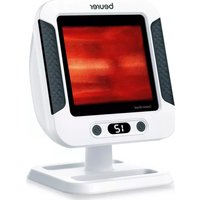
TL 95 - Light therapy BEURER - Free user manual and instructions
Find the device manual for free TL 95 BEURER in PDF.

User questions about TL 95 BEURER
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Light therapy in PDF format for free! Find your manual TL 95 - BEURER and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. TL 95 by BEURER.
USER MANUAL TL 95 BEURER
natural_image
White beige electronic device labeled 'beurer' with a silver base and small flower icon, no visible text or symbols on the device itself.EN Daylight therapy lamp Instructions for use....15
natural_image
Illustration of an open book with a geometric window and horizontal lines, no text or symbols presentEN Unfold page 3 before reading the instructions for use.
Read these instructions for use carefully. Observe the warnings and safety notes. Keep these instructions for use for future reference. Make the instructions for use accessible to other users. If the device is passed on, provide the instructions for use to the next user as well.
Contents
- Signs and symbols......16
- Intended purpose....17
- Warnings and safety notes......17
- Included in delivery .....19
- Device description....19
- Initial use ....19
-
Usage 20
-
Cleaning and maintenance.....21
- Accessories and replacement parts.....21
- What if there are problems? 22
- Disposal....22
- Technical specifications .....22
- Warranty ......24
Why use a daylight therapy lamp?
When the hours of daylight are noticeably shorter in autumn and people increasingly stay inside in winter, the effects of a lack of light may become apparent. This is often described as “winter depression”. The symptoms can present themselves in a number of ways:
- Imbalance
- Need for more sleep
- Subdued mood • Loss of appetite
- Lack of energy and listlessness • Difficulty concentrating
- Generally feeling under the weather
The cause of these symptoms is the fact that light – particularly sunlight – is essential for life and has a direct effect upon the human body. Sunlight indirectly controls the production of melatonin, which is only passed to the blood in darkness. This hormone shows your body that it’s time to sleep. That’s why more melatonin is produced in months with less sunshine, making it difficult to get up in the mornings because your body functions are powered down. Use the daylight therapy lamp immediately after waking up (i.e. as early as possible) to end the production of melatonin and to brighten your mood.
Lack of light also prevents the production of the happy hormone serotonin, which is said to significantly influence our well-being. The application of light thus yields quantitative changes to hormones and neurotransmitters in the brain that have an effect on our activity levels, our feelings and our well-being. To compensate for such a hormonal imbalance, daylight therapy lamps can create a suitable replacement for natural sunlight.
In the medical field, daylight therapy lamps are used to combat the effects of a lack of light. Daylight therapy lamps simulate daylight over 10,000 lux. This light can influence the human body and be used as a treatment or as a preventative measure. Normal electric light, however, is not sufficient to influence the hormonal balance. This is because in a well-lit office, the light intensity is just 500 lux, for example.
1. SIGNS AND SYMBOLS
The following symbols are used on the device, in these instructions for use, on the packaging and on the type plate for the device:
 | WARNINGWarning notice indicating a risk of injury or damage to health |  | CE labellingThis product satisfies the requirements of the applicable European and national directives. |
 | IMPORTANTSafety note indicating possible damage to the device/accessory | Storage | Permissible storage temperature and humidity |
 | Product informationNote on important information | Operating | Permissible operating temperature and humidity |
 | Observe the instructionsRead the instructions before starting work and/or operating devices or machines | IP21 | Protected against solid foreign objects 12.5 mm in diameter and larger, and against vertically falling drops of water |
| IP24 | Protection against solid objects greater than 12,5mm and agains splashes of water from all directions. | ||
 | Manufacturer Serial number |  | |
 | Protection class II deviceThe device is double-insulated and is, therefore, in protection class 2 |  | Dispose of packaging in an environmentally friendly manner |
 | ON/OFF |  | Marking to identify the packaging material.A = Material code, B = Material number: 1-7 = Plastics, 20-22 = Paper and cardboard |
 | Medical device For indoor use only |  | |
 | Item number |  | Authorised representative in the Europe-an Community |
 | Date of manufacture Importer symbol |  | |
 | atmospheric pressure limitation |  | Unique Device Identifier (UDI) for unique product identification |
 | Disposal in accordance with the Waste Electrical and Electronic Equipment EC Directive - WEEE |  | Direct currentThe device is suitable for use with direct current only |
 | Batch Code Type |  | |
 | Separate the product and packaging elements and dispose of them in accordance with local regulations. | ||
2. INTENDED PURPOSE
Purpose
The Bright Light Therapy lamp is intended to compensate the effects of lack of light, particularly sunlight, and provide relief from winter seasonal affective disorders, mood disorders and circadian phase sleep disorders.
