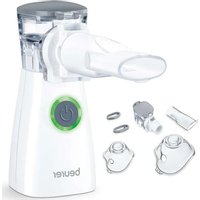
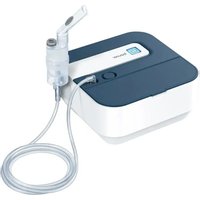
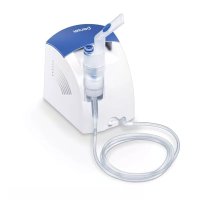
IH 57 - Inhaler BEURER - Free user manual and instructions
Find the device manual for free IH 57 BEURER in PDF.

User questions about IH 57 BEURER
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Inhaler in PDF format for free! Find your manual IH 57 - BEURER and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. IH 57 by BEURER.
USER MANUAL IH 57 BEURER
natural_image
White beurer device with clear blue cap and control knob, no visible text or symbols on device bodynatural_image
Illustration of an open book with a geometric window and horizontal lines, no text or symbols presentEN Unfold page 3 before reading the instructions for use.
Software Version V1.0.2.3
Read these instructions for use carefully and keep them for later use. Make them accessible to other users and note the information they contain.
CONTENTS
- Included in delivery 34
- Signs and symbols ......35
- Intended use....37
- Warnings and safety notes ....39
- Device and atomiser set description .....43
- Initial use 44
-
Usage 45
-
Cleaning and maintenance....50
- Atomiser set and replacement parts ..... 55
- What if there are problems? 56
- Disposal....57
- Technical specifications .... 58
- Guarantee / Service 61
1. INCLUDED IN DELIVERY
Check that the exterior of the cardboard delivery packaging is intact and make sure that all contents are present. Before use, ensure that there is no visible damage to the device or atomiser set (mouthpiece, masks) and that all packaging material has been removed. If you have any doubts, do not use the device and contact your retailer or the specified Customer Services address.
- Nebuliser
- Mesh atomiser with medication container (hereinafter referred to as “medication container”)
- Mouthpiece
- PVC adult mask
- PVC children's mask
- Micro USB cable
- Storage bag
• These instructions for use
2. SIGNS AND SYMBOLS
The following symbols appear in these instructions for use.
| WarningWarning indicating a risk of injury or damage to your health | |
| ImportantSafety note indicating possible damage to the device/accessory | |
| ImportantIndicates that caution is necessary when operating devices or controls in the vicinity of this symbol, or that the current situation requires the operator to be vigilant or the operator to take action to avoid undesirable consequences | |
| Product informationNote on important information |
The following symbols are used on the packaging and on the type plate for the device and atomiser set.
 | Follow the instructionsRead the instructions before starting work and/or operating devices or machines |  | CE labellingThis product meets the requirements of the applicable European and national directives. |
| Isolation of applied parts, type BFGalvanically isolated applied part (F stands for “floating”); meets the requirements for leakage currents for type B |  | Marking to identify the packaging material.A = material abbreviation,B = material number:1-6 = plastics,20-22 = paper and cardboard | |
| Manufacturer On/Off |  | ||
| MD | Medical device Article number | [SZC4] | |
| SN | Serial number | [73BH] | Unique device identifier (UDI)Identifier for unique product identification |
| IP 22 | Protected against foreign objects ≥12.5 mm and against water dripping at an angle | Storage/Transport [C4DS] [C4DS] | Permissible storage and transport temperature and humidity |
| Dispose of packaging in an environmentally friendly manner |  | Reuse on a single patient | |
| Disposal in accordance with the Waste Electrical and Electronic Equipment EC Directive (WEEE) |  | Protection class II deviceThe device is double-insulated and is, therefore, in protection class 2 | |
 | Do not dispose of batteries containing harmful substances with household waste |  | Date of manufacture |
3. INTENDED USE
Purpose
Nebulisers (including compressor, ultrasonic and mesh nebulisers) are medical devices for the nebulisation of liquids and liquid medication. This device produces aerosols by combining an oscillating mesh with holes and a liquid medication.
The aerosol treatment is suitable for treating the upper and lower airways.
By nebulising and inhaling the medication prescribed/recommended by your doctor, you can prevent diseases affecting the airways, or in the case that you contract such an illness, you can alleviate symptoms and speed up your recovery.
Target group
The nebuliser is only intended for medical care in a home environment.
The nebuliser is not intended for health care in professional facilities.
The nebuliser can be used on persons over 2 years of age under supervision; it can be used for self-treatment by persons over 12 years of age.
The shape and size of the person's face determines whether or not the device can be used under supervision. As such, it may be possible to use the device under supervision on someone who is younger, or conversely it may not be possible to use the device until they are older. When using a mask to inhale, take care to ensure the mask fits well and the eyes are unobstructed.
Clinical benefits
Inhalation is the most effective way to administer medication for most respiratory system conditions.
The benefits are:
- The medication is carried directly to the target organs
- The local bioavailability of the medication is significantly increased
• Systemic diffusion is reduced substantially - Only very low doses of the medication are required
- Quick and effective therapeutic effect
- Side effects are significantly reduced compared to systemic administration
• Humidification of the airways - Loosening and making (bronchial) secretions more liquid
- Releasing bronchospasms (spasmolysis)
- Relieving swollen and inflamed bronchial mucosa
- Coughing up secretions
- Fighting viruses that affect the upper and lower airways
Indication
The nebuliser can be used for diseases of the upper and/or lower airways.
Examples of upper respiratory system diseases include:
- Nasal mucosal inflammation
• Allergic nasal mucosal inflammation - Nasal sinus infection
- Pharyngitis
• Laryngitis
Examples of lower respiratory system diseases include:
- Bronchial asthma
- Bronchitis
• COPD (chronic obstructive pulmonary disease) - Bronchiectasis
- Acute tracheobronchitis
- Cystic fibrosis
- Pneumonia

