BC 54 - Blood pressure monitor BEURER - Free user manual and instructions
Find the device manual for free BC 54 BEURER in PDF.

User questions about BC 54 BEURER
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Blood pressure monitor in PDF format for free! Find your manual BC 54 - BEURER and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. BC 54 by BEURER.
USER MANUAL BC 54 BEURER
EN Blood pressure monitor Instructions for use 23
Notes on electromagnetic compatibility .....166
DEUTSCH

natural_image
Line drawings of four different types of mugs or containers, showing front and side views with no text or symbols.natural_image
Silhouette of a person sitting with heart symbols on their arm, holding a hand near a table (no text or symbols present)Quelle: WHO, 1999 (World Health Organization)
Read these instructions for use carefully and keep them for later use, be sure to make them accessible to other users and observe the information they contain.
Dear customer,
thank you for choosing a product from our range. Our name stands for high-quality, thoroughly tested products for applications in the areas of heat, weight, blood pressure, body temperature, pulse, gentle therapy, massage, beauty and air.
With kind regards, your Beurer team
Table of contents
- Included in delivery .....24
- Signs and symbols....24
- Proper use....25
- Warnings and safety notes ....26
- Device description....28
-
Initial use ....29
-
Usage ......31
- Cleaning and maintenance....38
- What if there are problems? ....38
- Disposal....39
- Technical specifications ....40
- Warranty / Service 41
1. Included in delivery
Check that the exterior of the cardboard delivery packaging is intact and make sure that all contents are present. Before use, ensure that there is no visible damage to the device or accessories and that all packaging material has been removed. If you have any doubts, do not use the device and contact your retailer or the specified Customer Service address.
1 x Wrist blood pressure monitor with cuff
1 x Instructions for use
1 x Quick Start Guide
1 x Storage box
2 x 1.5V AAA batteries LR03
2. Signs and symbols
The following symbols are used on the device, in these instructions for use, on the packaging and on the type plate for the device:
 | WARNINGWarning instruction indicating a risk of injury or damage to health |
 | CAUTIONSafety note indicating possible damage to the device/accessory |
 | Product informationNote on important information |
 | Observe the instructionsRead the instructions before starting work and/or operating devices or machines |
 | Isolation of applied partsType BFGalvanically isolated application part (F stands for “floating”); meets the requirements for leakage currents for type B |
| KKHG] | Direct currentThe device is suitable for use with direct current only |
 | Disposal in accordance with the Waste Electrical and Electronic Equipment EC Directive – WEEE |
 | Do not dispose of batteries containing hazardous substances with household waste |
 | Separate the packaging elements and dispose of them in accordance with local regulations. |
 | Marking to identify the packaging material. A = Material code, B = Material number: 1-7 = Plastics, 20-22 = Paper and cardboard |
| Separate the product and packaging elements and dispose of them in accordance with local regulations. | |
 | Manufacturer |
 | Temperature limitThe temperature limit values to which the medical device can safely be exposed are indicated. |
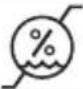 | Humidity, limitIndicates the humidity range to which the medical device can safely be exposed. |
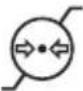 | Atmospheric pressure, limitIndicates the range of atmospheric pressures to which the medical device can be safely exposed |
| IP22 | IP classDevice protected against foreign objects ≥ 12.5 mm and against water dripping at an angle |
 | Serial number |
 | CE labellingThis product satisfies the requirements of the applicable European and national directives. |
 | Medical device |
| REF | Item number |
3. Proper use
Intended use
The blood pressure monitor is intended for the fully automatic, non-invasive measurement of arterial blood pressure and pulse values on the wrist.
Target group
It is designed for self-measurement by adults in the home environment and is suitable for users whose wrist circumference is within the range printed on the cuff.
Indication/clinical benefits
The user can record their blood pressure and pulse values quickly and easily using the device. The recorded values are classified according to internationally applicable guidelines and evaluated graphically. Furthermore, the device can detect any irregular heart beats that occur during measurement and inform the user via a symbol in the display. The device saves the recorded measurements and can also output average values of previous measurements.
The recorded data can provide healthcare service providers with support during the diagnosis and treatment of blood pressure problems, and therefore plays a part in the long-term monitoring of the user's health.
4. Warnings and safety notes

Contraindications
- Do not use the blood pressure monitor on newborns, children or pets.
• People with restricted physical, sensory or mental skills should be supervised by a person responsible for their safety and receive instructions from this person on how to use the device. - If you have any of the following conditions, it is essential you consult your doctor before using the device: cardiac arrhythmia, circulatory problems, diabetes, pregnancy, pre-eclampsia, hypotension, chills, shaking.
• People with pacemakers or other electrical implants should consult their doctor before using the device. - The blood pressure monitor must not be used in connection with a high-frequency surgical unit.
- Do not use the cuff on people who have undergone a mastectomy.
- Do not place the cuff over wounds as this may cause further injury.
- Make sure that the cuff is not placed on an arm in which the arteries or veins are undergoing medical treatment, e.g. intravascular access or intravascular therapy, or an arteriovenous (AV) shunt.

