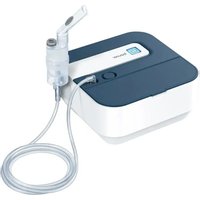
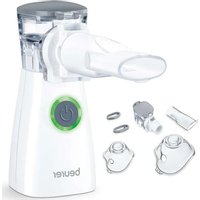
IH 30 - Inhaler BEURER - Free user manual and instructions
Find the device manual for free IH 30 BEURER in PDF.

Frequently Asked Questions - IH 30 BEURER
User questions about IH 30 BEURER
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Inhaler in PDF format for free! Find your manual IH 30 - BEURER and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. IH 30 by BEURER.
USER MANUAL IH 30 BEURER
Instructions for use
F Inhalalateur
Mode d'emploi
E Inhalatore
- Top cover
- Decoration ring
- Medication cup
- Watertank
- Indication lamp
- ON/OFF button
- Main body
- Back cover
-
Filter case
-
Air filter

-1 extension tube
- 1 adult mask
-1 children mask
-1 rechargeable battery
-1adapter
- 1 protective bag
F Accessoires
We are happy that you have decided on a product from our range. Our name stands for high-qua- lity and thoroughly tested products from the areas of warmth, blood pressure, body temperature, weight, gentle therapy, massage and air. Please read these instructions for use carefully and follow the directions.
Best regards, The Beurer Team
Indications
This nebuliser is suitable for the treatment of asthma, chronic bronchitis and other respiratory tract diseases e.g. the common cold. This innovative ultrasound technology allows the medically effective inhalation of aerosol medicines into the upper and lower airways. This inhalation prevents respiratory tract diseases, alleviates their accompanying symptoms and speeds recovery. You can obtain further information on suitable applications from your doctor or pharmacist.
The device is suitable for inhalation at home or when travelling. Inhalation of medication should be used only under a doctor's supervision. Carry out inhalation in a quiet and relaxed state, and inhale slowly and deeply, so that the medicine can enter the fine, deep bronchial tubes. Exhale normally.
The following symbols are used on the pa- ckaging and on the nameplate of the device and accessories.
| Application component type B | |
| Observe the instructions for use | |
| Manufacturer | |
| SN | Serial number |
| Keep dry |

Attention: Important Notes Keep for later use
- After opening the packaging, check that the Inhalator and accessories have no damage or defects. Otherwise contact your dealer immediately.
- Use the Inhalator only as specified, i.e. for aerosol therapy. All other use is improper and therefore dangerous. The manufacturer cannot be held responsible for damage caused by improper or incorrect usage.
- Use the Inhalator on frail persons and children only under supervision.
- The Inhalator and accessories must never be left in the unsupervised hands of children.
- Before using your Inhalator, ensure that the supply voltage stated on the power line adapter is the same as that of the local supply voltage.
- Use only the power line adapter supplied.
- Always use only pure water in the water tank. Other liquids may cause the Inhalator to become defective.
- The Inhalator must not be tilted or shaken during use. Otherwise, the medicament could be mixed with the water in the tank.
- Keep the Inhalator away from your eyes during use. The medicament mist could cause injury.
- Never use the Inhalator when the water tank or medicament cup is empty.
- Always comply with the doctor's instructions concerning the type of medicament to be used, the dosage and duration of inhalation.
- Do not operate the Inhalator in the presence of flammable gases.
- Never touch the power cord or power line adapter with wet hands - you could receive an electric shock.
- Comply with the cleaning instructions.
- Only one person should use the Inhalator in order to avoid cross infection.
- If you find that your Inhalator is not suctioning correctly or is damaged, switch it off and disconnect it from the power line adapter or battery. Then contact your authorized dealer. Only Beurer spare parts must be used.
The Inhalator consists only of parts that can be repaired or exchanged by the authorized dealer/service agency.
- Batteries that are defective or no longer required should not be deposited in domestic waste. Dispos of the fully discharged battery at disposal depots established for this purpose. You can fully discharge the battery yourself ba operating the Inhalator until its indicator lamp lights red or flashes.
Power supply
The Inhalator can be operated in two different ways:
1. With the power line adapter
The power line adapter is suitable for use in homes provided with a 230V supply voltage. Insert the small plug of the power line adapter into the socket underneath the Inhalator. Then plug the power plug of the power line adapter into the socket outlet. The Inhalator is now ready to use.