CONTRAINDICATIONS
- The atomiser is not intended for the treatment of life-threatening conditions.
- This device must not be used by children under the age of 12 and by people with reduced physical, sensory (e.g. reduced sensitivity to pain) or mental abilities or a lack of experience or knowledge, unless they are supervised or have been instructed on how to use the device safely, and are fully aware of the resulting risks of use.
- Do not use the device on persons who are ventilated and/or unconscious.
- Check whether there are contraindications for use with the usual systems for aerosol treatment on the medication instruction leaflet.
- If the device does not work properly, or if you feel unwell or experience pain, stop using it immediately.
4. WARNINGS AND SAFETY NOTES

GENERAL WARNINGS
- The atomiser has no significant effect on the efficacy and safety of the medication administered and is not intended for the treatment of life-threatening conditions.
- Once it has been properly prepared, the device can be used again. The preparation work includes replacing all components, including the atomiser. We recommend that you replace the medication container and other components after one year at the latest. This replacement period is based on the device being used for max. 3 cycles per day and for ≤ 10 min per cycle, and provided it is regularly disinfected and cleaned in accordance with the instructions for use (8. Cleaning and maintenance). For hygiene reasons, it is essential that every user uses their own components.
- You should always follow the instructions of your doctor or pharmacist regarding the type of medication to use, the dosage, and the frequency and duration of inhalation. In the event of use with medication/nebulisation of medication, the conditions and restrictions applicable to such medication
must be adhered to. Please note: For treatment, only use the medications indicated by your doctor according to the particular diagnosis.
- The device is not a substitute for medical consultation and treatment. Always consult your doctor first in the event of pain or illness.
- If you have health concerns of any kind, consult your GP!
- If the liquid you wish to use is not compatible with steel, PP, PC, silicone and PVC, do not use this liquid with our nebuliser. If the information provided with the liquid does not indicate whether the liquid is compatible with these materials, please contact the liquid manufacturer. Examples of compatible liquids include salbutamol solution and sodium chloride solution.
- Please note the general hygiene measures when using the nebuliser.
- Before use, ensure that there is no visible damage to the device or components. If you have any doubts, do not use the device and contact your retailer or the specified Customer Services address.
• Changes to the device and the components are not permitted.
- Do not use any additional parts that are not recommended by the manufacturer.
- The nebuliser may only be operated with compatible Beurer atomisers and with the appropriate Beurer components. The use of atomisers and components made by other companies may result in less efficient treatment and could damage the device.
- Keep the device away from your eyes when it is in use, as the mist of medication could be harmful.
- Never use the device near flammable gases, oxygen or nitrogen oxide.
- Store the device and components out of the reach of children and pets.
- Keep packaging material away from children (risk of suffocation).
- The device must be switched off and the plug of the micro USB cable pulled out before every cleaning and/or maintenance procedure.
- If the device was at the maximum storage temperature, a period of 4 hours is required before using it in order for the device to cool down to a level where it is ready for the intended use. Likewise, if the device was at the minimum storage temperature, a period of 4 hours is required before using it until the device is ready for the intended use.
- Never submerge the device in water and do not use it in the bathroom. Under no circumstances may liquid enter the device.
- If the device is dropped, exposed to high levels of moisture or suffers any other damage, it must no longer be used. If in doubt, contact Customer Services or the retailer.
- Power cuts, sudden interference or other unfavourable conditions could lead to the device becoming inoperable. We, therefore, recommend that you obtain a replacement device or medication (the latter should be agreed with your doctor).
- To avoid the risk of entanglement and strangulation, store the cable out of the reach of small children.
- The device must only be connected to the mains voltage that is specified on the type plate.
- Never touch the micro USB cable with wet hands, as you could get an electric shock.
- Do not pull the mains adapter out of the socket using the micro USB cable.
- Do not crush or bend the micro USB cable, pull it over sharp-edged objects or leave it dangling down, and protect it from sources of heat.
- We recommend that the micro USB cable is completely unrolled to avoid dangerous overheating.
- If the micro USB cable of this device is damaged, it must be disposed of. Please contact Customer Services or the retailer.
- If the device is opened, there is a risk of electric shock. The power supply network not disconnected unless the adapter is unplugged (and the micro USB cable has no other power connection).