General warnings
- The measurements taken by you are for your information only – they are no substitute for a medical examination! Discuss the measured values with your doctor and never make your own medical decisions based on them (e.g. regarding dosages of medicines).
- The device is only intended for the purpose described in these instructions for use. The manufacturer is not liable for damage resulting from improper or incorrect use.
- Using the blood pressure monitor outside your home environment or whilst on the move (e.g. whilst travelling in a car, ambulance or helicopter, or whilst undertaking physical activity such as playing sport) can influence the measurement accuracy and cause incorrect measurements.
- Cardiovascular diseases may lead to incorrect measurements or have a detrimental effect on measurement accuracy.
- Do not use the device at the same time as other medical electrical devices (ME equipment). This could lead to a malfunction of the measuring device and/or an inaccurate measurement.
-
Do not use the device outside of the specified storage and operating conditions. This could lead to incorrect measurements.
-
Only use the cuffs included in delivery or cuffs described in these instructions for use for the device. Using another cuff may lead to measurement inaccuracies.
- Note that when inflating the cuff, the functions of the limb in question may be impaired.
- Do not perform measurements more frequently than necessary. Due to the restriction of blood flow, some bruising may occur.
- During the blood pressure measurement, the blood circulation must not be stopped for an unnecessarily long time. If the device malfunctions remove the cuff from the arm.
- Place the cuff on your wrist only. Do not place the cuff on other parts of the body.

General precautions
- The blood pressure monitor is made from precision and electronic components. The accuracy of the measurements and service life of the device depend on its careful handling.
- Protect the device from impacts, moisture, dirt, marked temperature fluctuations and direct sunlight.
-
Ensure the device is at room temperature before measuring. If the measuring device has been stored close to the maximum or minimum storage and transport temperatures and is placed in an environment with a temperature of 20^ C, it is recommended that you wait approx. 2 hours before using the measuring device.
-
Do not drop the device.
- Do not use the device in the vicinity of strong electromagnetic fields and keep it away from radio systems or mobile telephones.
- We recommend that the batteries be removed if the device will not be used for a prolonged period of time.
Measures for handling batteries

- If your skin or eyes come into contact with battery fluid, rinse the affected areas with water and seek medical assistance.
- Choking hazard! Small children may swallow and choke on batteries. Therefore, store batteries out of the reach of small children.
- Risk of explosion! Do not throw batteries into a fire.
- If a battery has leaked, put on protective gloves and clean the battery compartment with a dry cloth.
- Do not disassemble, open or crush the batteries.

- Observe the plus (+) and minus (-) polarity signs.
- Protect batteries from excessive heat.
- Do not charge or short-circuit batteries.
-
If the device is not to be used for a long period of time, remove the batteries from the battery compartment.
-
Use identical or equivalent battery types only.
• Always replace all batteries at the same time. - Do not use rechargeable batteries.

Notes on electromagnetic compatibility
- The device is suitable for use in all environments listed in these instructions for use, including domestic environments.
- The use of the device may be limited in the presence of electromagnetic disturbances. This could result in issues such as error messages or the failure of the display/device.
- Avoid using this device directly next to other devices or stacked on top of other devices, as this could lead to faulty operation. If, however, it is necessary to use the device in the manner stated, this device as well as the other devices must be monitored to ensure they are working properly.
- The use of accessories other than those specified or provided by the manufacturer of this device can lead to an increase in electromagnetic emissions or a decrease in the device's electromagnetic immunity; this can result in faulty operation.
- Failure to comply with the above can impair the performance of the device.
5. Device description
- Display
- START/STOP button ①
- Memory button M1
- Memory button M2
- Wrist cuff
- Battery compartment lid
- Risk indicator

text_image
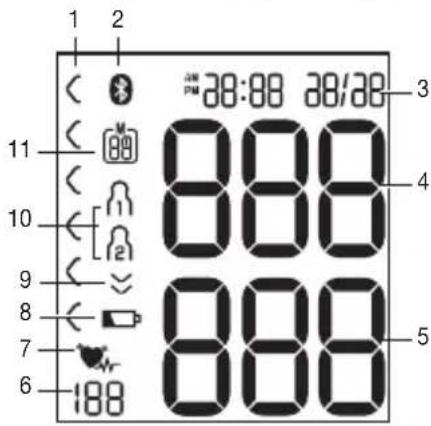
beurer 1 2 3 4 5 6 7Information on the display:
- Risk indicator
- Symbol for Bluetooth® transfer
- Time and date
- Systolic pressure
- Diastolic pressure
- Calculated pulse value
-
Cardiac arrhythmia symbol 🤨 Pulse symbol ♥
-
Battery status indicator
-
Release air
-
User memory
-
Memory space number/memory display average value (R), morning (RMT), evening (PMT)
text_image
1 2 11 10 9 8 7 6 88 88 88 3 4 56. Initialuse
Inserting the batteries
- Remove the battery compartment lid on the left side of the device.
- Insert two 1.5 V AAA
micro (alkaline type LR03) batteries. Make sure that the batteries are inserted the correct way round. Do not use rechargeable batteries. - Close the battery compartment lid again carefully.
Now set the date and time as described below.