2. With rechargeable battery
The battery allows you to use the Inhalator where no other power sources are available. Insert the small battery plug into the socket underneath the Inhalator. Its operating time lasts up to 30 min. If the indicator lamp lights red or flashes, this means that the battery is discharged and must be charged up. Use only the rechargeable battery supplied by the manufacturer. The use of other batteries can damage the Inhalator.
Starting up
The Inhalator must be thoroughly cleaned before first use. This also applies if the Inhalator has not been used for a long time (see "cleaning").
Filling the water tank
- Connect the Inhalator to the power line adapter or the battery.
- Ensure that the Inhalator is switched off (the indicator lamp must be not be lit, i.e. it must neither flash green nor light up green!).
- Remove the top cover from the Inhalator.
- Fill the tank only with tap water or distilled water (20 - 30^) otherwise you could damage your Inhalator.
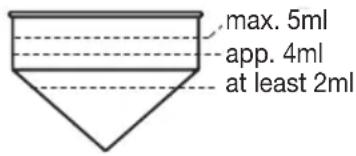
We recommend that you boil the water beforehand. The correct filling level is between the two markings on the inside of the tank. If the water temperature is lower than 20^ , nebulization of the medicament will be considerably less than at the specified temperature. The warmer the water, the greater the degree of nebulization.
- Empty the tank after every session and fill it again each time the Inhalator is used.
Filling the medicament cup
- Place the medicament cup on the water tank.
- Pour the medicament directly into the cup. Do not overfill the medicament cup! The filling level is at max. 5 ml!
- Use medicaments only under your doctor's instructions and check the inhalation time and quantity appropriate for you.
- If the prescribed quantity of medicament is less than 2ml , top it up to at least 4ml with distilled water or isotonic saline solution! Viscous medicaments must also be diluted. In this case also, follow your doctor's instructions.
- Replace the top cover back on the Inhalator again and ensure that it snaps into place.
Stop using the medicament cup as soon as signs of wear appear at the tip (cracks, white marks). Do not use a medicament cup for more than three sessions.
Using the Inhalator with accessories (optional)
The Inhalator can be used both with and without a mask. It is advisable for children to use a mask.
During the following procedures, take care not to tilt the Inhalator to prevent mixing of the water and medicament.
- Make sure that the Inhalator is switched off.
- Now push the short, white tube of the inhalation mask (with children the smaller mask) into the open end of the extension tube.
- Fit the extension tube into the aperture in the top cover by pushing the tube together slightly at a smooth end, and push it into the aperture until a slight snap engagement can be heard.
Operation
Make sure that the Inhalator is in the vertical position before you switch it on and thus ensure that it operates correctly with maximum effectiveness.
- Press the ON/OFF button to switch on the Inhalator. The indicator lamp lights up green.
- Adjust the filter case at the back of the Inhalator to ensure the required amount of airflow. At maximum setting, three air holes can be seen.
-
If desired, use the inhalation mask. We also recommend during use of the mask that you breath in and out through your mouth in order to reach the bronchial system. If your nose is to be included, breath in through your nose and out through your mouth.
-
Inhale the medicament prescribed by your doctor. The Inhalator is designed so that the
inhalation rate increases after a few minutes. It begins at a minimum.
- When the mist is no longer constant, this indicates that the medicament cup is empty. Inhale until no more mist is produced or the Inhalator switches off (the Inhalator switches off automatically after 10 min. of use).
- Switch off the Inhalator by pressing the ON/OFF button. The indicator lamp goes out. Unplug the power plug of the power line adapter from the socket outlet and unplug the small plug from the socket underneath the Inhalator.
- Empty the medicament and water residues (WARNING: HOT!) and clean the Inhalator as described under "Cleaning". Allow the Inhalator to cool down for 30 minutes before re-use.
Troubleshooting
If problems still occur despite the following checks, send the Inhalator for repair to the address appropriate for you.
| Problem cause/solution | |
| The Inhalator does not react when the ON/OFF button is pressed | Check that the power line adapter/battery is connected to the Inhalator correctly. Check that the water tank has been properly filled. Check that the battery is properly charged. |
| Low or no nebulization Make sure that the medicament cup is sufficiently full (min. 2ml - max. 5ml) and is not broken. Check that the Inhalator under the medicament cup is clean. | |
| The fan is not operating correctly Check that the fan is securely and correctly connected to the motor shaft. | |
Cleaning and storage
Disconnect the power supply before cleaning. Do not immerse the Inhalator in water; it could be irreparably damaged. On each occasion after use, we advise you to clean the Inhalator as follows:
- Remove the top cover, the medicament cup, air filter and filter case (pull down and out from the rear protective sheath) and clean these parts with a damp cloth and a mild detergent or disinfectant according to the manufacturer's instructions. Clean the extension tubing and inhalation masks in the same manner.