Patient population
Adult and child over 3 years old.
Intended users
The use of the device does not require a specific knowledge or professional ability. The patient is the intended operator except in case patient that required special assistance.
Indication
The device simulates daylight to provide relief from seasonal or mood disorders.
3. WARNINGS AND SAFETY NOTES

- The daylight therapy lamp is intended for radiation on human bodies only.
- Before use, ensure that there is no visible damage to the device or accessories and that all packaging material has been removed. If you have any doubts, do not use the device and contact your retailer or the specified Customer Services address.
- Ensure that the daylight therapy lamp is positioned on a stable base.
- The device must only be connected to the mains voltage that is specified on the type plate.
- Never submerge the device in water and do not use it in the wet rooms.
- Do not use the daylight therapy lamp on animals!
- Do not use device on patient is taking a photosensitizing medication or herb.
- Do not use device on:
- children under age 3
- people insensitive to heat
- people with skin lesions due to illness
- people that has a condition that might render his or her eyes more vulnerable to phototoxicity
- people that has a photosensitive skin condi
- Keep packaging material away from children (risk of suffocation).
- If the device is warm, do not cover it or pack and store it.
- Always unplug the mains adapter and allow the device to cool down before touching it.
- Do not touch the device with wet hands when it is plugged in and do not allow water to spray on the device. Only operate the device if it is completely dry.
- Ensure that you only insert and remove the mains adapter with dry hands and that you only press the On/Off button with dry hands.
- Keep the mains cable away from hot objects and naked flames.
- Protect the device from heavy impacts.
- Do not pull the mains adapter out of the socket using the mains cable.
- Do not use the device if it shows signs of damage or does not function properly. In such cases, contact Customer Services.
- Do not leave the equipment unattended when it is switched on to avoid the risk of fire or burns.
- This equipment is not intended for use by children under three years. Children under three years should be supervised to ensure that they do not play with the equipment to avoid the risk of fire and burns.
- If the wall socket used to power the equipment has poor connections, the plug of the equipment becomes hot. Make sure you plug the equipment into a properly installed wall socket to avoid the risk of fire and burns.
- Do not subject the equipment to heavy shocks to avoid risk of damage to the lamp.
- In the event of damage to the mains connection cable for this device, the cable must be disposed of. If it is not a removable cable, the device must be disposed of.
- Disconnection from the power supply network is only guaranteed when the mains adapter is unplugged.
- Do not use the device in the presence of flammable anaesthetic gas connections with air, oxygen or nitrogen oxide.
- No calibration and no preventive checks or maintenance need to be carried out on this device.
- You cannot repair the device. The device contains no parts that you can repair.
- Do not make any changes to the device without the manufacturer's permission.
- If the device has been changed, thorough tests and checks must be carried out to ensure the continued safety of the device for any future use.
• To avoid strangulation and entanglement, keep cable out of reach of young children.
- The device is intended for use by the patient themselves. The device and accessories must be used in accordance with these instructions for use.
General notes

IMPORTANT
- Diabetics and people who suffer from retinal diseases must be examined by an optician before using the daylight therapy lamp.
- Please do not use the device if you suffer from an eye disease such as cataracts, glaucoma, diseases of the optic nerve or inflammation of the vitreous body.
- Please do not use in case of a recent eye surgery or a diagnosed eye condition whereby doctor has advised you to avoid bright light.
- Always consult a doctor before using the daylight therapy lamp if you have a strong sensitivity to light, skin sensitive to light or you are prone to migraine attacks.
- It must not be used on people with disabilities, children under 3 years of age or people with reduced sensitivity to heat (people with skin alterations due to illness).
- Always consult a doctor before using the daylight therapy lamp if you are taking medication such as pain relief medication, medication to reduce high blood pressure or antidepressant medication.