natural_image
Three line drawings of a mug with handles and a partially open lid, shown from different angles (no text or symbols)If the battery change symbol is flashing and Er6 appears, no further measurements are possible and you must replace all batteries. Once the batteries have been removed from the device, the date and time must be set again. Any saved measured values are retained.
Making settings
You must make sure that the device has the correct settings before use in order to be able to make full use of all functions. Only by doing so can your measurements with associated date and time be saved and accessed later by you.
There are two different ways to access the menu from which you can adjust the settings:
- Before initial use and after each time you replace the battery:
When inserting batteries into the device, you will be taken to the relevant menu automatically.
- If the batteries have already been inserted:
Press and hold the START/STOP button on the device when switched off for approx. 5 seconds.
In this menu you can adjust the following settings in succession:
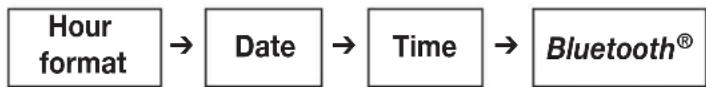
flowchart
graph LR
A["Hour format"] --> B["Date"]
B --> C["Time"]
C --> D["Bluetooth®"]
It is essential to set the date and time. Otherwise, you will not be able to save your measured values correctly with a date and time and access them again later.
Hour format
The hour format now flashes on the display.
- Select the desired hour format using the memory button M1 or M2 and confirm with the START/STOP button ⏻

Date
The year flashes on the display.
- Select the desired year using the memory button M1 or M2 and confirm with the START/STOP button ①.
The month flashes on the display.
- Select the desired month using the memory button M1 or M2 and confirm with the START/STOP button ①.
The day flashes on the display.
- Select the desired day using the memory button M1 or M2 and confirm with the START/STOP button ①.
If the hour format is set as 124, the day/month display sequence is reversed.
20 19
/
-11
The hours flash on the display.
- Select the desired hour using the memory button M1 or M2 and confirm with the START/STOP button ①.
The minutes flash on the display.
- Select the desired minute using the memory button M1 or M2 and confirm with the START/STOP button ⏻
The Bluetooth ^® symbol is shown in the display.
- Use the memory button M1 or M2 to select whether automatic Bluetooth® data transfer is activated (Bluetooth® symbol is displayed) or deactivated (Bluetooth® symbol is not shown) and confirm with the START/STOP button①
i Bluetooth® transfers will reduce the battery life.

7. Usage
General rules when measuring blood pressure yourself
- In order to generate as informative a profile of the progression of your blood pressure as possible and ensure that the measured values can be compared, you should measure your blood pressure regularly and always at the same times of day. It is recommended that you measure your blood pressure twice a day: once in the morning after getting up and once in the evening.
- You should always carry out the measurement when you are sufficiently physically rested. You should therefore avoid taking measurements during stressful periods.
- Do not take a measurement within 30 minutes of eating, drinking, smoking or exercising.
- Before the initial blood pressure measurement, make sure always to rest for about 5 minutes.
- Furthermore, if you want to take several measurements in succession, make sure always to wait for at least 1 minute between the individual measurements.
- Repeat the measurement if you are unsure of the measured value.
Attaching the cuff
- Fundamentally, blood pressure can be measured on both arms. Certain deviations between the measured blood pressure on the right arm and left arm are due to physiological causes and completely normal. You should always perform the measurement on the arm with the highest blood pressure values. Before starting self-measurement, consult your doctor in this regard. From this point on, always take measurements on the same arm.
- The device may only be operated with the cuff attached when supplied. Before using the device, the user should check the fit of the cuff and, in doing so, ensure that their wrist circumference is within the range printed on the cuff.
- Uncover your wrist. Ensure that the circulation of the arm is not hindered by tight clothing or similar.
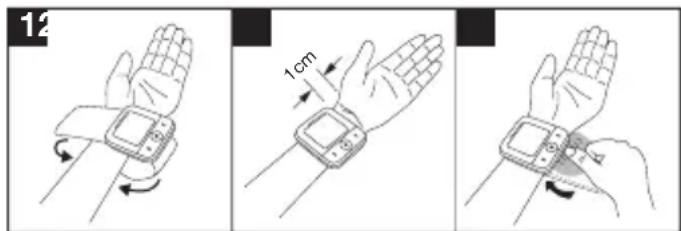
- Now place the cuff on the wrist so that the palm of your hand and the device display are facing upwards.
- Position the cuff so that there is a distance of 1.0 - 1.5 cm between it and the heel of your hand.
- Now fasten the cuff tightly around your wrist using the hook-and-loop fastener. Make sure that it is tight but that it does not cut into your wrist.
Adopting the correct posture
- To carry out a blood pressure measurement, make sure you are sitting upright and comfortably. Lean back and place your arm on a surface. Do not cross your legs. Place your feet next to each other flat on the floor.
- Always make sure that the device is at heart level during the measurement. Otherwise significant measure-
ment deviations can occur due to physiological causes. To do so, place your elbow on a table to support your arm. In order to make the measurement even more comfortable for you, you can place your lower arm on a suitable object (e.g. t