GENERAL PRECAUTIONS
- Never store the device (or the micro USB cable) near to sources of heat.
- Do not use the device in a room in which a spray has previously been used. Air the room before carrying out the treatment.
- Never use the device if it is making an abnormal sound.
- Store the device in a location protected from climatic effects. The device must be stored in the environmental conditions specified.
- Do not operate the device near electromagnetic transmitters.
-
Protect the device from severe knocks.
-
After the battery has been successfully charged, disconnect the device from the mains.
- Should you require an adapter or extension lead, it must meet the applicable safety requirements. The power limit and the maximum output specified on the adapter must not be exceeded.
Prior to initial use
- Remove all packaging material before using the device.
- Protect the device from dust, dirt and humidity and never cover the device while it is in use.
- Do not operate the device in a very dusty area.
- Switch the device off immediately if it is faulty or not working properly.
- The manufacturer is not liable for damage resulting from improper or incorrect use.
Repairs
- Under no circumstances should you open or repair the device yourself. If you do so, it may no longer function correctly. Failure to comply with this instruction will void the guarantee.
- The device is maintenance-free.
- For repairs, please contact Customer Services or an authorised retailer.
Measures for handling batteries

- If fluid from a battery cell comes into contact with your skin or eyes, wash the affected areas with water and seek medical assistance.
- Choking hazard! Small children could swallow and choke on batteries. So, keep batteries out of the reach of small children!
- Protect batteries from excessive heat.
- Risk of explosion! Never throw batteries into a fire.
- Do not disassemble, split or crush the batteries.
- Batteries must be charged correctly prior to use. The instructions from the manufacturer and the specifications in these instructions for use regarding correct charging must be observed at all times.
- Fully charge the battery prior to initial use (see “Warnings and safety notes” section).
- To obtain the maximum battery life, fully charge the battery at least 2 times a year.


NOTES ON ELECTROMAGNETIC COMPATIBILITY
- The device is suitable for use in all environments listed in these instructions for use, including domestic environments.
- The use of the device may be restricted if electromagnetic disturbance occurs. This could result in issues such as the failure of the device.
- Avoid using this device directly next to other devices or stacked on top of other devices, as this could lead to faulty operation. If, however, it is necessary to use the device in the manner stated, this device as well as the other devices must be monitored to ensure they are working properly.
- The use of an atomiser set other than that specified or provided by the manufacturer of this device can lead to an increase in electromagnetic emissions or a decrease in the device's electromagnetic immunity; this can result in faulty operation.
- Keep portable RF communication devices (including peripheral equipment, such as antenna cables or external antennas) at least 30 cm away from all device parts, including all cables included in delivery.
- Failure to comply with the above could impair the performance of the device.
5. DEVICE AND ATOMISER SET DESCRIPTION
The associated drawings are shown on page 3.
Device overview
1 Cover for medication container
2 Mesh atomiser with medication container (hereinafter referred to as "medication container")
3 Housing
4 On/Off switch
5 LED indicator
6 Button for unlocking the medication container ("PRESS")
7 Micro USB port
8 Micro USB cable
Atomiser set overview
9 PVC adult mask
10 PVC children's mask
11 Mouthpiece
Atomiser set
Only use the atomiser set recommended by the manufacturer. If this is not respected, the device may not function safely.

