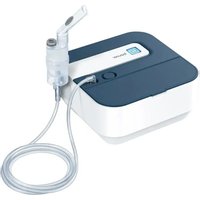
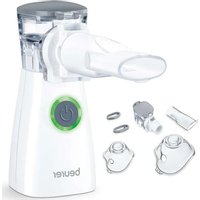
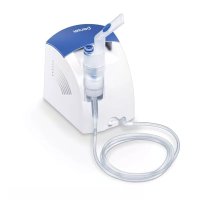
IH 47 - Inhaler BEURER - Free user manual and instructions
Find the device manual for free IH 47 BEURER in PDF.

User questions about IH 47 BEURER
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Inhaler in PDF format for free! Find your manual IH 47 - BEURER and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. IH 47 by BEURER.
USER MANUAL IH 47 BEURER
EN Unfold page 3 before reading the instructions for use.
Read these instructions for use carefully. Follow the warnings and safety notes. Keep these instructions for use for future reference. Make the instructions for use accessible to other users. If the device is passed on, provide the instructions for use to the next user as well.
CONTENTS
- Included in delivery 36
- Signs and symbols 37
- Intended purpose 39
4.Warnings and safety notes.. 41 - Device description 46
- Initial use 47
-
Usage 48
-
Cleaning and maintenance 53
- Accessories and/or replacement parts 58
10.What if there are problems? 58 - Disposal 60
- Technical specifications 60
13.Guarantee/Service. 63
1. INCLUDED IN DELIVERY
Check that the exterior of the cardboard delivery packaging is intact and make sure that all contents are present. Before use, ensure that there is no visible damage to the device or atomiser set (mouthpiece, masks) and that all packaging material has been removed. If you have any doubts, do not use the device and contact your retailer or the specified Customer Service address.
- Nebuliser
- Mesh atomiser with medication container (hereinafter referred to as "medication container")
- Mouthpiece
Adult mask made from PVC
Children's mask made from PVC
USB-C cable - Storage bag
These instructions for use
2. SIGNS AND SYMBOLS
The following symbols are used on the device, in these instructions for use, on the packaging and on the type plate for the device:

WARNING
Indicates a potentially impending danger. If it is not avoided, there is a risk of death or serious injury.

CAUTION
Indicates a potentially impending danger. If it is not avoided, slight or minor injuries may result.
NOTICE
Indicates a potentially harmful situation. If it is not avoided, the device or something in its vicinity may be damaged.
The following symbols are used on the packaging and on the type plate for the device and atomiser set:
| Observe the instructions Read the instructions before starting work and/or operating devices or machines | Separate the product and packaging materials and dispose of them in accordance with local regulations. | ||
| B A | Identification of the packaging material. A = material code, B = material number: 1-7 = plastics, 20-22 = paper and cardboard | CE | CE labelling This product satisfies the requirements of the applicable European and national directives. |
| Applied part, type BF Importer | |||
| Manufacturer Date of manufacture | |||
| MD | Medical device Article number | REF | |
| SN | Serial number | UDI | Unique device identifier (UDI) to uniquely identify the product |
| IP22 | The device is protected against foreign objects ≥ 12.5 mm and against dripping water falling at an angle | # | Type number |
| Air humidity range Temperature range | |||
| Atmospheric pressure limit Protection class | device |
| CH REP | Swiss authorised representative Single patient multiple use | |
| Pb Ca Hg | Do not dispose of batteries containing harmful substances with household waste | The electronic device must not be disposed of with household waste |
| --- | Direct current Importer |
3. INTENDED PURPOSE
Intended use
Nebulisers (including compressor, ultrasonic and mesh nebulisers) are medical devices for the nebulisation of liquids and liquid medication (aerosols). This device produces aerosols by combining an oscillating mesh with holes and a liquid medication.
The aerosol treatment is suitable for treating the upper and lower airways.
By nebulising and inhaling the medication prescribed or recommended by your doctor, you can prevent diseases affecting the airways, or in the case that you contract such an illness, you can alleviate symptoms and speed up your recovery.
Intended users
The nebuliser is intended for use in the home healthcare environment, not in professional healthcare facilities.
The nebuliser can be used on anyone over 2 years of age under supervision; it can be used for self-treatment by anyone over 12 years of age.
The shape and size of the person's face determine whether or not the device can be used under supervision. As such, it may not be possible to use the device under supervision until they are older. When using a mask to inhale, take care to ensure the mask fits well and the eyes are unobstructed.
Clinical benefits
Inhalation is the most effective way to administer medication for most respiratory diseases.
It offers the following benefits:
- The medication is transported directly to the target organs
- The local bioavailability of the medication is significantly increased
Systemic diffusion is significantly reduced - Only very low doses of the medication are required
- Quick and effective therapeutic effect
- Side effects are significantly lower compared to systemic administration
- Humidifying the airways
- Loosening and making (bronchial) secretions more liquid
- Releasing bronchospasms (spasmolysis)
Relieving swollen and inflamed bronchial mucosa - Coughing up secretions
- Fighting viruses that affect the upper and lower airways
Indication
The nebuliser can be used for diseases of the upper and/or lower airways.
Examples of upper airway diseases include:
- Nasal mucosal inflammation
- Allergic nasal mucosal inflammation
- Nasal sinus infection
- Pharyngitis
Laryngitis
Examples of lower airway diseases include:
- Bronchial asthma
- Bronchitis
COPD (chronic obstructive pulmonary disease)
Bronchiectasis
- Acute tracheobronchitis
Cystic fibrosis
Pneumonia
Contraindications
- The atomiser is not intended for the treatment of life-threatening diseases.
- This device must not be used by children under the age of 12 and by people with reduced physical, sensory (e.g. reduced sensitivity to pain) or mental skills or a lack of experience or knowledge, unless they are supervised or have been instructed on how to use the device safely, and are fully aware of the consequent risks of use.
- Do not use the device on persons who are ventilated and/or unconscious.
- Check whether there are contraindications for use with the usual systems for aerosol treatment on the medication instruction leaflet.
- If the device does not work properly, or if you feel unwell or if you experience pain, stop using it immediately.
4. WARNINGS AND SAFETY NOTES
GENERALWARNINGS
- The atomiser has no significant effect on the efficacy and safety of the medication administered and is not intended for the treatment of life-threatening conditions. The atomiser is not suitable for use in anaesthesia or other ventilation systems.
- After proper preparation, the device can be reused. This preparation includes the replacement of all components, including the atomiser. We recommend that you replace the medication container and other components after one year at the latest. This recommendation for the timing of the replacement is based on the assumption that the device is used for a maximum of 2 cycles per day and
for ≤ 10 min per cycle and is regularly disinfected and cleaned in accordance with the instructions for use (8. Cleaning and maintenance). For hygiene reasons, it is essential that each user uses their own components.
- Always follow the instructions of your doctor or pharmacist regarding the type of medication to be used, the dosage and the frequency and duration of inhalation. When using with medications/medication nebulisation, observe the conditions and restrictions applicable to these medications. Notice: Use only the medications prescribed by your doctor for treatment according to the respective diagnosis.
- The device is not a substitute for medical advice and treatment. Always consult your doctor first in case of pain or illness.