natural_image
Silhouette of a person holding a heart with hands placed on the chest (no text or symbols)- Relax your arm and the palm of your hand.
- To avoid distorting the measurement, you should remain as still as possible during the measurement and not speak.
Selecting the user
You have 2 memories, each with 60 memory spaces, to enable the separate storage of measurements for 2 different people.
To select the corresponding user memory, press the M1 memory button (for user 📄) or M2 (for user 🔒 on the switched-off device. Confirm your selection by pressing the START/STOP button ⏻
Performing the blood pressure measurement
As described above, attach the cuff and adopt the posture in which you want to perform the measurement.
- Press the START/STOP button ① to start the blood pressure monitor. All displays will illuminate briefly.
The blood pressure monitor will begin the measurement automatically after 5 seconds.
The cuff automatically inflates. The measurement itself is taken during the inflation phase.
As soon as a pulse is found, the pulse symbol ♥ displayed.
You can cancel the measurement at any time by pressing the START/STOP button
- The systolic pressure, diastolic pressure and pulse rate measurements are displayed.
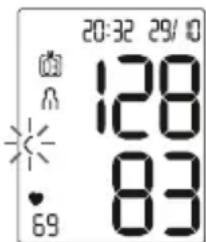
text_image
20:32 29/10 128 83 69Er_ appears if the measurement could not be performed properly. Observe the chapter „9. What if there are problems?“ in these instructions for use measurement.