- Severe cases of SAD should, in any case, be closely supervised by a clinician.
• If you have health concerns of any kind, consult your general practitioner!
- Remove all packaging material before using the device.
• Light sources are excluded from the warranty.
- If the device has been in storage or recently transported, keep it for at least two hours at room temperature before using it.
• Power adapter is part of the ME eugipment.
- Power-on check item: please check whether light flashes, dark areas/shadows and other abnormalities occur after power-on. If there is any abnormality, please contact the after-sales hotline.
- The patient cannot undergo MRI scan while using this device.
- Please report any serious incident that has occurred in relation to the device injury or adverse event to the local competent authority and to the Manufacturer or to the European Authorised Representative (EC REP) Vigilance contact point: https://ec.europa.eu/growth/sectors/medical-devices/contacts/
- PRC is the abbreviation of the People's Republic of China.
Instruction for repairs

IMPORTANT
- Do not open the device. Do not attempt to repair the device yourself. This could result in serious injury. Failure to comply with this instruction will void the warranty.
- For repairs, please contact Customer Services or an authorised retailer.
4. INCLUDED IN DELIVERY
Check that the exterior of the cardboard delivery packaging is intact and make sure that all contents are present. Before use, ensure that there is no visible damage to the device or accessories and that all packaging material has been removed. If you have any doubts, do not use the device and contact your retailer or the specified Customer Services address.
• 1 daylight therapy lamp
• 1 set of instructions for use
- 1 mains adapter
5. DEVICE DESCRIPTION
The corresponding drawings are shown on page 3.
1 Fluorescent screen
5 On/Off button
2 Rear of the device housing
6 Timer setting
3 Stand
7 Brightness setting (dimmer)
4 Mains adapter connector
6. INITIAL USE
Take the device out of the plastic wrapping. Check the device for damage or faults. If you notice any damage or faults on the device, do not use it and contact Customer Services or your supplier.
Setting up the device
Place the device on a level surface. The position should be chosen to ensure a distance of between 20 cm and 45 cm between the user and the device. The lamp is most effective at this distance.
Mains connection
- To prevent possible damage to the device, the daylight therapy lamp must only be used with the mains adapter described here.
- Insert the mains adapter into the connector provided for this purpose on the rear of the daylight therapy lamp. The mains adapter must only be connected to the mains voltage that is specified on the type plate.
- After using the daylight therapy lamp, unplug the mains adapter from the mains socket first and then disconnect it from the daylight therapy lamp.
i Note
Ensure that there is a mains socket close to the set-up area.
Arrange the mains cable in such a way that no-one will trip over it.
7. USAGE
| 1 | Insert the mains adapter into the connector. | |
| 2 Switching on the lampPress and hold the On/Off button5 for 2 seconds. When the device is next switched on, it will start with the saved brightness level and treatment time. | ||
| 3 LED display/timerThe TL 95 Daylight therapy lamp indicates your current treatment time using 4 LEDs. The treatment time can be set at the following 4 levels.The timer function will not be active when the lamp is switched on for the first time. The lamp now runs until it is switched off again or a treatment time is set (timer = 0 min). In order to use the timer, set the desired level from 1 to 4 by pressing briefly the On/Off button5. Level 1 sets a treatment time of 30 minutes. The other levels are as follows:Treatment time Level (Number of lit LEDs)30 minutes 160 minutes 290 minutes 3120 minutes 4The lamp switches off automatically after this treatment time and steadily becomes darker. To deactivate the timer function, briefly touch the On/Off button until the LEDs go out. | ||
| 4 Brightness settingThe TL 95 Daylight therapy lamp has 6 brightness levels. The desired brightness level can be set using the flower button on the side – this level should be set so that it is pleasant for the eye during the treatment. Settings 1 to 6 correspond to 25%, 33%, 43%, 57%, 75% and 100% intensity respectively. When the device is switched off, the last level that was set is saved.Note:The device can also be used as a reading lamp from level 1. | ||