If you have any health concerns, consult your GP. - Do not use liquids with our nebuliser that are not compatible with steel, PP, PC, silicone or PVC. If the information supplied with the liquid does not indicate its compatibility with these materials, contact the liquid manufacturer. Compatible liquids include, for example, albuterol solution.
- The particle size distribution curve, MMAD, aerosol delivery and/or aerosol delivery rate may change if a different medication, suspension, emulsion or highly viscous solution is used than the one referenced in the technical specifications for the aerosol values.
- Please observe the general hygiene measures when using the nebuliser.
- Before use, ensure that the device or its components show no visible damage. If you have any doubts, do not use the device and contact your retailer or the specified Customer Service address.
- No modifications may be made to the device or its components.
- Use only accessories recommended by the manufacturer.
- The nebuliser may only be operated with compatible Beurer atomisers and with the appropriate Beurer components. The use of nebulisers and components from other manufacturers may impair the effectiveness of the treatment and damage the device.
- Keep the device away from your eyes when it is in use, as the mist of medication could be harmful.
- Never use the device in the presence of flammable or explosive gases, oxygen or nitrous oxide.
-
Store the device and components out of the reach of children and pets.
-
Keep the packaging material away from children (risk of suffocation).
- The device must be switched off and the plug of the USB-C cable pulled out before every cleaning and/or maintenance.
- If the device has been stored at maximum storage temperature, it must cool for 4 hours before use until it is ready for its intended use. If the device has been stored at minimum storage temperature, it will also take 4 hours until the device is ready for its intended use.
- Never submerge the device in water and do not use it in the bathroom. Under no circumstances may liquid enter the device.
- If the device has been dropped, exposed to high levels of moisture or suffered any other damage, it must no longer be used. If in doubt, contact Customer Services or the retailer.
- Power outages, sudden interference or other unfavourable conditions may cause the device to stop working. We therefore recommend that you stock up on a replacement device or replacement medication (the latter should be agreed with your doctor).
- Keep the cable out of reach of small children to avoid the danger of constriction and strangulation.
- The device must only be connected to the mains voltage that is specified on the type plate.
- Never touch the USB-C cable with wet hands as this could cause an electric shock.
- Do not use the USB-C cable to pull the mains adapter out of the socket.
- Do not crush or bend the USB-C cable, pull it over sharp-edges or leave it dangling, and protect it from sources of heat.
We recommend that the USB-C cable be completely unwound to avoid dangerous overheating. - If the USB-C cable of this device is damaged, it must be disposed of. Please contact Customer Services or the retailer.
- There is a risk of electric shock when opening the device. The device is not disconnected from the power supply unless the adapter is unplugged (and the USB-C cable has no other power connection).
GENERAL PRECAUTIONS
- Never store the device (or the USB-C cable) near heat sources.
- Do not use the device in a room where a spray has previously been used. Ventilate the room before performing the treatment.
- Never use the device if it makes unusual noises.
- Store the device in a location protected against weather conditions. The device must be stored under the specified ambient conditions.
- Do not operate the device near electromagnetic transmitters.
- Protect the device from heavy impacts.
- Disconnect the device from the power supply after the battery is fully charged.
- If you need an adapter or extension cable, it must comply with the applicable safety requirements. The power limit and maximum power specified on the adapter must not be exceeded.
Prior to initial use
- Remove all packaging material before using the device.
- Protect the device from dust, dirt and humidity and never cover the device while it is in use.
- Do not operate the device in a very dusty area.
- Switch the device off immediately if it is faulty or not working properly.
- The manufacturer is not liable for damage resulting from improper or incorrect use.
Repairs
- Under no circumstances should you open or repair the device yourself. If you do so, it may no longer function correctly. Failure to comply with this instruction will void the guarantee.
- The device is maintenance-free.
- For repairs, please contact Customer Services or an authorised retailer.
Notes on handling batteries
WARNING
- Risk of explosion! Risk of fire! Failure to comply with the points mentioned can result in personal injury, overheating, leakage, venting, breakage, explosion, or fire.
- Always use the correct or supplied charging cable/charger/Mains adapter for charging.
- Avoid continuous charging or overcharging Unplug the charger when charging is complete.
- Charge the device under supervision, paying attention to any heat generated, deformation, or release of gases. If in doubt, stop charging.
- If batteries/charging cables/chargers are defective, stop using them and dispose of them properly as soon as possible (see chapter on disposal).
- Do not throw the device or batteries into a fire.
- Never forcibly discharge, heat, disassemble, open, crush, deform, encapsulate, modify, or knock the device or batteries.
- Never short-circuit batteries or the connections of the battery-powered device.
- Protect the device or batteries from direct sunlight, rain, heat, and water.
- Exposure of batteries to an environment with extremely high temperatures or an extremely low air pressure may result in explosion or leakage of flammable liquids and gases.
- If fluid from a battery comes into contact with your skin or eyes, wash the affected areas with water and seek medical assistance.
NOTICE
- This device contains a battery that is not replaceable. Once a battery has reached the end of its service life, the device must be disposed of properly (see chapter on disposal).
NOTES ON ELECTROMAGNETIC COMPATIBILITY
- The device is suitable for use in all environments listed in these instructions for use, including domestic environments.
- The use of the device may be restricted if electromagnetic disturbance occurs. This could result in issues such as the failure of the device.
- Avoid using this device directly next to other devices or stacked on top of other devices, as this could lead to faulty operation. If, however, it is necessary to use the device in the manner stated, this device as well as the other devices must be monitored to ensure they are working properly.
- The use of an atomiser set other than that specified or provided by the manufacturer of this device can lead to an increase in electromagnetic emissions or a decrease in the device's electromagnetic immunity; this can result in faulty operation.
- Keep portable RF communication devices (including peripheral equipment, such as antenna cables or external antennas) at least 30~cm away from all device parts, including all cables included in delivery.
- Failure to comply with the above could impair the performance of the device.
5. DEVICE DESCRIPTION
The associated drawings are shown on page 3.
Device overview
1 Lid for medication container
3 Housing
5 LED display
7 USB-C connection
Mesh atomiser with medication container (hereinafter referred to as "medication container")
4 On/Off control
6 Button for unlocking the medication container ("PRESS")
8 USB-C cable
Atomiser set overview
9 Adult mask made from PVC
Children's mask made from PVC
11 Mouthpiece
Atomiser set
Only use the atomiser set recommended by the manufacturer. If this is not respected, the device may not function safely.
CAUTION
Leakage protection
When pouring the medication into the medication container, ensure that you only fill it up to the maximum mark (10 ml). The recommended filling volume for the device is 0.5 to 10 ml.
The substance to be nebulised must be in contact with the mesh for nebulisation to take place. If this is not the case, the nebulisation will stop automatically.
Therefore, keep the device as vertical as possible.
6. INITIAL USE
Before using the device for the first time
NOTICE
- Clean and disinfect the medication container 2 and the atomiser set before first use. See the "Cleaning and maintenance" section in this regard.
Assembly
Remove the device from the packaging.
If the medication container 2 is not assembled, slide it horizontally A from above onto the housing 3.
Charging the nebuliser battery
Proceed as follows to charge the nebuliser battery:
- Connect the USB-C cable 8 included in delivery to the USB-C port 7 on the bottom of the nebuliser and a USB mains adapter (not included in delivery; the mains adapter must comply with protection class 2 and have been tested in accordance with the European standard EN 60601-1). Plug the USB mains adapter into a suitable socket. B
Lie the nebuliser flat on its back during the charging process. - While charging, the LED display will continuously light up orange. As soon as the battery is fully charged, the LED display will continuously light up green.