IMPORTANT
Leakage protection
When pouring the medication into the medication container, ensure that you only fill it up to the maximum mark (8 ml). It is recommended to fill the device up to between 0.5 and 8 ml.
The substance to be nebulised must be contact with the mesh for nebullisation to take place. If this is not the case, the nebulisation will stop automatically.
So, please try to hold the device as vertically as possible.
6. INITIAL USE
Before using the device for the first time

NOTICE
- Clean and disinfect the medication container 2 and atomiser set before using them for the first time. See section “Cleaning and maintenance” on this process.
Assembly
Take the device out of the packaging.
If the medication container 2 has not been assembled, slide it onto the housing 3 horizontally from the front. A
Charging the nebuliser battery
To charge the nebuliser battery, proceed as follows:
- Connect the micro USB cable 8 included in delivery to the micro USB port 7 on the bottom of the nebuliser and a USB mains adapter (not included in delivery; the mains adapter should comply with protection class 2 and have been tested in accordance with the European standard EN 60601-1). Insert the USB mains adapter into a suitable socket. B Lie the nebuliser flat on its back during the charging process.
- While charging, the LED indicator 5 will light up continuously orange. As soon as the battery is fully charged, the LED indicator will stay on green.
- It usually takes approx. 4 hours to fully charge the battery.
- As soon as the battery is completely charged, remove the micro USB cable 8 with the USB mains adapter from the socket and the micro USB port 7 on the nebuliser.
- When the battery is fully charged, the battery life is approx. 4 hours. If the LED indicator 5 lights up orange while you are using the device, this indicates that the battery level is low.
- The way in which a low battery level is indicated depends on how you are using the device. If you have opted for a high nebulisation rate, the LED indicator 5 will stay on orange; if you have chosen a low nebulisation rate, the LED indicator 5 will flash orange slowly.
- The nebuliser cannot be used while it is being charged.
7. USAGE
7.1 Preparing the nebuliser
- For hygiene reasons, it is essential to clean and regularly disinfect the medication container 2 and the atomiser set after each treatment. For this purpose, see section “Cleaning and maintenance”.
- If the therapy involves inhaling several different medications one after the other, please be aware that the medication container 2 must be rinsed under warm tap water after each use. See section "Cleaning and maintenance" on this process.
7.2 Filling the medication container
- Open the cover of the medication container 2 by lifting up the flap 1 on the rear C and fill the medication container with an isotonic saline solution or pour the medication straight in. Avoid overfilling.
- The maximum recommended filling quantity is 8 ml.
- Use medication only as instructed by your doctor and ask about the appropriate inhalation period and quantity for you.
- If the prescribed quantity of medication is less than 0.5 ml, it must be topped up with isotonic saline solution. Dilution is also necessary with viscous medications (viscosity < 3). Please observe the instructions of your doctor in this case, too.
7.3 Closing the medication container
- Close the cover of the medication container 2 by pressing the flap on the rear down and locking it. D
7.4 Connecting the atomiser set
- Securely connect the part of the atomiser set that you would like to use (mouthpiece 11, adult mask 9 or children's mask 10) to the mounted medication container 2 E. If you are using the adult mask 9 or children's mask 10, you may attach one of the straps included. Ensure that you securely fix the strap on the mask with a knot.
- Now move the device to your mouth and put your lips firmly around the mouthpiece. When using the mask(s), please place it over your nose and mouth.
- Ensure that you have added fluid before switching on the device. Start the device by pressing the On/Off switch 4.
- If the spray mist is flowing out of the device and the LED indicator 5 is lit up continuously blue, the device is operating correctly.

NOTICE
The most effective way of nebulising is by using the mouthpiece. Nebulisation using a mask is only recommended if it is not possible to use a mouthpiece (e.g. for children who are not yet able to inhale using a mouthpiece). When using a mask to inhale, take care to ensure the mask fits well and the eyes are unobstructed.
The device medication flow rate is set to “high” in the factory settings. You can adjust the medication flow rate during use by pressing and holding the On/Off switch 4 for 3 seconds until the LED indicator 5 flashes blue. You can switch from the high to the low nebulisation rate and back again. If the LED indicator 5 lights up continuously blue, this indicates a high nebulisation rate of ≥ 0.4 ml/min; if the LED indicator 5 flashes blue slowly, this indicates a low nebulisation rate of ≥ 0.2 ml/min.
Adjust the medication flow rate according to your requirements.
7.5 Inhaling correctly
Breathing technique
- It is important to use the correct breathing technique to ensure that the particles are distributed as widely as possible throughout the airways. To ensure that the particles reach your airways and lungs, you must breathe in slowly and deeply, then hold your breath briefly (5 to 10 seconds) and then breathe out quickly.
- Nebulisers should only be used for the treatment of respiratory diseases after consultation with your doctor. They will advise you about choosing the medications suitable for inhalation therapy, and at what dose and how to use them.
- Certain medications are only available with a prescription.