| 5 Enjoying the lightPosition yourself as close as possible to the lamp, at a distance of between 20 cm and 45 cm. You can continue to go about your day-to-day activities during the treatment, including reading, writing, making telephone calls, etc.Keep looking directly into the light for a short period of time, as it is absorbed and shows an effect via the eyes/retina.Use the daylight therapy lamp as often as you want. However, the treatment is most effective if you carry out the light therapy for at least 7 successive days according to the prescribed times.The most effective time of day for the treatment is between 6 am and 8 pm, and we recommend that you use the device for 2 hours per day.However, do not look directly into the light for the entire treatment time, as this may cause over-stimulation of the retina.Start with a brief treatment time and gradually increase the time over the course of a week.① NoteYou may experience pain in the eyes and headaches after the first few applications. This pain should disappear in further sessions as the nervous system becomes accustomed to the new stimuli. | ||
| 6 Things to considerWe recommend a distance of between 20 and 45 cm from the face to the lamp during treatment.The treatment time depends on the distance: | ||
| Lux Distance Treatment time | ||
| 13,000 approx. 20 cm 20 min. | ||
| 10,000 approx. 25 cm 0.5 hours | ||
| 5,000 approx. 30 cm 1 hour | ||
| 2,500 approx. 45 cm 2 hours | ||
| In principle:The closer you are to the light source, the shorter the treatment time. | ||
| 7 Enjoying the light over longer periodsRepeat the treatment on at least 7 consecutive days during darker periods of the year, or for longer depending on your individual needs. The treatment should take place during the mornings where possible. | ||
| 8 Switching off the lampPress and hold the On/Off button 5 for 2 seconds. The LEDs switch off. Unplug the mains adapter from the mains socket.⚠️ IMPORTANTThe lamp remains hot after use. Allow the lamp to cool down sufficiently before putting it away and/or packing it away. | ||
8. CLEANING AND MAINTENANCE
The device should be cleaned from time to time.

IMPORTANT
- Ensure that no water gets inside the device!
The device must be switched off, disconnected from the mains and allowed to cool down each time before cleaning. - Do not clean the device in the dishwasher. Use a slightly damp cloth to clean the device.
- Do not use any abrasive cleaning products and never submerge the device in water.
Do not touch the device with wet hands when it is plugged in and do not allow water to spray on the device. Only operate the device if it is completely dry.
Storage
If you are not going to use the device for an extended period of time, disconnect it and store it in a dry place, out of the reach of children.
Follow the storage instructions provided in the “Technical specifications” chapter.
9. ACCESSORIES AND REPLACEMENT PARTS
To purchase accessories and replacement parts, please visit www.beurer.com or contact the service address for your country (see the service address list). Accessories and replacement parts are also available from retailers.
10. WHAT IF THERE ARE PROBLEMS?
| Problem Possible cause Solution | ||
| Device does not light up | On/Off button 5 switched off Switch on the On/Off button 5. | |
| No power Connect the mains adapter correctly. | ||
| No power | The mains adapter is faulty. Contact Customer Services or your retailer. | |
| LEDs have reached the end of their service life. LEDs faulty | For repairs, please contact Customer Services or an authorised retailer. | |
11. DISPOSAL
For environmental reasons, do not dispose of the device in household waste at the end of its service life. Dispose of the device at a suitable local collection or recycling point in your country. Observe the local regulations for material disposal. Dispose of the device in accordance with EC Directive – WEEE (Waste Electrical and Electronic Equipment). If you have any questions, please contact the local authorities responsible for waste disposal. You can obtain the location of collection points for old devices from the local or municipal authorities, local waste disposal company or your retailer, for example.