- It usually takes approximately 1.5 hours for the battery to be fully charged.
- As soon as the battery is fully charged, remove the USB-C cable with the USB mains adapter from the socket and the USB-C port on the nebuliser.
- When the battery is fully charged, the battery life is approximately 1 hour and 15 minutes. If the LED display 5 lights up orange while the device is in use, this indicates low a battery level.
- The type of low battery level display depends on how the device is used. If you have selected a high nebulisation rate, the LED display will be a continuous orange. If you have selected a low nebulisation rate, the LED display will slowly flash orange.
- The nebuliser cannot be used during the charging process.
7. USAGE
7.1 Preparing the nebuliser
-
For hygiene reasons, it is essential to clean and regularly disinfect the medication container 2 and the atomiser set after each treatment. For this purpose, see section "Cleaning and maintenance".
-
If the therapy involves inhaling several different medications one after the other, it should be noted that the medication container 2 must be rinsed under warm tap water following every usage. For this purpose, see section "Cleaning and maintenance".
7.2 Filling the medication container
- Open the lid of the medication container 2 by lifting up the flap 1 on the rear C and fill the medication container with an isotonic saline solution or pour the medication straight in. Do not overfill the medication container.
- The maximum recommended filling quantity is 10ml
- Use medication only as instructed by your doctor and ask about the appropriate inhalation duration and quantity for you.
- If the prescribed quantity of medication is less than 0.5ml , it must be topped up with isotonic saline solution. Dilution is also required for viscous medication (viscosity < 3 ). Please follow your doctor's instructions in this case as well.
7.3 Closing the medication container
- Close the lid of the medication container 2 by pressing the flap on the rear down and locking it. D
7.4 Connecting the atomiser set
- Securely connect the part of the atomiser set that you would like to use (mouthpiece 11, adult mask 9 or children's mask 10) to the mounted medication container 2 E. If you are using the adult mask 9 or children's mask 10, you may use one of the straps included. Ensure that you tie the straps securely to the mask with a knot.
- Now move the device to your mouth and put your lips firmly around the mouthpiece. When using the mask(s), please place it over your nose and mouth.
-
Ensure that you have filled it up with liquid before switching on the device. Start the device by pressing the On/Off control 4.
-
If the spray mist comes out of the device and the LED display 5 continuously lights up blue, the device is working correctly.
NOTICE
The most effective inhalation is when using the mouthpiece. Inhalation when using a mask is only recommended if the use of a mouthpiece is not possible (e.g. for children who are not yet able to inhale with a mouthpiece). When inhaling with a mask, make sure that the mask fits well and that the eyes are not covered.
The flow rate for the medication is set to high in the factory settings of the device. You can adjust the medication flow rate during use by pressing and holding the On/Off control 4 for 3 seconds until the LED display 5 flashes blue. You can switch from high to low nebulisation rate and back again. A continuously blue illuminated LED display 5 indicates a high nebulisation rate of ≥ 0.25ml / min ; a slowly flashing blue LED display 5 indicates a low nebulisation rate of ≥ 0.15ml / min .
Adjust the medication flow rate to your needs.
7.5 Inhaling correctly
Breathing technique
- It is important to use the correct breathing technique to ensure that the aerosols are distributed as widely as possible throughout the airways. For the aerosols to reach your airways and lungs, you need to breathe in slowly and deeply, then hold your breath briefly (5 to 10 seconds) and then exhale quickly.
- Only use nebulisers for the treatment of respiratory diseases after consulting your doctor. They will advise you on selecting suitable medications for inhalation therapy, as well as their dosage and usage.
- Certain medications require a prescription. Only liquids and medications indicated for inhalation may be used.
NOTICE
Keep the device as vertical as possible. However, a small angle does not affect usage as the container is leak-proof. Make sure that the medication is in contact with the mesh for proper functioning of the device during use. If the device is tilted slightly by 45 degrees with a maximum fluid volume of 10ml , the device will work as usual.
CAUTION
Pentamidine preparations, essential oils and medicines containing oils and lipids must not be used with this atomiser. Otherwise the nebuliser will be damaged. For health reasons, cough mixtures, gurgling solutions and drops for rubbing into the skin or for use in steam baths are completely unsuitable for inhalation with a nebuliser. These additives are also often viscous and can impair the correct functioning of the device and therefore the effectiveness of its usage in the long term.
In people with a hypersensitive bronchial system, medications that contain essential oils can sometimes cause acute bronchospasm (a sudden cramp-like narrowing of the bronchi with shortness of breath). Talk to your doctor or pharmacist about this.
7.6 Stopping the inhalation
a) Manually ending nebulisation:
- When you have finished the treatment, switch off the device using the On/Off control 4.
The LED display 5 will go out.
b) Automatically ending nebulisation:
- If the inhalant has been nebulised, the LED display will flash blue quickly and the device will automatically switch off. For technical reasons, a small amount of the medication remains in the medication container. Do not use this.
NOTICE
Certain medications (such as budesonide) might foam up during their application. If foam builds up within the medication container, or if the fill level is low and nebulisation can no longer occur, switch off the device. Automatic switch-off may no longer function reliably due to the foam; the mesh might continue to vibrate and eventually break
7.7 Cleaning
See section "Cleaning and maintenance".
7.8 Automatic switch-off
The device has an automatic switch-off function. When there is only a very small amount of medication or liquid left or when the substance to be nebulised is no longer in contact with the mesh, the device switches off automatically to prevent damage to the mesh. If nebulisation ends automatically, the automatic switch-off has been activated. The LED display 5 flashes blue quickly and the device then switches off. After a nebulisation period of 15 minutes, the device switches off automatically.
Do not operate the device if the medication container 2 is empty. The device detects when there is no medication in the medication container and will switch off automatically.
| LED display colour | Meaning |
| Green | Fully charged: As soon as the battery is fully charged, the LED display continuously lights up green. |
| Orange | Charging in progress:While the battery is charging, the LED display continuously lights up orange. Low battery level:If the LED display lights up orange while the device is in use, this indicates low a battery level. • At a high nebulisation rate: The LED display continuously lights up orange. • At a low nebulisation rate: The LED display flashes orange slowly. Self-cleaning mode:If you press and hold the On/Off control for 3 seconds while the device is switched off, the LED display flashes orange quickly and the device switches to self-cleaning mode. |
| Blue | Operation and medication flow:The blue LED display indicates that the device is functioning correctly. • At a high nebulisation rate of ≥ 0.25 ml/min: The LED display continuously lights up blue. • At a low nebulisation rate of ≥ 0.15 ml/min: The LED display flashes blue slowly. Insufficient medication and automatic switch-off:When the inhalant has been nebulised, the LED display will flash blue quickly and the device will automatically switch off. |
8. CLEANING AND MAINTENANCE

WARNING
Follow these hygiene instructions to avoid health risks.
- The medication container 2 and the atomiser set are intended for multiple use. Please note that different areas of application involve different requirements in terms of cleaning and hygienic preparation.
NOTICE
- Do not clean the mesh or the atomiser set mechanically with a brush or similar, as this can lead to irreparable damage and optimal treatment results can no longer be guaranteed.