- Clean the case with a damp, soft cloth and exclusively with water. Rinse the water tank thoroughly with clear water.
- Use only a soft, dry cloth to clean the small fan and the air inflow under the rear cover. Only the cover can be sterilized with boiling water. The Inhalator, the power line adapter and the rechargeable battery must not be exposed to direct sunlight, high temperatures or dampness.
Charging the battery
- Plug the small plug of the power line adapter into the socket underneath the battery.
- Now plug the power plug of the power line adapter into the socket outlet. The battery indicator lamp lights. For first use and after a long period of non-use the charging time is approx. 24 hours, otherwise 10 - 12 hours according to the state of discharge of the battery.
- Do not place the battery in metal containers, on any kind of metal or on wet surfaces. This could cause the battery to be short-circuited with subsequent over-heating and possibly cause an explosion.
Disposal
Please dispose of the blanket in accordance with the directive 2002/96/EG - WEEE (Waste Electrical and Electronic Equipment). If you have any queries, please refer to the local autho- rities responsible for waste disposal.

Technical data: Inhalator
| Power input: approx. 10 Watt | |
| Ultrasonic frequency: 2.5 MH | |
| Nebulization rate: | 0.2 to 0.7 ml/min according to medicament density |
| Particle size: 5 μm MMAD | |
| Med. cup capacity: max. 5 ml | |
| Automatic shutdown: after approx. 10 min. | |
| Storage conditions: -10°C to+50°C; 45% to 85% atmospheric humidity | |
| Operating conditions: +15°C to+30°C; 45% to 65% atmospheric humidity | |
| Dimensions: 65 x 50 x 140 mm | |
| Weight: approx. 185g | |
| Accessories: | power line adapter, re-chargeable battery, adult/child mask, extension tube, protective bag, 5 medicament cup |
Re-chargeable battery:
| Type: Ni-Mh | |
| Rated voltage: 12V DC | |
| Charging current: 40 to 120 mA | |
| Charging time: initial charge min. 24 hrs. normal 10 to 12 hrs. | |
| normal | 10 to 12 hrs. |
| Rated capacity: | 650 mAh |
| Storage conditions: | -18°C to +45°C |
| Operating conditions: | +15°C to +30°C |
| Weight: | 183g |
Replacement items:
| Designation REF | |
| Filter + medication container 071.00 | |
Power line adapter
| Input voltage: 230V 50Hz | |
| Output voltage: 12V DC | |
| Rated current: 1.1A | |
| Rated power: 13.2 VA | |
| Protective class: | II ☑ |
Note:
If the unit is used outside of the specifications, proper function is no longer guaranteed! We reserve the right to make technical changes to improve and further develop the product. This device and its accessories comply with European standards EN60601-1 and EN60601-1-2, as well as EN13544-1, and is subject to special safety measures in terms of electromagnetic tolerance. Note that portable and mobile RF communication equipment can affect this unit. More details can be requested from the stated Customer Service address or found at the end of the instructions for use. The unit conforms to the requirements of the European Directive for Medical Products 93/42/EC, the MPG (German Medical units Act).
Electromagnetic compatibility
- The device complies with current specifications with regard to electromagnetic compatibility and is suitable for use in all premises, including those designated for private residential purposes. The radio frequency emissions of the device are extremely low and in all probability do not cause any interference with other devices in the proximity.
- It is recommended that you do not place the device on top of or close to other devices. Should you notice any interference with other electrical devices, move the unit or connect it to a different socket.
- Radio equipment may affect the operation of this device.
FRANÇAIS
Madame, Monsieur
Batterie rechargeable :
Ybaxaemblnokynatelb,
MbI 6IaIopMm Bac 3a BbIbOp npOdyKuHn HaWei npMbI. Mbl npOn3BODm COBpeMeHHbIe, TzataJIbHO IpOTecTIPOBaHHbIe, BbICOKOKaueCTBeHHbIe N3dJIINy IJRA O6OrpeBa, MraKoI TepaNN, IN3MepeHnMaCCbI, apTePnaJIbHO rABJIeHNy, IJRA DnArHocTnKn, MaCCaJaN OuchTKn BO3duYa. BHMATEJIbHO npOHTte 3Ty IHCTpyKUHIO n CJeDyInTe yKa3aHnAIM.