| Component | Disposal Description | Photo |
| Device The component mainly is PC, metal and ABS. All comply with RoHS and REACH, and all could be safety disposal. |  | |
| Power adapter The adapter mainly includes plastic and electronic components, All comply with RoHS and REACH, and all could be safety disposal. |  | |
12. TECHNICAL SPECIFICATIONS
| Type GCE505 | |
| Model no. TL 95 | |
| Dimensions (L x W x H) 30.0 x 15.8 | x 47.5 cm |
| Weight approx. 1900 g | |
| Light | Full-spectrum LEDs |
| Power output | max. 60 watts |
| CRI | min. 95 |
| Light intensity 13,000 lux (at a distance of approx. 20 cm)10,000 lux (at a distance of approx. 25 cm) |
| Radiation Radiance output outside of the visible range (infrared and UV) is low enough that no eye or skin damage is anticipated. |
| Operating conditions 0°C to +35°C, 15 - 90% relative humidity,700 – 1060 hPa ambient pressure |
| Transport and storage conditions -20°C to +60°C, 15 - 90% relative humidity,700 – 1060 hPa ambient pressure |
| Product classification Protection class II, IP21 |
| Accessories Mains adapter, instructions for use |
| Colour temperature of the LEDs 6500K ±400 Kelvin |
| Threshold of short wavelength 420 - 680 nm |
| Maximum light output 66.88 W / m2 |
| Expected service life of the device 10.000 hours |
Maximum radiance output of the TL 95
| Radiance output Risk group classified | in accordance with IEC 62471 | Maximum value |
| E_UVA: Eye UV-A Exempt Group 0 | ||
| ES: Actinic UV skin & eye | Exempt Group 0 | |
| EIR: Infrared radiation hazard exposure limits for the eye | Exempt Group 2.146e-1 | |
| LIR: Retinal thermal (mild visual irritation) | Exempt Group 7.057e-2 | |
| LB: Blue light | Exempt Group 8.867e0 | |
| LR: Retinal thermal | Exempt Group 1.187e2 |
Subject to technical changes.
The serial number is located on the device or in the battery compartment.
Brightness: 10,000 lux (this point about the light intensity is merely for information purposes. With regard to the standard IEC 60601-2-83, this light source is classified as an Exempt Group).
Notes on electromagnetic compatibility
The device complies with Regulation (EU) 2017/745 of the European Parliament and of the Council on medical devices and the respective national regulations and the European standard EN 60601-1-2 (in accordance with CISPR 11, IEC61000-3-2, IEC61000-3-3, IEC 61000-4-2, IEC 61000-4-3, IEC 61000-4-4, IEC 61000-4-5, IEC 61000-4-6, IEC 61000-4-11, IEC 61000-4-8) and is subject to particular precautions with regard to electromagnetic compatibility.
- The device is suitable for use in all environments listed in these instructions for use, including domestic environments.
- The use of the device may be limited in the presence of electromagnetic disturbances. This could result in issues such as error messages or the failure of the display/device.
-
Avoid using this device directly next to other devices or stacked on top of other devices, as this could lead to faulty operation. If, however, it is necessary to use the device in the manner stated, this device as well as the other devices must be monitored to ensure they are working properly.
-
The use of accessories other than those specified or provided by the manufacturer of this device can lead to an increase in electromagnetic emissions or a decrease in the device's electromagnetic immunity; this can result in faulty operation.
- Failure to comply with the above can impair the performance of the device.
Mains adapter
| Model no. LXCP62 (II)-240 | |
| Input 100–240 V ~ 50/60 Hz, 1.5 A max | |
| Output 24 V DC, 2.5 A | |
| Protection The device is double-protected. | |
| ◇◇◇ | Polarity of the DC voltage connection |
| Classification IP24, protection class II | |
13. WARRANTY
Further information on the warranty and warranty conditions can be found in the warranty leaflet supplied.
Notification of incidents
For users/patients in the European Union and identical regulation systems (EU Medical Device Regulation (MDR) 2017/745), the following applies: If during or through use of the product a major incident occurs, notify the manufacturer and/or their representative of this as well as the respective national authority of the member state in which the user/patient is located.