- Press the START/STOP button ① to switch off the blood pressure monitor. The measurement is then stored in the selected user memory. If Bluetooth® data transfer is activated, data is transferred after pressing the START/STOP button ①. TheBluetooth® symbol flashes on the display. The blood pressure monitor now attempts to connect to the app for approx. 30 seconds. TheBluetooth® symbol stops flashing as soon as a connection is established. All measurement data is automatically transferred to the app. Once the data has been successfully transferred, the device switches off.
- If a connection to the app cannot be established after 30 seconds, the Bluetooth® symbol goes out and the
blood pressure monitor switches off automatically after 5 seconds.
- If you forget to turn off the device, it will switch off automatically. In this case too, the value is stored in the selected or most recently used user memory.
Evaluating the results
General information about blood pressure
- Blood pressure is the force with which the bloodstream presses against the arterial walls. Arterial blood pressure constantly changes in the course of a cardiac cycle.
- Blood pressure is always stated in the form of two values:
- The highest pressure in the cycle is called systolic blood pressure. This arises when the heart muscle contracts and blood is pumped into the blood vessels.
- The lowest is diastolic blood pressure, which is when the heart muscle has completely stretched back out and the heart fills with blood.
- Fluctuations in blood pressure are normal. Even during repeat measurements, considerable differences between the measured values may occur. One-off or irregular measurements therefore do not provide reliable information about the actual blood pressure. Reliable assessment is only possible when you perform the measurement regularly under comparable conditions.
Risk indicator
The World Health Organization (WHO) has defined the internationally recognised classification for the evaluation of measured blood pressure values listed in the table below:
| Measured blood pressure value range | Classification | Colour of the risk indicator | |
| Systole (in mmHg) | Diastole (in mmHg) | ||
| ≥180≥110 | High blood pressure stage 3 (severe) | Red | |
| 160–17910 | 0–109 | High blood pressure stage 2 (moderate) | Orange |
| 140–15990 | -99 | High blood pressure stage 1 (mild) | Yellow |
| 130–13985 | -89 High normal | Normal Green | |
| 120–12980 | -84 Normal | Green | |
| <120<80Optimal Green | |||
Source: WHO, 1999 (World Health Organization)
The risk indicator (the arrow in the display and the associated scale on the device) shows which category the recorded blood pressure values fall into. If the measured values are in two different classifications (e.g. systole in the high normal category and diastole in the normal category), the risk indicator then always shows you the higher category – “high normal” in the example described.
Please be aware that these standard values can only serve as a general guideline, as the individual blood pressure varies in different people and different age groups, etc.
Furthermore, it must be noted that measurements taken yourself while at home are generally lower than those that are taken by the doctor. For this reason, it is important that you regularly consult your doctor for advice. Only they are able to give you your personal target values for controlled blood pressure – in particular if you receive medicinal therapy.
Cardiac arrhythmia
This device can identify any cardiac rhythm disturbances as part of the analysis of your recorded pulse signal during blood pressure measurement. In this case, after the measurement, the device will indicate any irregularities in your pulse by displaying the symbol in the display. This can be an indicator for arrhythmia. Arrhythmia is an illness in which the heart rhythm is abnormal because of flaws in the bioelectrical system that regulates the heartbeat. The symptoms (skipped or premature heart beats, pulse being slow or too fast) can be caused by factors such as heart disease, age, physical disposition, excess alcohol and tobacco, stress or lack of sleep. If the symbol appears on the display after the measurement, the measurement must be repeated as the measurement accuracy may be impaired. To assess your blood pressure, only use the results that have been recorded without corresponding irregularities in your pulse. If the symbol appears frequently, please consult your doctor. Only they can establish the existence of an arrhythmia during a checkup, using their means of diagnosis.
Saving, displaying and deleting measured values
User memory
The results of every successful measurement are stored together with the date and time. The oldest measurement is overwritten in the event of more than 60 measurements.
- To select the corresponding user memory, press the M1 memory button (for user 🔒) or M2 (for user 🔒) on the switched-off device. Confirm your selection by pressing the START/STOP button ⏻
- If Bluetooth ^ is activated (the 8 symbol flashes on the display), the blood pressure monitor attempts to connect to the app. The buttons are deactivated and the 3 symbol ceases to flash as soon as a connection is established and the data is transferred.
If you press the memory button M1 or M2 in the meantime, the transfer is cancelled. The symbol is no longer shown.
HAs displayed.
The average value of all saved measured values in this user memory is displayed.
- Press the memory button M1.
ARTAS displayed.
The average value of the morning measurements for the last 7 days is displayed (morning: 5.00 a.m. - 9.00 a.m.).
- Press the memory button M1.
PPR displayed.
The average value of the evening measurements for the last 7 days is displayed (evening: 6.00 p.m. - 8.00 p.m.).



Individual measured values
- If you press the memory button M1 again, the last individual measurement is displayed (in this example, measurement 03).

- If you press the memory button M1 once more, you can view your individual measured values.
- To switch the device off again, press the START/STOP button Ⓞ
You can exit the menu at any time by pressing the START/STOP button.
- To erase a user's memory, first select the user memory to be erased by pressing the M1 or M2 memory button on the off device and confirm your selection by pressing the START/STOP button - The average value of all measurements appears on the display; in parallel, Rights up. - Press and hold down both memory buttons M1 and M2 simultaneously for 5 seconds.
CL00 appears on the display. All the values in the selected user memory have now been deleted.

Transfer via Bluetooth®
It is also possible to transfer the measured values saved on the device to your smartphone via Bluetooth®.
You will need the “ beurer Health Manager” app for this. This is available in the Apple App Store and from Google Play.
System requirements for the app
"beurerHealthManager":
- iOS ≥ 10.0, Android™ ≥ 5.0
- Bluetooth® ≥ 4.0
List of compatible devices:

Proceed as follows to transfer values:
If Bluetooth ^® is activated in the settings menu, the data is transferred automatically after the measurement.
The ⚙ symbol appears in the top left of the display (see chapter „6. Initial use“, section “Bluetooth®”).
When connecting for the first time, a randomly-generated six-digit PIN code is displayed on device, and at the same time an input field appears on the smart-
phone in which you must enter this six-digit PIN code. After successfully entering the code, the device will be connected to your smartphone.

Step 1: BC 54
Activate Bluetooth® on your device (see chapter „6. Initial use“, section “Bluetooth®”).

Step 2: “beurer Health Manager” app In the “beurer Health Manager” app, add the BC 54 under “Settings/My devices”.

Step 3: BC 54
Take a measurement.

Step 4: BC 54:
Data transfer immediately following measurements.
- If Bluetooth ^ data transfer has been activated, data is transferred after having confirmed the user memory and pressing the START/STOP button ①.