NOTICE
You should hold the device as vertically as possible. However, a slight angle will not affect usage, as the container is leak proof. For the device to work properly, ensure when using it that the medication is in contact with the mesh. If the device is tipped gently by 45 degrees with a maximum volume of 8 ml of liquid, the device will work as before.

IMPORTANT
For health reasons, essential oils, cough syrups, gargling solutions and drops to be used as a rub or in a steam bath are totally unsuitable for inhalation with a nebuliser. These additives are also often viscous and can impair the correct functioning of the device and therefore the effectiveness of usage in the long term.
For individuals with a hypersensitive bronchial system, medications containing essential oils may in some cases cause an acute bronchospasm (a sudden cramp-like restriction of the bronchi with shortness of breath). Talk to your doctor or pharmacist about this.
7.6 Stopping inhalation
a) Manually ending nebulisation:
- When you have finished the treatment, switch off the device using the On/Off switch 4.
- The LED indicator 5 will go off.
b) Automatically ending nebulisation:
- If the inhalant has been nebulised, the LED indicator 5 will flash blue quickly and the device will switch itself off. For technical reasons, a small amount of medication will remain in the medication container. Do not use this.
7.7 Cleaning
See section "Cleaning and maintenance".
7.8 Automatic switch-off
The device has an automatic switch-off function. If there is only a very small amount of medication or fluid left or if the substance to be nebulised is no longer in contact with the mesh, the device will switch off automatically to avoid causing damage to the mesh. If the nebulisation stops automatically, the automatic switch-off has been activated. The LED indicator 5 will flash blue quickly and the device will then switch off. After 15 minutes of nebulisation time, the device switches off automatically.
Do not operate the device when the medication container 2 is empty. The device detects when there is no medication in the medication container and will switch off automatically.
| Colour of the LED indicator | Explanation |
Green | Fully charged:As soon as the battery is fully charged, the LED indicator will light up continuously-ly green. |
Orange | Charging process:While the battery is being charged, the LED indicator will light up orange continuously.Low battery level:If the LED indicator lights up orange whilst you are using the device, this indicates that the battery level is low.At a high nebulisation rate: it will light up orange continuouslyAt a low nebulisation rate: it will flash orange slowlySelf-cleaning mode:If you press and hold the On/Off switch for 3 seconds while the device is switched off, the LED indicator will flash orange quickly and the device will go into self-cleaning mode. |
Blue | Operation and medication flow:The blue LED indicator indicates that the device is working correctly.High nebulisation rate of ≥ 0.4 ml/min: the LED indicator will light up blue continuouslyLow nebulisation rate of ≥ 0.2 ml/min: the LED indicator will flash blue slowlyToo little medication and automatic switch-off:If the inhalant has been nebulised, the LED indicator will flash blue quickly and the device will switch itself off. |
8. CLEANING AND MAINTENANCE

WARNING
Follow these hygiene instructions to avoid health risks.
- The medication container 2 and atomiser set are intended for multiple use. Please note that different areas of use involve different cleaning and hygiene preparation requirements.

NOTICE
- Do not clean the mesh or the atomiser set mechanically using a brush or similar device, as this could cause irreparable damage and optimum treatment results may no longer be guaranteed.
- Please consult your doctor about the additional requirements for hygiene preparation (hand care, handling of medication/inhalation solutions) for high-risk groups (e.g. patients with cystic fibrosis).
- The cleaning instructions mention water later on; it should be as soft as possible. Distilled water is suitable for all cleaning steps except for self-cleaning.
Disassembling
- Hold the device with one hand. The button 6 on the rear of the device should be facing you. Press the button 6 using your thumb and horizontally pull off the medication container 2 forwards using your free hand. F
- Remove the mouthpiece 11 or the mask (9 or 10) from the medication container 2.
- Open the cover of the medication container 2 by lifting up the flap 1 on the rear G.
- Reassembly is carried out in reverse order.
Cleaning
After each use, the medication container 2 and the parts of the atomiser set that were used, such as the mouthpiece 11, mask (9 or 10), must be cleaned for approx. 5 minutes using (ideally distilled) warm water at a max. temperature of 40°C. To clean the medication container, please follow the self-cleaning instructions described below. Dry the parts carefully using a soft cloth. Put the parts together again once they are completely dry and place them in a dry, sealed container or disinfect them.
- Please perform the self-cleaning function afterwards (see “Self-cleaning function” section).