C HannyuHmN noXeJahnAMyN, KomnaHn Beurer
PekomehdaunK npimehenHIO
Даньи Игалларop pekomendуETСдЯ leueHn acTmbi, xpoHnuecknx 6poHXNTOB
I npOuHx pecnnpaTopnbix 3abOJIeBAHn. IcNoIb3yEma HOBeIshaYbItpa3ByKOBa
TexHONORn OecneUBAeT aΦΦeKTINBHyO INHraJIaCIO BepXHIX N HIXHIX DbIXaTeJIbHbIX
nyTe C npIMeHNem a3po3OJIbHbIX leue6HbIX npenapatob. TakaA INHraJIaCnno3BOJAE
IpeDToBpaTITb 3abOJIeBAHn pecnnpaTopHO TpaKaTa, NOdABJIeT COpOBoKDaIOUcne 3TN
3abOJIeBAHn CNMTOMbl N yCKOPaET Bbl3OpOBLeHne.
Bolee noPpO6Hyo INΦopMaun Ho Na cel Chet Bbl MoKeTe nOLyHTb y CBOero BpaayuN
fapMaueBTA. YCTPOINCTBO IO3BOJAE TPOBODITb INHraJIaCm KAc B DOMaSHNX ycIOBnX, TaK
N B POe3dkax. PpOBeHeNe INHraJIaCm Heo6XoDIMO BblONHrTb JInMb NO peKOMEnDAcIyM N
Iod HaI3OpOM Bpaay. BblONHЯITE INHraJIaCm B cNOKOIHOM n paccna6JeHHom COCTOHN,
BdIxaiTe MeIeHNO n rIy6OKo, qTObI JekapCTBeHHbI npenapat MOr npOHnKaTb B 6poHXn KaK
MOxHo rIy6Ke. BblIxaiTe HopMaJIbHO.
Ha ynaKOBke n Ha 3aBODCKoT a6nUKe np6opa n npHaJnxHocTe NcNoIb3yOTc
CleDyUOuHe CmBOJbl.
HanoJIHeHne emKoCTn dJa MeDnKaMeHTOB
- YCTaHOBNTe EMKOCTb IJRA MeINKaMeHTOB Ha EMKOCTb IJRA BOIbl.
- HanibaTe JekapCTBO nprMo B emKocTb. He nepeJeIe!
MaKcImaIbHbI yPoBeHb 3aNoJIHeHnA-5Mn!
- PpOeDpya DOnxHa npOdoJkaTbCra COOTBeTCTByIOoee BpeM.
- Ecnn npednncanha do3a lekapctba coctablaet Mehee 2 Ml, IOBeHnte ero ypoBHeb do, no Mehbwe Mepe, 4Ml, pa36abN BINCTUNIPOBAHHO BODOn NnN 30TOHueCKIM cOJHBm pactbOpom! B83Kne IekapctBa TaKxe DoJXhbl 6bITb pactbOpEnbI. B clyuaynx nCNoJIb3OBAHnra TaKOBbIX, TaKxe CJeNyTe npednncanm Bpaua.
- YcTaHOBInTe Ha MeCTO BepXHIOU KpbIiUKy I y6eINTECb, YTO OHa IIOTHO BCTaJa Ha MeCTO.
Kak Tolbko Ha HakoHeuHke EMKocTn dIe MeiKamEHTOB NOBraTc np3HaKn 13Hoca (TpueHHbl, 6elbIe MetKN), Heo6xOIMo OTka3aTbCra OT ee NcNoJIb30BaHna. He nCpOJIb3yIte EMKocTb 6OJee Tpex ceaHcoB.
Bo3MOxHoe nCNoJIb3OBAHne INrAJaTopa c Akceccyapamn
INHraJIaTOp MoXeT NcNoIb3OBAbTcR KaK C MaCKoI, TaK I 6e3 Hee.ДЯ npOBeHnN INHraJIaCmI DeTAM peKOMeHNyETcR NcNoJIb3OBAHnE MaCKn.Пpr npOBeHnN OINcMbIAembIX DaJIee npOceDyp No3a60TbTeCb, YTO6bl INHraJIaTOp He HaKIOHJaC R BO I36exKaHne CMeuHBaHnJ leKapCTBeHHoro npenapata c BODoI.