- Ask your doctor for additional requirements regarding hygienic preparation (hand cleaning, handling of medications/inhalation solutions) for high-risk groups (e.g. patients with cystic fibrosis).
- The water mentioned later in the cleaning instructions should be as soft as possible. Distilled water is suitable for all cleaning steps except self-cleaning.
Disassembling
- Hold the device with one hand. The button 6 on the back of the device should face towards you. Press the button 6 with your thumb and pull the medication container 2 horizontally forwards with your free hand. F
- Remove the mouthpiece 11 or the mask 9 or 10 from the medication container 2.
- Open the lid of the medication container 2 by lifting up the flap 1 on the rear C.
Assembly is carried out in the reverse order.
Cleaning
The medication container 2 and the used parts of the atomiser set such as the mouthpiece 11 or mask (9 or 10) must be cleaned after each use with warm (ideally distilled) water at a maximum temperature of 40^ for about 5 minutes. To clean the medication container, please follow the self-cleaning instructions described below. Dry the parts carefully using a soft cloth. When completely dry, reassemble the parts and place in a dry, sealed container or disinfect them.
- Please perform the self-cleaning function afterwards (see "Self-cleaning function" section).
CAUTION
Never hold the entire device under running water to clean it.
When cleaning, ensure that any residue is removed and dry all parts carefully.
Do not use cleaning products that can be potentially toxic in contact with skin or mucous membranes if swallowed or inhaled.
If required, clean the housing of the device using a slightly damp cloth, which you can moisten with a mild soapy solution.
- If there is any residue from medical solutions or contamination on the silver/gold contacts of the device or medication container 2, clean with a cotton bud moistened with ethyl alcohol.
- The device must be switched off and cooled down before each cleaning.
- Do not use any abrasive cleaning products.
CAUTION
- Ensure that no water enters the device!
- Do not clean the device and atomiser set in the dishwasher.
- Avoid the device coming into contact with splash water. Only operate the device when it is completely dry.
CAUTION
We recommend performing the inhalation without interruption, as the inhalant can clog the medication container 2 during prolonged interruptions of treatment, which may impair the effectiveness of the administration.
Self-cleaning function
You can use the device's self-cleaning function to completely remove medication residues and any blockages.
To do so, proceed as follows:
-
Disassemble the device as described above.
-
Open the lid of the medication container 1 and pour out the remaining liquid.
- Pour a sufficient amount (4-6 ml) of tap water (soft water) into the medication container 2. Close the lid. Shake the container slightly so that the remaining liquid mixes well with the cleaning liquid.
- Open the lid of the medication container, pour out all the liquid and then fill it with enough tap water (soft water) again.
- Place the medication container on the housing 3
- Press and hold the On/Off control for 3 seconds when the device is switched off. The LED display then flashes orange quickly and the device switches to self-cleaning mode. The device will automatically run for 10 minutes. Please make sure that there is enough liquid in the medication container.
Disinfection
Please observe the following points when disinfecting your medication container 2 and the atomiser set. Regular disinfection of the individual parts is recommended.
First, clean the medication container 2 and atomiser set as described in the "Cleaning" section. Then proceed with disinfection.
CAUTION
Ensure that you do not touch the mesh if possible, as this may destroy it.
Do not use sodium hypochlorite disinfectant for disinfection as it may damage the atomiser.
Disinfection with ethyl alcohol (70-75%)
- Place the mouthpiece 11 and masks 9 10 in a container and cover all parts with ethyl alcohol. Leave it to take effect for 10 minutes.
- Finally, clean all parts again with water.
- Then dry all parts thoroughly. For more information, see the "Drying" section.
Disinfection with boiling water
- The medication container 2, lid 1 and the mouthpiece can be placed in boiling water for 15 minutes in order to disinfect them. Ideally, distilled water should be used to avoid contamination and
limescale formation on the mesh. The parts should not come into contact with the hot base of the pan.
- Then dry all parts thoroughly. For more information, see the "Drying" section.
- The medication container 2 must not be placed in the microwave.
- We guarantee 52 disinfection cycles with boiling water per medication container.
NOTICE
The masks must not be placed in hot water!
We recommend disinfecting the masks with 70 - 75% ethyl alcohol.
Drying
- Dry the parts carefully using a soft cloth.
- Gently shake the medication container 2 a few times so that any water is removed from the tiny holes in the mesh.
- Place the individual parts on a clean, dry and absorbent surface and leave them to dry completely (for at least 4 hours).
- Do not use a microwave, dish dryer or hair dryer for drying or disinfection.
NOTICE
Please ensure that the parts are completely dried after cleaning, otherwise there is a high risk of bacterial growth.
When completely dry, reassemble the parts and place in a dry, sealed container.
Durability of materials
- Like all plastic parts, the medication container 2 and the atomiser set are subject to a certain degree of wear and tear if used frequently and prepared hygienically. Over time, this may lead to a change in the aerosol, which may reduce the effectiveness of the treatment. We therefore recommend that you replace the medication container 2 and atomiser set after one year at the most.
Storage
- Do not store in damp conditions (such as in a bathroom) and do not transport with any damp items.
- Protect from direct sunlight during storage and transport.
9. ACCESSORIES AND/OR REPLACEMENT PARTS
To purchase accessories and/or replacement parts, visit www.beurer.com or contact the corresponding service address (as per the service address list) for your country. Accessories and/or replacement parts are also available from retailers.
| Designation (Material) REF | |
| Mouthpiece (PP), adult mask (PVC), children's mask (PVC), mesh atomiser with medication container (PC) | 110.126 |
10. WHAT IF THERE ARE PROBLEMS?
| Problem/question Possible cause/remedy |
| The nebuliser pro-duces no or too little aerosol. |
| 1. Too little medication in the medication container 2. |
| 2. The nebuliser is not being held vertically. |
| 3. Medication liquid has been poured in that is not suitable for nebulisation (e.g. too viscous. The viscosity must not be greater than 3. Ask your pharmacist about the viscosity.) The medication liquid should have been prescribed by a doctor. |
| 4. The battery is flat. Charge it. |
| The output is too low.1. There are air bubbles in the medication container that prevent continuous contact with the mesh. Please check this and remove any air bubbles. |
| 2. Particles on the mesh are hindering output. In this case, you should pause the inhalation process and remove the mask or mouthpiece. Then clean and disinfect the medication container (see “Cleaning and maintenance” section). |
| 3. The mesh is worn. Replace the medication container 2. |
| What medications are suitable for inhaling?Naturally, only your doctor can decide which medication should be used for treating your condition.Please consult your doctor in relation to this matter.With the IH 47, you can nebulise medications with a viscosity of less than 3.Medications containing oil (particularly essential oils), pentamidine preparations and medications containing lipids, should not be used, as these affect the material of the medication container, which can lead to faults. |
| There is inhalation solution residue in the medication container 2. This is normal and due to technical reasons. Stop the inhalation as soon as the atomiser makes a significantly different noise or the device switches off automatically due to a lack of inhalant. |
| What should be taken into account when using the device with infants and children?1. For infants, the mask should cover the nose and mouth to ensure effective inhalation.2. For children, the mask should also cover the nose and mouth. It is not advisable to use the nebuliser on a sleeping person because too little medication enters the lungs.Notice: Inhalation should only be carried out under the supervision of an adult and with their assistance, and the child should not be left alone. |
| Problem/question Possible cause/remedy | |
| Should each person have their own atom- iser set? | Yes, this is absolutely necessary for hygiene reasons. |
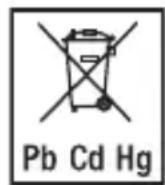
11. DISPOSAL
Battery disposal
- Empty, completely flat batteries must be disposed of in specially designated collection boxes, at recycling points or at electronics retailers. You are legally required to dispose of the batteries.