IMPORTANT
Never hold the whole device under running water to clean it.
When cleaning, ensure that any residue is removed and dry all parts carefully.
Never use any substances for cleaning that could potentially be toxic if they were to come into contact with the skin or mucous membranes, or if they were swallowed or inhaled.
If required, clean the housing of the device using a slightly damp cloth, which you can moisten with a mild soapy solution.
- If there is any residue from medical solutions or contamination on the silver/gold-coloured contacts of the device or medication container 2, clean with a cotton bud moistened with ethyl alcohol.
- Also clean the outer sides of the mesh and medication container with a cotton bud moistened with ethyl alcohol.
- The device must be switched off and allowed to cool down each time before cleaning.
- Do not use any abrasive cleaning products.

IMPORTANT
- Make sure that no water gets inside the device!
- Do not clean the device or atomiser set in the dishwasher!
- Do not allow water to splash the device. Only operate the device if it is completely dry.

IMPORTANT
We recommend carrying out inhalation continuously since the inhalant may clog the medication container 2 if the treatment is paused for a significant time, and the effectiveness of the administration may be impaired as a result.
Self-cleaning function
To completely remove medication residue and any blockages, you can use the device's self-cleaning function.
To do this, proceed as follows:
- Disassemble the device as described above.
-
Open the cover of the medication container 1 and pour out the remaining fluid.
-
Pour a sufficient amount (3-6 ml) of tap water (soft water) into the medication container 2. Then close the cover. Shake the container slightly so that the remaining fluid mixes properly with the cleaning fluid.
-
Open the cover of the medication container, pour out all the liquid and then add enough tap water (soft water) again.
-
Place the medication container on the housing 3.
-
Press and hold the On/Off switch 4 for 3 seconds when the device is switched off. The LED indicator 5 will then flash orange quickly and the device will go to self-cleaning mode. The device will automatically run for 3 minutes. Please make sure there is sufficient fluid in the medication container.
Disinfection
Please carefully observe the points below when disinfecting your medication container 2 and the atomiser set. Regular disinfection of the individual parts is recommended.
First, clean the medication container 2 and atomiser set as described in the “Cleaning” section. Then continue with the disinfection.