9. ACCESSORI E RICAMBI
3. WAARSCHUWINGEN EN VEILIGHEIDSOPMERKINGEN

ARSCHUWING
5. BESCHRIJVING VAN HET APPARAAT
Electromagnetic compatibility
Table 1:
| Guidance and manufacturer's declaration - electromagnetic emissions | ||
| The Device is intended for use in the electromagnetic environment specified below. The customer or the user of the Device should assure that it is used in such an environment | ||
| Emissions test Compliance Electromagnetic environment – guidance | ||
| RF emissionsCISPR 11 | Group 1 The Device uses RF | energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. |
| RF emissionsCISPR 11 | Class B The Device is suitable | for use in all establishments including domestic and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes. |
| Harmonic emissionsIEC 61000-3-2 | Class A | |
| Voltage fluctuations/flicker emissionsIEC 61000-3-3 | Complies | |
Table 2:
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The Device is intended for use in the electromagnetic environment specified below. The customer or the user of the Device should assure that it is used in such an environment | |||
| Immunity test IEC 60601test level | Compliance level | Electromagnetic environment - guidance | |
| Electrostatic discharge (ESD)IEC 61000-4-2 | ±2kV ±4kV ±6kV ±8kV±15kV | ±8 kV contact±2kV ±4kV ±8kV ±15kVair | Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30 % |
| Electrical fast transient/burstIEC 61000-4-4 | ±1kV, ±2kV, 100 kHz repetition frequency | Power supply lines ±2 kV M | Mains power quality should be that of a typical commercial or hospital environ-ment. |
| SurgeIEC 61000-4-5 | ±0.5kV, ±1kV (Line to line)±0.5kV, ±1kV, ±2kV(Line to Ground) | Line to line: ±0.5kV, ±1kVLine to Ground: ±0.5kV, ±1kV, ±2kV | Mains power quality should be that of a typical commercial or hospital environ-ment. |
| Voltage dips, short interrupti-ons and voltage variations on power supply input lines | 0% UTfor 0.5 cycle0% UTfor 1 cycle70% UT | 0% UTfor 0.5 cycle0% UTfor 1 cycle70% UT | Mains power quality should be that of a typical commercial or hospital environ-ment. |
| Voltage dips, short interrupti-ons and voltage variations on power supply input linesIEC 61000-4-11 | for 25/30 cycles0% UTfor 250/300 cycles | for 25/30 cycles0% UTfor 250/300 cycles | Mains power quality should be that of a typical commercial or hospital environ-ment. If the user of the model TL55 re-quire continued operation during power mains interruptions, it is recommended that the model 168 DAYLUX daylight therapy lamp be powered from an unin-terruptible power supply or a battery. |
| Power frequency (50/60 Hz) magnetic field IEC 61000-4-8 | 30 A/m50Hz/60Hz | 30 A/m50Hz/60Hz | Power frequency magnetic fields should be at levels characteristic of a typical location in a typical commercial or hospital environment. |
| NOTE U_T is the a.c. mains voltage prior to application of the test level. | |||
Table 3:
| Guidance and manufacturer's declaration - electromagnetic immunity | ||||
| The Device is intended for use in the electromagnetic environment specified below. The customer or the user of the Device should assure that it is used in such an environment | ||||
| Immunity test IEC 60601test level | Compliancelevel | Electromagnetic environment -guidance | ||
| Conduced RFIEC61000-4-6 | 150KHz to 80MHz _T 3Vrms6Vrms (in ISMand amateur radiobands)80% Am at 1kHz | 150KHz to 80MHz _T 3Vrms6Vrms (in ISMand amateur radiobands)80% Am at 1kHz | Portable and mobile RF communications equipment should be used no closer to any part of the Device ,including cables, than the recommended separation distance calculated from the equation appropriate for the frequency of the transmitter.Recommended separation distances: d=1.2 ; d=2 | |
| Radiated RFIEC61000-4-3 | 10V/m 10V/m 80MHz to 800MHz | d=1.2 ^T 800MHz to 2.7GHz _T d=2.3 | Where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer, d is the recommended separation distance in meters (m) Field strengths from fixed RF transmitters, as determined by an electromagnetic site surveya ,should be less than the compliance level in each frequency range bInterference may occur in the vicinity of equipment marked with the following symbol: | |
| NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies.NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | ||||