1.Y6eNTecbB TOM,HTO INHraJTop BbIKIOueH.
2. BCTaBbTe KopoTkyo 6enyu Tpy6ky INHraJauOnHOm Mackn (IJIy DeTeN IcNoJb3yeTcMaCKa MeHbJero pa3Mepa) B OTKpbItbI KoHeU Tpy6Kn-ynHInHTeJI.
3. BtopoJ KOHeTpy6Kn-udHnHTeJI BCTaBbTe B OTBepCTne BepxHeK KpbIuKN, JERKO npotaIKNBAra TlaAdkN KOHeU DO MOMeHTa, KOrDa yCJIbIuNTe UeLqOK, CBnDeTeJIbCTByIOuN O fHKcaun.
Pa6ota
Ipeed BKJIIOUeHHeM INHraJIaTopa Heo6XoJIMO y6eINTbC8 B TOM, YTO yCTpoiCTBO HAXODITcB BEPTNkAJbHOM NIOJOxEHHN I, CJIeIOBaTeJIbHO, 6ydt pa60TaTb C MaKcImaJIbHOH 3ΦΦeKTNUBHOCTbIO.
- YTo6bI BkIIOHTb INHraJIaTOp, HaxMITE KHOJKy ON/OFF. Iprn 3Tom INHdIKaTOPHaJ lamna CBETITcR 3eJIeHbIM CBEtOM.
- Otperynpyte pnlbTp, paonoloxeHHbI Ha TblbHoi CTOpOHe INHraJrTopa, taKIM o6pa30M, yTo6bl obecneuBancra docTaTOUHbI nOTOK BO3dyxa. B noLoxeHm fNl8tpa dJa MaKcImaJIbHO rNOTOK aTKePTna.
- Пи Келанн Дд Игалци может Испьзоваься мackа. Пи Испьзован Мackи pekomeHdyetcдышатb ртOM Дд ГлбokOTO поннховеня napOB B дixaTeHBhepyTN. EcIn Heo6xOIMa INrAЯци HOCOBОД NOLOCTN, BdIXaIte Ype3 Hoc, a BblbIXaIte Ype3 pot.
- Истору Скоковский Доступа ведим的基础上,在我国的国际社会上,俄国在1900年以后一直保持着高度的国际地位。
- П配电ашени Выдени Ингалуюserо napa уka3bBaet Ha To, YTO emKocTb Дя MeDNkamenta nycta. Рп NTOM поцетусу сеуET р配电оть, ТAK KaK yCTPOICTBO BvIKLOUaETc. (КрOME TOrO, Ингалутор abTomatueckn OTKLOUaETc cnyctr 10 MInHyT POCJIe BKIOUeHnA).
- Дя Вькюеця усточа Heохдимо NOВТОнгу Нжати ON/ OFF. Пп NTOM ИндКаTOHЯ Лмпа ratchet. OTКИчHTe aanTep ot po3etKu cetn 3JIeKTPoNTaHЯ IN BbITAHTe MaIeHbKN pa3bem n3 rHe3da, paCNoLOXeHHORO B HIXHNe YactN INHraJToPA.
- CneIte ocTaTkn IeKapCTBeHHoro npenapaTa n BoDbI (OCTOPOXHO: TOPAHO!) n OunCTnTe yCTpoiCTBO B COOTBeTCTBmC INHΦopMaUneB NyHKTe „OunCTka". Npeed NOBTOpHBIM NCNoJIb3OBaHNEM Heo6xOДMоДaTb yCTpoiCTBy B TeueHne 30 MInHyT.
Bo3MOxHbIe HeucnpaBHOCTu
Ecni, HeCMOTpHa OTCyTCTBnE BHeuHnx NOBpeKdHn IIN DeΦeKTOB, yCTPOIcTBO BCE Jx He pa6oTaET, OTouHnTe erO npedCTaBntelIO npOn3BODntela.