- The codes below are printed on batteries containing harmful substances:
Pb = battery contains lead
Cd = battery contains cadmium
Hg = battery contains mercury
General disposal
For environmental reasons, do not dispose of the device in household waste at the end of its service life. Dispose of the device at a suitable local collection or recycling point in your country. Dispose of the device in accordance with EC Directive - WEEE (Waste Electrical and Electronic Equipment). If you have any questions, please contact the local authorities responsible for waste disposal.
12. TECHNICAL SPECIFICATIONS
Type IH47
Dimensions (L × W × H) 48 × 59 × 115 ~mm
Weight 0.0926kg± 0.005kg
Input 5 V DC; 1.2 A

| Energy consumption ≤ 2.5 W | |
| Filling volume Min. 0.5 mlMax. 10 ml | |
| Medication flow rate Low nebulisation rate: ≥ 0.15 ml/minHigh nebulisation rate: ≥ 0.25 ml/min | |
| Vibration frequency 110 kHz ± 10 kHz | |
| Sound pressure Approx. ≤45 dB(A) | |
| Operating conditions Temperature: +5°C – +40°CRelative humidity: 15–90%, non-condensingAmbient pressure: 700 – 1060 hPa | |
| Storage and transportation conditions | Temperature: -40°C – +55°CRelative humidity: 5–93%, non-condensing |
| Expected service life of the device | Information on the service life of the product can be found on the homepage |
| Battery: | |
| Capacity | 600 mAh |
| Nominal voltage | 3.7 V |
| Type designation | Lithium-ion |
Aerosol values according to ISO 27427:2023 with salbutamol:
Aerosol delivery: 0.72ml
Aerosol delivery rate: 0.13 ml/min
- Fill volume dispensed in percent per minute: 6.47%
Residual volume: 0.04ml
- Particle size (MMAD): 3.43 m
GSD (geometrical standard deviation): 1.77
- RF (respirable fraction < 5 m ): 74.67%
Large particle range (>5 m) .. 25.33%
Medium particle range (2 to 5 m ): 57.27%
- Small particle range (< 2 m) : 17.40%
Software version V1.0.3.
The serial number is located on the device.
Subject to technical changes. (see "Cleaning and maintenance" section).
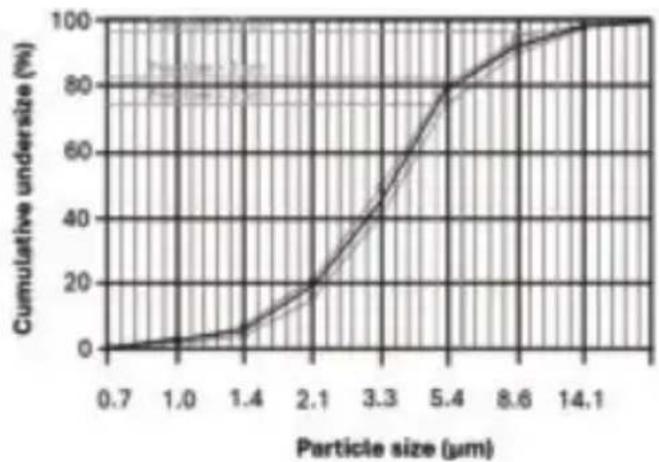
Particle size chart
Measurements were performed using a salbutamol solution with a "Next Generation Impactor" (NGI). The diagram may therefore not be applicable to suspensions or highly viscous medications. You can obtain more detailed information from the manufacturer of your medication.
Aerosol values are based on adult breathing patterns and are likely to vary for paediatric or infant populations.
NOTICE
If the device is not used according to the specifications, it may not work correctly! We reserve the right to make technical changes to improve and develop the product.
This device and its atomiser set conform with the European standard EN 60601-1-2 (Group 1, Class B, in accordance with CISPR 11, IEC 61000-3-2, IEC 61000-3-3, IEC 61000-4-2, IEC 61000-4-3, IEC 61000-4-4, IEC 61000-4-5, IEC 61000-4-6, IEC 61000-4-8, IEC 61000-4-11) and are subject to particular precautions with regard to electromagnetic compatibility.
13. GUARANTEE / SERVICE
More information on the guarantee and guarantee conditions can be found in the guarantee leaflet supplied.
Notification of incidents
For users/patients in the European Union and identical regulation systems, the following applies: If a major incident occurs during or through use of the product, notify the manufacturer and/or their representative of this as well as the respective national authority of the member state in which you are located.
FRANÇAIS

Instructions relatives aux piles
A AVENTISSEMENT
1 Couvercle du recipient a medicaments
9. ACCESSORI E/O RICAMBI
I P E D U N P E X K D E H N E
Yka3bIbaet Ha noteHnauhnyo onachoctb. Ecn ee He npedotbpaTntb, nocJeCTBnM MOryT 6bITb CMeptb NIN TjXKeJIbIe yBeYbIa.
BHUMAHNE
Yka3bIbaet Ha notehnnaIbHyo onaHocTb. Ecnn ee He npedotBpaTntb, nocJeCTBnM MOryT 6bITb IeKne nn He3HaunTeNbHbIe TpaBMbl.
YBEOMJIENHE
Yka3bIbaeT Ha BO3MOxHbIM MaTePnAJIbHbIM yUeep6. EcIn erO He npEDoTbPaTITb, BO3MOXHO NOBpeKdHeNe npi6opa IIN OkpyKaIOuIX ObekTOB.
Ha ynapokBke n Ha TnnoBoi Ta6nueKe npnbopa n KOMnIeKta npnaadJeXHocTei paCnblnteJnCNOlb-3yOTcra CneDyIOUne CNMBOJIb.
OBsIe IpeDyIpyKdEHNr
- Pacnbintelb He OKa3bIbaet cyueCTBeHHoro BnHnHa 3ΦΦeKTHBHOCTb N 6e3OnacHOCTb npHNMaembIX cero nOMOuIIO JeKapCTBeHHbIX npenapaTOB IN He npedHa3HaueH dJI NaeHn ONaChbIXdJI Xn3Hn COCToHn. Pacnbintelb He npedHa3HaueH dJI NCNoJb3OBaHn B aHeCTe3N0JOrHcCKNX IIN Dpyrnx CnCTeMax NCKycCTBeHHo BEHTNJraCNI JERKnx.