| a Field strengths from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the [XXXXX] is used exceeds the applicable RF compliance level above, the [XXXXX] should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as re-orienting or relocating the [XXXXX].b Over the frequency range 150 kHz to 80 MHz, field strengths should be less than 3 V/m. | ||||
Table 4:
| Recommended separation distances between portable and mobile RF communications equipment and the [] | ||||
| The Device is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the Device can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and the Device as recommended below, according to the maximum output power of the communications equipment. | ||||
| Rated maximum output power of transmitter W | Separation distance according to frequency of transmitter m | |||
| 150 kHz to 80 MHz (out ISM and ama- teur radio bands) d=1.2 | 150 kHz to 80 MHz (in ISM and ama- teur radio bands) d=2 | 80MHz to 800MHz d=1.2 | 800MHz to 2.7GHz d=2.3 | |
| 0.01 0.12 0.2 | 0.12 0.23 | |||
| 0.1 0.38 0.6 | 32 0.38 0.73 | |||
| 1 1.2 2 1.2 | 2.3 | |||
| 10 3.8 6.32 | 3.8 7.3 | |||
| 100 12 20 12 | 23 | |||
| For transmitters rated at a maximum output power not listed above, the recommended separation distance d in meters (m) can be estimated using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) accordable to the transmitter manufacturer.NOTE 1 At 80 MHz and 800 MHz. the separation distance for the higher frequency range applies.NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | ||||
Table 5:
| Guidance and manufacturer's declaration – electromagnetic immunity |
| The Device is intended for use in the electromagnetic environment specified below. The customer or the user of the Device should assure that it is used in such an environment |
| Radiated RF IEC61000-4-3 (Test specifications for ENCLOSURE PORT IMMU-NITY to RF wireless communications equipment) | Test Frequen-cy (MHz) | Band a) (MHz) | Service a) Modulation b) Modulation b) Modulation b) (W) | Distance (m) | IMMUNI-TY TEST LEVEL (V/m) | ||
| 385 380 | -390 TETRA | 400 Pulse | modulation b) 18 Hz | 1,8 0,3 27 | |||
| 450 380 | -390 GMRS | 460, FM c) | ± 5 kHz de-viation 1 kHz sine | 2 0,3 28 | |||
| 710 704 | -787 LTE Band | 13, 17 | Pulse modulation b) 217 Hz | 0,2 0,3 9 | |||
| 745 | |||||||
| 780 | |||||||
| 810 800 | -960 GSM | 800/900, TETRA 800, iDEN 820, CDMA 850, LTE Band 5 | Pulse modulation b) 18 Hz | 2 0,3 28 | |||
| 870 | |||||||
| 930 | |||||||
| 1720 | 1 700 - 1 990 | GSM 1800; CDMA 1900; GSM 1900; DECT; LTE Band 1, 3, 4, 25; UMTS | Pulse modulation b) 217 Hz | 2 | 0,3 | 28 | |
| 1845 | |||||||
| 1970 | |||||||
| 2450 | 2 400 - 2 570 | Bluetooth, WLAN, 802.11 b/g/n, RFID 2450, LTE Band 7 | Pulse modulation b) 217 Hz | 2 | 0,3 | 28 | |
| 5240 | 5 100 - 5 800 | WLAN 802.11 a/n | Pulse modulation b) 217 Hz | 0,2 | 0,3 | 9 | |
| 5500 | |||||||
| 5785 | |||||||
| NOTE UT If necessary to achieve the IMMUNITY TEST LEVEL, the distance between the transmitting antenna and the ME EQUIPMENT or ME SYSTEM may be reduced to 1 m. The 1 m test distance is permitted by IEC 61000-4-3. | |||||||
| a) For some services, only the uplink frequencies are included.b) The carrier shall be modulated using a 50 % duty cycle square wave signal.c) As an alternative to FM modulation, 50 % pulse modulation at 18 Hz may be used because while it does not represent actual modulation, it would be worst case. | |||||||
The MANUFACTURER should consider reducing the minimum separation distance, based on RISK MANAGEMENT, and using higher IMMUNITY TEST LEVELS that are appropriate for the reduced minimum separation distance. Minimum separation distances for higher IMMUNITY TEST LEVELS shall be calculated using the following equation:
$$ E = 6 / d \sqrt {P} $$
Where P is the maximum power in W, d is the minimum separation distance in m, and E is the IMMUNITY TEST LEVEL in V/m.
For transmitters rated at a maximum output power not listed above, the recommended separation distance d in metres (m) can be estimated using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer.
NOTE 1 At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies.
NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people.
CE0123

Globalcare Medical Technology Co., Ltd.
7th Building, 39 Middle Industrial Main Road, European Industrial Zone,
Xiaolan Town, 528415
Zhongshan City, Guangdong Province, P.R. China
Phone : +86 760 22589901 / http://www.globalcare.com.hk/contact/
BEUER GmbH • Söflinger Str. 218 • 89077 Ulm (Germany)
Doravall'fescience, Piazza Albania, 10, 00153 Rome, Italy
https://www.donawa.com/wli/main/contatti.index