Step 4: BC 54:
Data transfer at a later point:
- Go to memory mode (chapter „7. Use“, section „Saving, displaying and deleting measured values“). Select the desired user memory. The Bluetooth® transfer starts automatically.
The “beurer Health Manager” app must be active to allow data transfer.
If your smartphone has a protective cover, remove this to ensure that there is no interference during the transfer. Begin the data transfer in the “beurer Health Manager” app.
We would explicitly draw attention to the fact that the software to hand is not a medical product in accordance with EU Directive 93/42/EEC.
The values displayed are purely for visualisation purposes and must not be used as a basis for therapeutic treatments. The software is not part of a diagnostic-medical system.
8. Cleaning and maintenance
- Clean the device and cuff carefully using a slightly damp cloth only.
- Do not use any cleaning agents or solvents.
- Under no circumstances hold the device and cuff under water, as this can cause liquid to enter and damage the device and cuff.
- If you store the device and cuff, do not place heavy objects on the device and cuff. Remove the batteries.
9. What if there are problems?
| Error message | Possible cause | Solution |
| Er1 | Unable to record a pulse. | Please wait one minute and repeat the measurement.Ensure that you do not speak or move during the measurement. |
| Er2 | You have moved or spoken during the measurement. | |
| Er3 | The cuff was not attached correctly. | Please observe the notes in chapter "7. Usage" under the section "Attaching the cuff". During a new measurement, check whether the cuff can now be inflated properly. |
| Er4 | An error occurred during measurement. | Please wait one minute and repeat the measurement. Ensure that you do not speak or move during the measurement. |
| Er5 | The inflation pressure is higher than 300 mmHg or the measured blood pressure is outside the measuring range. | |
| Er6 | The batteries are almost empty. | Insert new batteries into the device. |
| Error message | Possible cause | Solution |
| Er7 | The data could not be sent via Bluetooth®. | Proceed as described in chapter „7. Usage“ under the section „Transfer via Bluetooth®“ into the manual memory mode and try to restart the data transfer. |
| Er8 | A device error has occurred. | Please repeat the measurement after a pause of one minute. |
If the problems persist despite the proposed corrective actions, please contact customer service.
10. Disposal
i Repairing and disposing of the device
- Do not repair or adjust the device yourself. Proper operation can no longer be guaranteed in this case.
- Do not open the device. Failure to comply will result in voiding of the warranty.
- Repairs must only be carried out by Customer Services or authorised suppliers. Before making a claim, please check the batteries first and replace them if necessary.
- For environmental reasons, do not dispose of the device in the household waste at the end of its useful life. Dispose of the unit at a suitable local collection or recycling point. Dispose of the device in accordance with EC Directive – WEEE (Waste Electrical and Electronic Equipment). If you have any questions, please contact the local authorities responsible for waste disposal.
i Disposing of the batteries
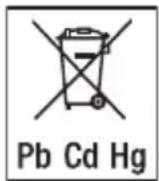
- Batteries must not be disposed of with household waste. They may contain toxic heavy metals and are subject to special waste treatment.
- The codes below are printed on batteries containing harmful substances: Pb = Battery contains lead, Cd = Battery contains cadmium, Hg = Battery contains mercury.
11. Technical specifications
Model no. BC 54
| Measurement method | Oscillometric, non-invasive blood pressure measurement on the wrist |
| Measurement range | Cuff pressure 0-299 mmHg,Systolic 60-230 mmHg,Diastolic 40-130 mmHg,Pulse 40-199 beats/minute |
| Display accuracy Systolic ± 3 mmHg, Diastolic ± 3 mmHg, Pulse ± 5 % of the value shown | |
| Measurement inaccuracy | Max. permissible standard deviation according to clinical testing: Systolic 8 mmHg / Diastolic 8 mmHg |
| Memory 2 x 60 memory spaces | |
| Dimensions L 80.5 mm x W 69.5 mm x H 25 mm | |
| Weight Approximately 116 g (without batteries, with cuff) | |
| Cuff size 135 to 215 mm | |
| Permissible operating conditions | +5 °C to +40 °C, 15-90 % relative humidity (non-condensing), 700-1060 hPa ambient pressure |
| Permissible storage and transport conditions | -20°C to +60°C, ≤ 93% relative humidity (non-condensing) |
| Power supply 2 x 1.5V | —— AAA batteries |
| Battery life For approx. 150 measurements, depending on levels of blood pressure and pump pressure | |
| Software version | A01 |
| Data transfer 2402 | MHz - 2480 MHz frequency bandTransmission power max. 7 dBmThe blood pressure monitor uses Bluetooth® low energy technologyCompatible with Bluetooth® ≥ 4.0 smartphones/tablets |
The serial number is located on the device or in the battery compartment.
Technical information is subject to change without notification to allow for updates.
- This device is in line with European Standard EN60601-1-2(In accordance with CISPR 11, IEC 61000-4-2, IEC 61000-4-3 and IEC 61000-4-8) and is subject to particular precautions with regard to electromagnetic compatibility (EMC). Please note that portable and mobile HF communication systems may interfere with this unit.
- This device corresponds to the EU Medical Devices Directive 93/42/EEC, the German Medical Devices Act (Medizinproduktgesetz) and the standards EN 1060-1 (non-invasive sphygmomanometers, Part 1: General requirements), EN 1060-3 (non-invasive sphygmomanometers, Part3: Supplementary requirements for electro-mechanical blood pressure measuring systems) and IEC 80601-2-30 (Medical electrical equipment –
Part 2-30: Particular requirements for the basic safety and essential performance of automated non-invasive sphygmomanometers).
- The accuracy of this blood pressure monitor has been carefully checked. Calibration is not necessary.
- The device has been developed with regard to a long useful life. The expected service life is 5 years.
- If using the device for commercial medical purposes, it must be regularly tested for accuracy by appropriate means. Precise instructions for checking accuracy may be requested from the service address.
- We hereby confirm that this product complies with the European RED Directive 2014/53/EU. The CE Declaration of Conformity for this product can be found under: www.beurer.com/web/we-landingpages/de/cedecla-rationofconformity.php
12. Warranty / Service
Further information on the guarantee and guarantee conditions can be found in the guarantee leaflet supplied.
FRANÇAIS