- Iocne Hndnexkaeien noTROBKn npnbop MOxHO nCNoIb3OBaTb NOBtOPO. TaKaI noTROBKa BKNIOaET 3aMeHy BcEx KOMNOHEHTOB, BKIOUay paCnblInTeJIb. PekOMeHdyEeTc 3aMeHryb EMKOCTB DIA JIeKApCTB IN DpyrNe KOMNOHEHTbI He N03dHee Yem CnYCTra RD NcNOb3OBaHN. DaHHa peKOmeHaIaIIN NO BpeMeHN 3aMeHbIO ChOBaHa Ha NpeDnIOJXeHN, YTO npnbop NcNOb3yETcR He 6oJIe 2 ZIKNob B DeH b V TeueHne ≤ 10 MInyT 3a ZIKN, a TAKKe peryIpaHO de3nHfNUPyETcN OUnIaETcB COOTBeTCTBUN C INHCTpyKUeN IO npImeHEnHO (8. «OuNcTKa UxOd'). IV3 coo6paHexHn ITRHeB KaKDbII NOB3OBaTeJIb DOJIXeH NcNOb3OBaTb NepcoHaJIbHbIE KOMNOHETbI.
Bcerda cIeDyTe yka3aHnM Bpaa IIN φapMaueBtA OTHOCHTbHO Tnna NcNoJIb3yeMOro MeiHckoro npenapaT,do3npOBKn, YactOtBI IN PPOJXKeJIbHOCTN INraJIaCm. Prn NcNoJIb3OBAHm CLeKapCTBeHHbIM npenapaTaMn/pacblHeHN JekapCTBeHHbIX npenapaTOB Co6JIIOaJTe ycNOBnI ORpaHnueHn, npimeHmble K 3Tm npenapaTam. YBeOMJeHne. NcNoJIb3yJrTe dJa JleEHnToJIbKO Te MeDiuHckne npenapaTbI, KOTopbie 6bln Ha3HaueHbI BpaOH M COOTBEcTBn C yCTaHOBJeHHbIM DnArHO3OM. - Пибор He заменяет собов Консультацю и leченье враuya. Рп ВОЗноковни 60лели пллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллллл.
- Балевни BCERда сана Na obpaшайтесь K врач.
- Ecni y Bac Bo3HnKIn npo6IeMbl co 3OpOBbEm, o6paTntecb K BpaCy!
He nCnoB3yIe B uHraJTope XnDkoCTn, HecOBMeCTmblc co CtaJIbH, NOINPpOnJIeHOM, NOLKap6oHaTOM, cnIKoHOM nn PBX. EcIn nHΦopMaun, npNlaRaemar K XnDkoCTn, He No3BOJareT onpeJeNTb ee COBMeCTUMOCTb C 3TmM MaTePnaJAmn, O6paTntEc b K pON3BOJNTeJIHO XnDkoCTn. K COBMeCTmblm XnDkoCTaM OTHCOTcR, HanpImep, pactBOp aJIb6yTePona.
KpnBa paCnpedeHn yacntu no pa3mepy, MMAD, oBem n/nn cKOpocb npdaun a3po3oJr MOryT n3MeHHTbcra npn nCNoJIb3OBAHn IekapCTBeHHoro CpeIcTBa, cycneH3n, 3Mylbcn nn BblCOKOBa3KOrO pactbopa, OTnUHO r OYka3aHHoro B TexHuecknx XapaKTepncTKkax dJa 3HaueHn a3p03oJr.
- Ppi nCnOJIb3OBaHm INrAJaTOpa BbIOnJIHJaTe 06Ue npHraTbe caHITapHO-rnHneHneckne Mepbl.
- Pered nCnoJIb3OBAHnem y6eIITecb, YTO npnbOp n erO KOMNoHEHTbI He NMeOT BUDIMbIX NOBpeJxHn. Pn HAnuHm ComHeHn He nCnoJIb3yIte npnbOp n O6paTITecb K npoDaBcy NIn No yKa3aHHOMy aIpeCy cepBnCHOn cnyK6bl.
3anpeaetcBHOCTb KaKne-JI6o NImeHnB Pnp6op nIe KOMnoHeTbl.
- IcnoJb3yIe ToJIbKO npuHaIeXHocTn, peKOMeHIOBaHHbIe npoI3BOIteJeM.
- INHraJrTop pa3peWaeTcR nCnOJIb3OBaTb TOJIbKO BMeCTe C NOxOJaUIMn paCnbIJIrTeJAmn Beurer n COOTBeTCTByIOUIMM KOMNoHEHTaMn Beurer. NcNoJIb3OBaHne paCnbIJIrTeJn KOMNoHErTOB dpyrNX npON3BOJNTeJn MOXET CHN3ITb 3ΦΦeKTINBHOCTb JueHn r n pInBecTn K NOBpeKdEHHIO np6opa.
- Bo Bpem 3KcnpnyatauHn Depxnte npnbop Ha DoCTaTOHOM pacctOHN OT rla3, NOCKoJIbky paCnblIeMbI JekapCTBeHHbI npenapaT MoKeT IN NOBpeDNTb.
- HnKoIa He nCnoJIb3yIte npI6Op pIOM C rOPIUcIMN IIN B3PbIBOONaCHbIMN ra3aMn, KNCLOPODOM nn OkCuIOM a3OTA.
XpaHnTe npi6op IN KOMNoHEtbl B MeCTe, HeIOCTyINHom IJr DeTeN I DomaunHX XJBOTbIX.
XpaHnTe ynaKOBouHbI MaTePnA1B HeIOCTynHom IyTei MeCe (OnaCHOctb ydysb).
- Pered BbINOJIHeHEm IIO6bIX pa60 Tn OOnCTKe n (nnn) TexHNueCKOMy 06ClyXnBaHnO BbIKIIOHTe np6op n n3BNeKIne uTekep Ka6eJ USB-C.
- Ecni np6op xpaHnIc npn MaKcImaJIbHOJ TeMnpaType xpaHeHnI, nepei nCNoJIb3OBAHnEM daITe emy ocTbITb B TeueHne 4 yacob, noka OH He 6ydt roTOB K nCNoJIb3OBAHnIO no Ha3HaueHnIO. Ecni np6op xpaHnIc npn MInHMaJIbHOJ TempepaType xpaHeHnI, To do erO rOTOBHOCTN K nCNoJIb3OBAHnIO no Ha3HaueHnIO taKke notpe6yETc 4 yaca.
- HnKoIa He norgykaIte np6op B Body n He nCnoJIb3yIte erO B BaHNoi KomHaTe. He dOnyckaIte nonadHna JxNdkOCTn BHyTpB np6opa.
- Ecni np6op yan, noBeprcn CunbHomy Bo3deNCTBIO BlaHn nnnoyn Hhie nobpeXDeHnra, ero daJIbHeiuee nCnoIb3ObaHne 3aPpeaetc. B cnyae comHeHn o6paTntecb B cepBnchHyoc Lyk6y nn K npodabuy.
C60n 3neKtponuTaHnB, BHe3aHbIe NomExn Hn DpyrNe He6laRopnTbIe ycIOBn MOryT npNBecTn K nolomKe np6opa. No3tomy MbI peKomeHdyem Imetb NOd pyko3aNaChO INp6Op nII 3a- NaChbIe MedunHckne npenapatbl (NocleDnE cJeDuET cornaOBaTb C BpaOM).
XpaHnTe uHyp B HeIOCTyNHom dIa DeTeMecTe BO n36eKaHne onaCHOCTu dyueHna. - Повлочаite npubop Tolyko K ceTeBOMy HappxeHIO, yka3aHHOMy Ha TINOBOI Ta6nUKe.