IMPORTANT
Make sure that you do not touch the mesh if possible, as this can destroy it.
Disinfection with ethyl alcohol (70 - 75%)
- Pour 8 ml of ethyl alcohol into the medication container 2. Close the container. Leave the alcohol in the container for at least 10 minutes.
- For better disinfection, shake the unit gently back and forth.
- Once this is complete, pour the ethyl alcohol out of the container.
- Repeat the process, but this time using water.
- Position the medication container 2 so that you can drip a few drops of ethyl alcohol onto the mesh from the outside. Leave this to work for 10 minutes.
- Finally, clean all parts again with water.
-
The same procedure is used for the mouthpiece 11 and the masks 9 10.
-
Then clean the masks 9 10 and the mouthpiece 11 with water.
- Then dry all parts thoroughly. See the section "Drying" for further details.
Disinfection with boiling water
- The medication container 2, the cover 1 and the mouthpiece can be placed in boiling water for 15 minutes in order to disinfect them. Ideally, distilled water should be used for this, to prevent contamination and limescale forming on the mesh. Contact between the parts and the hot base of the pan should be avoided.
- Then dry all parts thoroughly. See the section "Drying" for further details.
• The medication container 2 must not be placed in the microwave. - We guarantee 50 disinfection cycles with boiling water per medication container.
i NOTICE
The masks must not be placed in hot water!
- We recommend disinfecting the masks using 70% - 75% ethyl alcohol.
Drying
- Dry the parts carefully using a soft cloth.
- Gently shake the medication container 2 a few times so that any water in the mesh is removed from the tiny holes.
- Place the individual parts on a clean, dry and absorbent surface and leave them to dry completely (at least 4 hours).
i NOTICE
Please ensure that the parts are completely dry after cleaning, otherwise the risk of bacterial growth is increased.
Put the parts together again when they are completely dry and place them in a dry, sealed container.
Durability of materials
- As with any plastic parts, the medication container 2 and atomiser set are subject to a certain amount of wear and tear when used and hygienically prepared on a frequent basis. Over time, this can lead to a change in the aerosol, which can reduce the effectiveness of the treatment. We therefore recommend that you replace the medication container 2 and atomiser set after one year at the most.
Storage
- Do not store in damp conditions (such as in a bathroom) and do not transport with any damp items.
- When storing and transporting, protect from prolonged direct sunlight.
9. ATOMISER SET AND REPLACEMENT PARTS
The atomiser set parts and replacement parts are available from the service address indicated (in the service address list). Please state the corresponding order number.
| Name Material REF | ||
| Mouthpiece (PP), adult mask (PVC), children's mask (PVC), mesh atomiser with medication container (PC) | PP/PVC/PC 601 | .43 |
- WHAT IF THERE ARE PROBLEMS?
| Problems/questions Possible cause/remedy | |
| The nebuliser produces no or too little aerosol. | 1. Too little medication in the medication container2. |
| 2. Nebuliser not held vertically. | |
| 3. Unsuitable medication fluid added for nebulisation (e.g. too viscous. Viscosity must not be greater than 3. Check with the pharmacy regarding viscosity.) The medication fluid should be prescribed by a doctor. | |
| 4. The battery is flat. Charge it. | |
| The output is too low. | There are air bubbles in the medication container, which are preventing continuous contact with the mesh. Please check this and remove any air bubbles. |
| 2. Particles on the mesh are hindering output. In this case, you should pause the inhalation process and remove the mask or mouthpiece. Then clean and disinfect the medication container (see “Cleaning and maintenance” chapter). | |
| 3. The mesh is worn. Replace the medication container2. | |
| Which medications are suitable for inhaling? | Naturally, only your doctor can decide which medication should be used for treating your condition.Please consult your doctor about this.With the IH 57, you can nebulise medications with a viscosity of less than 3. Medications containing oil (particularly essential oils) should not be used, as these attack the material of the medication container, which can lead to faults. |
| Problems/questions | Possible cause/remedy |
| There is inhalation solution residue in the medication container2. | This is normal and is due to technical reasons. Stop the inhalation as soon as the atomiser starts to make a notably different sound or the device switches off automatically due to a lack of inhalant. |
| What should be taken into account when using the device with infants and children? | 1. For infants, the mask should cover the nose and mouth to ensure effective inhalation.2. For children, the mask should also cover both the nose and mouth. It makes no sense to use the nebuliser on someone who is sleeping, since not enough of the medication will reach the lungs.Notice: Inhalation should only be carried out under the supervision of an adult and with their assistance and the child should not be left alone. |
| Should each person have their own atomiser set? | Yes, this is absolutely essential for hygiene reasons. |
11. DISPOSAL
Battery disposal
- Empty, completely flat batteries must be disposed of in specially designated collection boxes, at recycling points or at electronics retailers. You are legally required to dispose of the batteries.
- The codes below are printed on batteries containing harmful substances:
Pb = battery contains lead
Cd = battery contains cadmium
Hg = battery contains mercury
General disposal
For environmental reasons, do not dispose of the device in household waste at the end of its service life. Dispose of the device at a suitable local collection or recycling point in your country. Dispose of the device in accordance with EC Directive – WEEE (Waste Electrical and Electronic Equipment). If you have any questions, please contact the local authorities responsible for waste disposal.