For all ME EQUIPMENT and ME SYSTEMS
| Guidance and manufacturer's declaration - electromagnetic emissions | ||
| The IH30 is intended for use in the electromagnetic environment specified below. The customer or the user of the IH30 should assure that it is used in such an environment. | ||
| Emissions test Compliance | Electromagnetic environment - guidance | |
| RF emissions CISPR 11 | Group 1 | The IH30 uses RF energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. |
| RF emissions CISPR 11 Class B The IH30 is suitable for use in all establishments, including domestic establishments and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes. | ||
| Harmonic emissions IEC 61000-3-2 | Class A | |
| Voltage fluctuations/flicker emissions IEC 61000-3-3 | Complies | |
Table 2
For all ME EQUIPMENT and ME SYSTEMS
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The IH30 is intended for use in the electromagnetic environment specified below. The customer or the user of the IH30 should assure that it is used in such an environment. | |||
| IMMUNITY test | IEC 60601 test level | Compliance level | Electromagnetic environment - guidance |
| Electrostatic discharge (ESD) IEC 61000-4-2 | ± 6 kV contact ± 8 kV air | ± 6 kV contact ± 8 kV air | Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30 %. |
| Electrical fast transient/burst IEC 61000-4-4 | ±2 kV for power supply lines | ±2 kV for power supply lines | Mains power quality should be that of a typical commercial or hospital environment. |
| Surge IEC 61000-4-5 | ±1 kV line(s) and neutral | ±1 kV line(s) and neutral | Mains power quality should be that of a typical commercial or hospital environment. |
| Voltage dips, short interruptions and voltage variations on power supply input lines IEC 61000-4-11 | <5 % U_T (>95 % dip in U_T) for 0,5 cycle 40 % U_T (60 % dip in U_T) for 5 cycles 70 % U_T (30 % dip in U_T) for 25 cycles <5 % U_T (>95 % dip in U_T) for 5s | <5 % U_T (>95 % dip in U_T) for 0,5 cycle 40 % U_T (60 % dip in U_T) for 5 cycles 70 % U_T (30 % dip in U_T) for 25 cycles <5 % U_T (>95 % dip in U_T) for 5s | Mains power quality should be that of a typical commercial or hospital environment. If the user of the IH30 requires continued operation during power mains interruptions, it is recommended that the IH30 be powered from an uninterruptible power supply or a battery. |
| Power frequency (50/60 Hz) magnetic field IEC 61000-4-8 | 3 A/m 3 A/m | Power frequency magnetic fields should be at levels characteristic of a typical location in a typical commercial or hospital environment. | |
| NOTE: U_T is the a.c. mains voltage prior to application of the test level. | |||
Table 3
For ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The IH30 is intended for use in the electromagnetic environment specified below. The customer or the user of the IH30 should assure that it is used in such an environment. | |||
| IMMUNITY test | IEC 60601 test level | Compliance level | Electromagnetic environment - guidance |
| Conducted RF IEC 61000-4-6 Radiated RF IEC 61000-4-3 | 3 Vrms 150 kHz to 80 MHz3 V/m 80 MHz to 2.5 GHz | 3 Vrms3 V/m | Portable and mobile RF communications equipment should be used no closer to any part of the IH30, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter.Recommended separation distance:d=1.2√Pd=1.2√P80 MHz to 800 MHzd=2.3√P 800 MHz to 2.5 GHzWhere P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and d is the recommended separation distance in meters (m).Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey, a should be less than the compliance level in each frequency range. bInterference may occur in the vicinity of equipment marked with the following symbol: |
| NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies.NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||
| aField strengths from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the IH30 is used exceeds the applicable RF compliance level above, the IH30 should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as re-orienting or relocating the IH30.bOver the frequency range 150 kHz to 80 MHz, field strengths should be less than 3 V/m. | |||
Table 4 For ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
| Recommended separation distances between portable and mobile RF communications equipment and the IH30 | |||
| The IH30 is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the IH30 can help electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and the IH30 as recommended below, according to the maximum output power of the communications equipment. | |||
| Rated maximum output power of transmitter (W) | Separation distance according to frequency of transmitter (m) | ||
| 150 kHz to 80 MHz d = 1.2 √P | 80 MHz to 800 MHz d = 1.2 √P | 800 MHz to 2.5 GHz d = 2.3 √P | |
| 0.01 0.12 0.12 0.23 | |||
| 0.1 0.38 0.38 0.73 | |||
| 1 1.2 1.2 2.3 | |||
| 10 3.8 3.8 7.3 | |||
| 100 12 12 23 | |||
| For transmitters rated at a maximum output power not listed above, the recommended separation distance d in meters (m) can be determined using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer. NOTE 1 At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies. NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||