- HnKoIa He npKacaiTecb K Ka6eIIO USB-C MoKpbIMn pyKaMn, TaK KaK 3TO MOxET npNBecTu K nOpapKeHIO 3JIeKTPuueCKIM TOKOM.
- Пии n3BneueHn ceTeBO r aanTepa n3 po3eTKn He TAnHe 3a Ka6JIb USB-C.
He cdaBnBaIe n He nepeRn6aIte Ka6enb USB-C, He npOTraNbaIte n He noDbEwNBaIte ero HaI npedMeTAMNC OCTpbIMKpaMn, a TaXKe 3aunuaiTe OT NCTOCHIKOB TeIIa. - PekomeHnyetcnoHoctbpoa3MaTbIBaTb Ka6eNb USB-CДЯпpeDoTbpaUeHnnepepeBa.
- Поврждений кабел USB-C стого полесь утлиицу.Образсвесь в сервicHyо сухь уллк рordаву.
- Пи OTкрытпnpибopa суцьсByet onachocь nopaxkeня anektpnueckm TOKOM. Пибор He otКлочen OT cetn nHTaHn, ecn He OTknioch aadanTep (a Ka6eJb USB-C He nmeet dpyrOro pa3beMa nITaHn).
OBLNE MEPbI INPEIOCTOPOXHOCTN
- Hukorda He xpaHnte npnbop (nIu Ka6eJIb USB-C) pIaOM c nCTOHyKamTenla.
He nCnoB3yIe np6op B nOmeuHn, B KOToPOM paHee nCnoB3oBaJc sIpei. Ipeep npOBedeHnem npOeDpybI npOBeTpIe nOmeUeHne. -
Hukorda He nCnoJb3yIte np6Op, ecIn OH n3daeT Heo6bUHbIe Wymbl.
XpaHnTe np6Op B MeCTe, 3aunuHHom OT aTMocpehBix BO3deIcTBn. Pnp6op DoJxeh XpaHnTbCra Byka3aHHbIX ycIOBnX OKpyKaHOSeI cpebl.
He nCnoJb3yIte np6op pyDom c 3JeKTpomarHnTHbIMn nepeJaTnKaMn. -
3aunuaine npnbop ot cnIbHbIX yapOB.
- Pocne noHOn 3apAdkn 6aTapeKn OTKJIIOHTe npIbOp OT cTeN.
- Ecni Tpe6yeTcA aanTep nn ydHnHtBbHbI Ka6eIb, OH dONKeH COOTBeTCTBOBaTb DeiCTByIOUIM Tpe6oBaHnM 6e3oNaChocTn. 3aPpeuaetc npeBbIwaTb yKa3aHHyHO Ha aanTpe MaKcImaJIbHyO MOuHOCtB.
Ipeed noDrotOBKoK pa6ote
- Pered nCnoJb3ObaHnem npnbopa ydaJIte BCE ynaKOBOHyIe MaTePnAbI.
- He donyckaite 3aqr3henna np6opa, 3aunuaite ero ot nbin n Bnarn. B npocce 3kcnnyatau Hn B KOem clyae He hakpbBaite np6op.
He nCnoJb3yIe np6Op B cnJIbHO 3aJIbeHHbIX NOMeJeHnX.
Bcnyae o6hapxhenn depekTOB nHn HnOlaKOB pa6oTe HEmeJeHHO OTKnIOHTe np60p. - ПОНЗВОДИТЕЛБ He HeCET OTBETCTBEHHOCTH 3a yUeP6, Bbl3BaHHbI HekBaJIHΦNUPOBAHbIM IJIH HeHaJIEXKaUIM NcNoJIb3OBAHHeM.
PemOH
- KaTeOpnueckn 3aIpeuaaetcR oTKpbIBaTb npnbop n peMOHTnpoBaTb erO caMoCToTeNbHO, noCKoJIbKy HAdexKHOCTb erO pa60Tb I B 3Tom clyae He rapaHTnpyETcR. HecobIOJeHne 3TuX Tpe6oBAHn BeDET K Notepe rapaHTmN.
- Пибор He Tpe6yET TexHnueckoro O6cLyKuBaHn.
- Ecπι Tpe6yeTcρ πeMOHT, σθραπιTEcB B cepBnCHyO cIyXkEy ηιN K ΜΦιζηλβHOMγ ΣιCτρι6bIoTopy.
Yka3aHnno 6paueHHc 6aTapeKamn
PPEyPExKdEHN
- Onachoctb B3pbIba! Onachoctb noxapa! HecobJIOeHne cJeDyUOx yKa3aHn MoKeT npNBecTN K TpaBMam IIN K NepeIpeBy, BblTeKaHnIO, BblNyCKy Bo3dyXa, NOlOMKe, B3pbIBy IIN BO3rOpAHIO.
-ДяЗарядн BCERda nCnoIb3yIte NOxOJaIu NII IN BXOJaIu B KOMnJIeK T 3apJHbI Ka6eIb/3aPraHoe yCTPOIcTBO/6BLOK nITAHN.
He donyckaIte dInTeIbHO nIN ype3MepHO 3apAdkn. IIO 3aBepseHn npouceca 3apAdkn OTKJIIO-HTe 3apAIDHOE YCTPOJCTBO OT CETN NITAHIA.
3apjkaTe yctpoCTBO nO npncMOTpOM, pni 3tOM cJeNTe 3a BbIeJIeHHeM TeNla, N3MeHeHEm fopMbI bIeJIeHHeM ra3OB. B cIyuae comHeHn IpePBITE npocecc 3apJkn.
- П配电и Te И сплбзовын Heисправьile 6атapesteКИ/3apяньй KaБeЛb/Зардhoe yCTpoIcTBO nΚak можно скоpeeуУтINn3npyuTe INx HAnLnxaUIM OБра3OM (cM. rIaby «YtInn3aUry»).
He 6pocaiTe npibOp nIb 6aTapeKn B orOnb.
3aPepaetc npHynteBHO pa3paTb, HarpeBaTb, pa3bPaTb, BCKpbBaTb, pa3bNaTb, deOpMnPOBaTb, repMeTHNtPOBaTb, MOHnHnUPoBaTb NII NIOBepraTb ydapaam npnbop nII batapeKu.
He donnyckaIte KOPOTKOro 3aMbIkaHn 6aTapeek n KOHTaKToB npu6opa, pa6oTaUoJero ot 6aTapeek.
3aunuane npnbop nnn 6aTapeKn OT BO3dEeCTBna npMbIX COJIHeHbIX Jyuei,doxjra,CINbHoro HapReBa I nonadaHnB OBDbl.
- Bo3deiCTBHe Ha 6aTapeKn Upe3BbUaHbO BbICOKo TeMnepaTypbI NJIu Upe3BbUaHNo Hn3KOrO daBJeHnBa3DVxa MoKET Bbl3BaTb B3PbIB NJIu VTeHV JERKOBOcJIaMeHJIOUHXc JNkKOCTeN Ira3OB.
- Пи поадани кдкости n3 6aTapeiKn Ha Koxy nIn B rJa3a npomoi Te nopaxeHbI yacTOK 6oJIbshm KOJIuYeCTBOM BObl I O6paNTTeCb K BpaV.
YBEOMJIEHNE
Y6eINTEcB, YTO npn OCHNTke BCE OCTaTKN 6bJIIN ydaJIeHbI, IN TuaTeJIbHO BbCyIJInTe BCE DeTaN.