Mesures relatives aux piles

natural_image
Line drawings of four different types of mugs or mugs with handle handles, showing internal structure and mounting points (no text or symbols)natural_image
Silhouette of a person with heart symbols on the chest, holding an otoscope (no text or symbols present)Transmission via Bluetooth®
natural_image
Line drawing of three identical mug designs with handles and a partially open lid (no text or symbols)natural_image
Silhouette of a person with heart symbols on their chest, holding a hand near a table (no text or symbols present)"beurer HealthManager"
- iOS ≥ 10.0, Android ^TM ≥ 5.0
- Bluetooth® ≥ 4.0
Paso 2: App "beurer HealthManager"
natural_image
Line drawings of three identical mugs with handles, showing different internal shapes and color variations (no text or symbols)natural_image
Illustration of a human hand wearing a smartwatch with wrist rotation arrows (no text or symbols)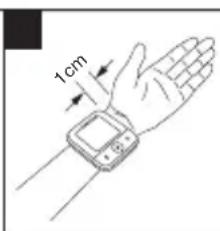
text_image
1 cm
natural_image
Line drawing of a hand wearing a wristwatch with a digital display (no text or symbols)natural_image
Silhouette of a person sitting at a desk with heart symbols on their arm, no text or labels presentPasso 2: App "beurer HealthManager"
natural_image
Line drawing of three identical mugs with handles and a partially inserted insert, shown in side-by-side assembly (no text or symbols)natural_image
Silhouette of a person sitting at a desk with heart symbols on the arm and hands, no text or labels present.natural_image
Line drawings of three identical mugs with handles and a partially inserted insert, shown in side-by-side outlines (no text or symbols)natural_image
Line drawing of a human hand wearing a smartwatch with wrist rotation arrows (no text or symbols)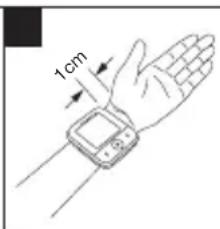
text_image
1cm
natural_image
Line drawing of a hand wearing a wristwatch (no text or symbols)natural_image
Silhouette of a person with heart symbols on their arm, resting on a table (no text or symbols present)natural_image
Line drawings of three identical mugs with handles, showing different internal shapes and color variations (no text or symbols)Wybranie ustawień
natural_image
Silhouette of a person with heart symbols on her hand, holding an object near a table (no text or symbols present)Notes on electromagnetic compatibility
The ME EQUIPMENT or ME SYSTEM is suitable for home healthcare environments and so on.
Warning: Don't near active HF surgical equipment and the RF shielded room of an ME system for magnetic resonance imaging, where the intensity of EM disturbances is high.
Warning: Use of this equipment adjacent to or stacked with other equipment should be avoided because it could result in improper operation. If such use is necessary, this equipment and the other equipment should be observed to verify that they are operating normally.
Warning: Use of accessories, transducers and cables other than those specified or provided by the manufacturer of this equipment could result in increased electromagnetic emissions or decreased electromagnetic immunity of this equipment and result in improper operation.
Warning: Portable RF communications equipment (including peripherals such as antenna cables and external antennas) should be used no closer than 30 cm (12 inches) to any part of the equipment (BC 54), including cables specified by the manufacturer. Otherwise, degradation of the performance of this equipment could result. If any: a list of all cables and maximum lengths of cables (if applicable), transducers and other ACCESSORIES that are replaceable by the RESPONSIBLE ORGANIZATION and that are likely to affect compliance of the ME EQUIPMENT or ME SYSTEM with the requirements of Clause 7 (EMISSIONS) and Clause 8 (IMMUNITY). ACCESSORIES may be specified either generically (e.g. shielded cable, load impedance) or specifically (e.g. by MANUFACTURER and EQUIPMENT OR TYPE REFERENCE). If any: the performance of the ME EQUIPMENT or ME SYSTEM that was determined to be ESSENTIAL PERFORMANCE and a description of what the OPERATOR can expect if the ESSENTIAL PERFORMANCE is lost or degraded due to EM DISTURBANCES (the defined term “ESSENTIAL PERFORMANCE” need not be used).