- TECHNICAL SPECIFICATIONS
| Type | IH 57 |
| Dimensions (LxWxH) 46 x 78 x 108 mm | |
| Weight 109 g ± 5 g | |
| Input 5 V DC; 1 A | |
| Energy consumption < 1.5 W | |
| Filling volume min. 0.5 ml | max. 8 ml |
| Medication flow rate Low nebulisation rate: ≥ 0.2 ml/minHigh nebulisation rate: ≥ 0.4 ml/min | |
| Vibration frequency 110 kHz ± 10 kHz | |
| Operating conditions Temperature: +5°C to +40°CRelative humidity: 15-90% non-condensingAmbient pressure: 700 to 1060 hPa | |
| Storage and transportation conditions | Temperature: -25°C to +70°CRelative humidity: ≤ 90% non-condensing |
| Expected service life of the device | Information on the service life of the product can be found on the homepage |
| Battery: | |
| Capacity | 1,800 mAh |
| Nominal voltage | 3.7 V |
| Type designation | Lithium-ion |
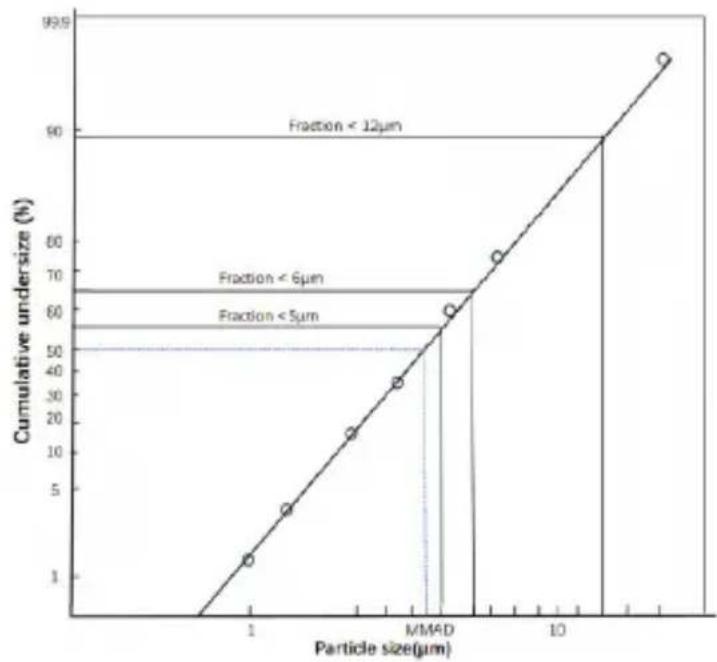
| Aerosol values according to EN 27427:2019 based on adult breathing patterns with salbutamol: | · Aerosol delivery: 0.90 ± 0.03 ml· Aerosol delivery rate: 0.21 ± 0.01 ml/min· Fill volume dispensed in percent per min.: 28.71 ± 3.12% · Residual volume: 0.004 ± 0.001 ml· Particle size (MMAD): 4.519 ± 0.09 μm· GSD (geometrical standard deviation): 2.054 ± 0.04 · RF (respirable fraction < 5 μm): 55.04 ± 1.21% · Large particle range (> 5 μm): 44.96 ± 1.21% · Medium particle range (2 to 5 μm): 41.03 ± 0.46% · Small particle range (< 2 μm): 14.01 ± 1.03% |
Software version V1.0.2.3
The serial number is located on the device.
Subject to technical changes.
(see "Cleaning and maintenance" chapter).
Particle size diagram
line
| Particle size(μm) | Cumulative undersize(%) | | ----------------- | ---------------------- | | 1 | 1 | | 2 | 3 | | 3 | 5 | | 4 | 7 | | 5 | 9 | | 6 | 11 | | 7 | 13 | | 8 | 15 | | 9 | 17 | | 10 | 19 | | 11 | 21 | | 12 | 23 | | 13 | 25 | | 14 | 27 | | 15 | 29 | | 16 | 31 | | 17 | 33 | | 18 | 35 | | 19 | 37 | | 20 | 39 | | 21 | 41 | | 22 | 43 | | 23 | 45 | | 24 | 47 | | 25 | 49 | | 26 | 51 | | 27 | 53 | | 28 | 55 | | 29 | 57 | | 30 | 59 | | 31 | 61 | | 32 | 63 | | 33 | 65 | | 34 | 67 | | 35 | 69 | | 36 | 71 | | 37 | 73 | | 38 | 75 | | 39 | 77 | | 40 | 79 | | 41 | 81 | | 42 | 83 | | 43 | 85 | | 44 | 87 | | 45 | 89 | | 46 | 91 | | 47 | 93 | | 48 | 95 | | 49 | 97 | | 50 | 99 |Measurements were performed using a salbutamol solution with a “Next Generation Impactor” (NGI). The diagram may, therefore, not be applicable to suspensions or highly viscous medications. You can obtain more detailed information from the manufacturer of your medication.

NOTICE
If the device is not used according to the specifications, it may not work correctly! We reserve the right to make technical changes to improve and develop the product.
This device and its atomiser set comply with the European standard EN 60601-1-2 (Group 1, Class B, in accordance with CISPR 11, IEC 61000-3-2, IEC 61000-3-3, IEC 61000-4-2, IEC 61000-4-3,
IEC 61000-4-4, IEC 61000-4-5, IEC 61000-4-6, IEC 61000-4-7, IEC 61000-4-8, IEC 61000-4-11) and is subject to particular precautions with regard to electromagnetic compatibility.
13. GUARANTEE / SERVICE
More information on the guarantee and guarantee conditions can be found in the guarantee leaflet supplied.
Notification of incidents
For users/patients in the European Union and identical regulation systems (EU Medical Device Regulation (MDR) 2017/745), the following applies: If a major incident occurs during or through use of the product, notify the manufacturer and/or their representative of this as well as the respective national authority of the member state in which the you are located.
FRANÇAIS

• BPCO (broncho-pneumopathie chronique obstructive)
- Pneumonie

CONTRE-INDICATIONS
Mesures relatives aux piles

Vers. software V1.0.2.3