He nCnoJb3yIte YnCTaIe CpeIcTBA, KOToPbIe MOrY T 6bITb NOTeHuaJIbHO TOKcNHyIMN npN KOHTaKTe C KoxKe IIN CNIn3NtBIMN OboIoQkAMN, a TaKKe pNpNpOrIaTbIBaHN INN BDbIXAHNI.
Ipn Heo6xOaUMocTn OuncTe Kopnyc npnbopa cnerka BnaXHo caPeTKo, KOtOpyIO MoXHO CMOuHTb cIa6bIM MbIbHbIM paCTBOPOM.
- Ecni Ha cepe6pncTbix/30JIoNTncTbIX KOHTaKTax npnbopa n emKocTn dIJIeKapct2 ocTaIINcB cIebl MeiHcHcknx pactBopOB IJIIN 3aIrp3HeHn, ONUCTnte INX BaTHOJ NaIOUcKO, CMOueHHoN 3TNIOBbIM CNIPTOM.
- Pered kaxdoon ochnTko npnbop Heo6xOIMOBbIKNHOHTb naTb emy OCTbITb.
He ncpnoIb3yIte a6pa3nBhIe HcTcIue cpeDCTBa.
BHUMAHVE
CJIeIte 3a TEM, YTO6bI B np6op He nonana BOda!
He MoIte npIbOp IN KOMJIeK T pINHaJIeXHOCTe I paCnblITeJI B NocydomoEChN MaIInHe!
He donyckaIte nonadaHna 6pbI3r BODbl Ha npIbOp. 3KcNpyaTupyIte npIbOp Tolbko nocne erO NOHoro BblcIxAHnA.

BHIMAHHE
PekomeHdyetc npoBOuTb HraJauIIO 6e3 nepepbioB, TaK KaK HraJauIOHOoe BeueCTBO npu dInTeNbHom npepbBaHn TepaHm MoKeT 3acOpNTb EMKocTB dJa JekapCTB 2, YTO MOKeT NOBJIyTaHa 3ΦΦeKTINBHOCTb NODaHn JekapCTBa.
ФункцяCAMOQUNTKN
Для поногудалия OCTаTKOB ЛекapCTВ И BO3MOЖнblX 3acopoB MOЖHO INCNoIb3OBAТь Функцюcamоочтkn пибopa.
ДяЗTORO BbINONHITe CJIeNyUOuNe DeiNCTBnJ.
1. Pa36epnTe np6op, KaK onncaHo Bblwe.
2. OTKpoIte KpbIuKy EMKoCTn IJIeKapCTB 1 n CJIeIte OCTaBswUocr XnIkoCTb.
3. 3aJIeIe T B EMKoCTb IJRA JekapCTB 2 IOCTaTOUHoe KOJIuYeCTBO BOODPPOBOHOB OBDbl (C Hn3KIM CO- depXaHHeM U3BeCTn) (4-6 M). 3aKpOte KpbIuKy. CJIerKa BCTpXHnTe EMKOCTb, YTObI OCTaBuaJcR XnIKoCTb XopoWTo CMeWJalacb C XnIKoCTbIO DnI ONUCTKn.
4. OTKpoIe KpbIuKU yEMKOCTn IJIa JIeKApCTB, BblJeIte BCIO XNlKoCTb N 3aJIeIte DoCTaTOUHoe KOJIuYe-CTBO BOJONPOBOHOB OBDI (C HN3KIM COePxAHNEM N3BecTn).
5. YctaHOBtTe emKocTb dIJIeKapCTB Ha Kopnyc 3
6. Пи ВьклioунHom пиборе нахмiteи удржиBaite BBKJIIOUaTeJB 4 habkaTbIMВ TeueHne 3 ceKHyI.CBeToIDNoHbI INHnKaTOp 5 6bICTpO Mmraet opaHKeBbIM CBETOM,И пиборпepexoNTВ peJxM caMOOuHCTKn.Пибор abTomatUeCKn paobTaET B TeueHne 10 MmHT. CJeIte 3a DoctaToHbIM OБьemOM XnIDKOCTN B EMKOCTN ДЯ ЛЕКAPCTB.
De3nHΦeKzna
Pn De3HfekuHm EMKoCTn Dn JekapCTB 2 n KOMnJIeKTa npHaJNeXHOCTe paCbIInTeJr CO6HIO- daIte cIeNyUOHe yka3aHn. PekomeHdyETcpeyIpyHO de3HfNUPOBaTb OTdJIbHbIe DetaII.
Chauana ouhctte emkoctb nIekapctb 2 n KOMnneKT npHaJnxHocte paCnbIInTeJIa, KaK OnilcaHO B rnaBe «Ouuctka>. 3aTe m npoDOnxnte de3HfekUHO.
BHUMAHNE
CTapaTecb He npNKacTbcK cETke, TAK KaK 3TO MoKet npNBeCTn K ee NOBpeXdeHIO.
He nCnoIb3yIe rIIOXIoPHT HaTpIa Ie3HΦeKcIu, TaK KaK 3TO MOKeT NOBpeIb paCnblnteJIb.
Дeзинф ekцялгпспгтOM (70-75%)
YCTOuHBOCTb MaTePnAna
- KaK i BCE pIaCTMaCCOBbIe DeTaN, EMKOCTb Ira JIeKApCTB 2 n KOMJIeKT npHaIeXHOCTeI paCnblNTeJI NaDBepXeHbI ONpeDeIeHHOMy N3HOCy npN YactOM IcNOJb3OBAHN I rIgHeHueCKoI o6pa6OTke. Co BpeMeHem 3TO MOKeT PpNBecTI K N3MeHEnIO a3Po3OJIa, YTO MOKeT CHN3HTb 3Φ-ΦeKTHBHOCTb IeueHn. Po3TOMy peKOMeHdyETC 3aMeHHTb EMKOCTb Ira JIeKApCTB 2 n KOMJIeKT npHaIeXHOCTeI paCnblNTeJI He No3DHee Yem Upe3 rO.I.
XpaHeHne
3aPepaetc npabntb npnbop BO BnaXhix NomeeHnax (Haepmep, B BaHHoKOMHaTe) npeBO3NTb eO BMeCTe C BnaXhIMn PpeMetam.
- Пиханени и Транспортурке зашицайт eero OT пямьix солпуновлучел.
9. AKCECCYAPbl N/NI 3ANACHbIE DETAJIN
Akcceccyapbl n/nn 3anachbIe deTaln MOxHO npno6pctn Ha caIte www.beurer.com nnnepe3 cepBnCHyO cnyk6y B CTpaHcNPOJb3OBaHnra N3dEInn (CM. CnncOK aDpecoB cepBnchOn cnjXkb). AkcecCyapbl n/nn 3anachbIe deTaln TaKKe MOxHO npno6pctn B po3HnHbIX MaRa3NHax.
12. TEXHnueCKne DAHHbIE
Beurer UK Ltd., Suite 16, Stonecross Place, Stonecross Lane North, WA3 2SH Lowton, United Kingdom
Beurer GmbH · Söflinger Str. 218 · 89077 Ulm, Germany www.beurer.com · www.beurer-healthguide.com www.beurer-gesundheitsratgeber.com