Table 1
| Guidance and manufacturer's declaration - electromagnetic emissions | |
| Emissions test Compliance | |
| RF emissionsCISPR 11 | Group 1 |
| RF emissionsCISPR 11 | Class B |
| Harmonic emissionsIEC 61000-3-2 | Not application |
| Voltage fluctuations/flicker emissionsIEC 61000-3-3 | Not application |
Table 2
| Guidance and manufacturer's declaration - electromagnetic immunity | ||
| Immunity test IEC | 60601-2test level | Compliance level |
| Electrostatic discharge (ESD)IEC 61000-4-2 | ±8 kV contact ±2 kV, ±4 kV, ±8 kV, ±15 kV air | ±8 kV contact ±2 kV, ±4 kV, ±8 kV, ±15 kV air |
| Electrical fast transient/burstIEC 61000-4-4 | power supply lines: ±2 kV input/output lines: ±1 kV | Not application |
| SurgeIEC61000-4-5 | line(s) to line(s): ±1 kV line(s) to earth: ±2 kV 100 kHz repetition frequency | Not application |
| Voltage dips, short interruptions and voltage variations on power supply input linesIEC 61000-4-11 | 0%U_T ; 0.5cycle At 0^ , 45^ , 90^ , 135^ , 180^ , 225^ , 270^ and 315^ 0%U_T ;1 cycle and 70%U_T ; 25/30cycles Single phase: at 0^ 0% U_T ; 300cycle | Not application |
| Power frequency (50Hz/60Hz)magnetic fieldIEC 61000-4-8 | 30 A/m50Hz/60Hz | 30 A/m50Hz/60Hz |
| Conduced RFIEC61000-4-6 | 150KHz to80MHz:3Vrms6Vrms (in ISM and amateur radio bands) 80%Am at 1kHz | Not application |
| Radiated RFIEC61000-4-3 | 10 V/m80 MHz – 2,7GHz80 % AM at1 kHz | 10 V/m80 MHz – 2,7GHz80 % AM at1 kHz |
| NOTE U_T is the a.c. mains voltage prior to application of the test level. | ||
Table 3
| Guidance and manufacturer's declaration - electromagnetic immunity | |||||||
| Radiated RF IEC61000-4-3 (Test specifications for ENCLOSURE PORT IMMUNITY to RF wireless communications equipment) | Test Frequency | Band (MHz) | Service Modulation | Modulation (W) | Distance (m) | MMUNITY TEST LEVEL (V/m) | |
| 385 380- | 390 TETRA | 400 | Pulse modulation b) 18Hz | 1.8 0.3 27 | |||
| 450 430- | 470 GMRS 460: | FRS 460 | FM c) ± 5kHz deviation 1kHz sine | 2 0.3 28 | |||
| 710 704- | 787 LTE Band | 13, 17 | Pulse modulati-on b) 217Hz | 0.2 0.3 9 | |||
| 745 | |||||||
| 780 | |||||||
| 810 800- | 960 | GSM 800/900, TETRA 800, iDEN 820, CDMA 850, LTE Band 5 | Pulse modulati-on b) 18Hz | 2 0.3 28 | |||
| 870 | |||||||
| 930 | |||||||
| 1720 1700 | -1990 GSM 18 | 00;CDMA 1900;GSM 1900;DECT;LTE Band1, 3, 4, 25;UMTS | Pulse modulati-on b)217Hz | 2 0.3 28 | |||
| 1845 | |||||||
| 1970 | |||||||
| 2450 2400 | -2570 Bluetooth | th,WLAN,802.11b/g/n, RFID2450, LTEBand 7 | Pulse modulati-on b)217Hz | 2 0.3 28 | |||
| 5240 5100 | -5800 WLAN | 802.11a/n | Pulse modulati-on b)217Hz | 0.2 0.3 9 | |||
| 5500 | |||||||
| 5785 | |||||||
TheBluetooth ^® word mark and logos are registered trademarks owned by Bluetooth SIG, Inc. and any use of such marks by Beurer GmbH is under license. Other trademarks and trade names are those of their respective owners. Apple and the Apple logo are trademarks of Apple Inc., registered in the U.S. and other countries. App Store is a service mark of Apple Inc., registered in the U.S. and other countries. Google Play and the Google Play logo are trademarks of Google LLC. Android is a trademark of Google LLC.

BEURER GmbH • Söflinger Str. 218 • 89077 Ulm (Germany)





