IH 21 - Inhaler BEURER - Free user manual and instructions
Find the device manual for free IH 21 BEURER in PDF.

Frequently Asked Questions - IH 21 BEURER
User questions about IH 21 BEURER
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Inhaler in PDF format for free! Find your manual IH 21 - BEURER and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. IH 21 by BEURER.
USER MANUAL IH 21 BEURER
Instructions for use 13
FR Inhalatateur
Mode d'emploi 23
ES Inhalador
- Getting to know your instrument 13
- Signs and symbols 13
- Safety information and warnings.. 14
4.Description of device and accessories.15 - Initial use. 16
- Operation 16
- Changing the filter 18
- Cleaning and disinfection 19
- Disposal 20
- Troubleshooting 20
- Technical specifications 21
- Replacement parts and wearing parts 21
13.Warranty/service 22
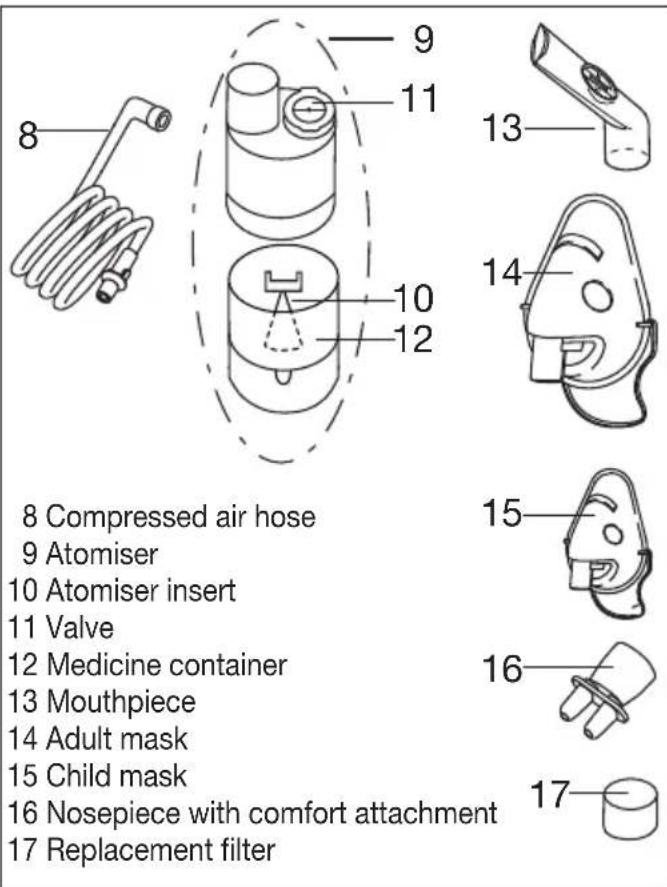
Included in delivery
For details of the delivery scope, see description of the device and accessories on page 15.
- Nebuliser
- Atomiser
- Compressed air hose
- Mouthpiece
- Adult mask
- Child mask
- Nosepiece with comfort attachment
- Spare filter
These instructions for use
1. Getting to know your instrument
Dear customer
Thank you for choosing one of our products. Our name stands for high-quality, thoroughly tested products for applications in the areas of heat, weight, blood pressure, body temperature, pulse, gentle therapy, massage and air.
Please read these instructions for use carefully and keep them for later use, be sure to make them accessible to other users and observe the information they contain.
With kind regards
Your Beurer team
Area of application
This nebuliser is a device used for the nebulisation of liquids and liquid medication (aerosols) and for the treatment of the upper and lower respiratory tract.
The nebulisation and inhalation of medication prescribed or recommended by a doctor can prevent against diseases of the respiratory tract, treat the side effects of these disorders and accelerate healing.
For more information on possible applications, consult your doctor or pharmacist.
The device is suitable for inhalation at home. Medicines should only be inhaled when instructed by a doctor. Inhalation should be performed in a calm and relaxed atmosphere. Inhale slowly and deeply to enable the medicine to reach the small bronchi deep into the lungs. Exhale normally.
Once it has been properly prepared, the device can be used again. Preparation involves replacing all the accessories, including the atomiser and air filter, and disinfecting the surface of the device using a standard disinfectant. Please note that all accessories should be replaced if the device is used by more than one person.
We recommend that you replace the atomiser and other accessories after one year.
2. Signs and symbols
The following symbols appear in these instructions.

Warning
Warning instruction indicating a risk of injury or damage to health.

Important
Safety note indicating possible damage to the unit/accessory.

Note
Note on important information.
The following symbols are used on the packaging and on the nameplate of the device and accessories.
| Type BF applied part | |
| Read the instructions for use | |
| Device in protection class 2 | |
| Manufacturer | |
| I | On |
| ○ | Off |
| SN | Serial number |
| 30 Min. ON/ 30 Min. OFF | 30 minute operation, followed by a 30 minute break before operation continues. |
| IP 21 | Protected against foreign objects equal to or greater than 12.5 mm in diameter and against vertically falling drops of water |
| CE0483 | The CE labelling certifies that the product complies with the essential requirements of Directive 93/42/EEC on medical products. |
| 20PAP | Dispose of packaging in an environmen- tally friendly manner |
3. Safety information and warnings

Warning
- Before use, ensure that there is no visible damage to the unit or accessories. When in doubt, do not use the unit and contact your dealer or the customer service address provided.
- In the event of device faults, please see Chapter "10. Troubleshooting".
- Using the unit is not a substitute for consultation with or treatment from a physician. Whenever you have any pain or illness, always contact your physician first.
- If you have health concerns of any kind, contact your general practitioner!
- When using the atomiser, always follow the basic hygiene procedures.
- Always follow your doctor's instructions regarding the type of medicine to be used and the dose, frequency, and duration of inhalation.
- Only use medicines that have been prescribed or recommended by your doctor or pharmacist.
Please note:
For treatment, only use parts indicated by your doctor according to the particular diagnosis.
- Check whether there are contraindications for use with the usual systems for aerosol therapy on the medication instruction leaflet.
- If the unit does not function correctly or if you begin to feel unwell or feel pain, stop using the unit immediately.
- During use, hold the unit away from the eyes, as some nebulised medicines may cause damage to the eyes.
- Do not operate the unit in the presence of flammable gases.
- This device is not intended to be used by children or persons with restricted physical, sensory (e.g. insensitivity to pain) or mental abilities or persons lacking in the required experience or knowledge for safe operation of the device, unless supervised or instructed by a person responsible for their safety.
- Check in the medicine package leaflet for possible contraindications for use with common aerosol therapy systems.
The device must be switched off and the plug pulled out before every cleaning and/or maintenance procedure.
- Keep children away from packaging materials (risk of suffocation).
- To avoid the risk of entanglement and strangulation, store cables and air lines out of the reach of small children.
- Do not use attachments that are not recommended by the manufacturer.
-
Connect the unit only to the mains voltage listed on the type plate.
-
Do not dip the unit into water and do not use it in wet rooms. Do not allow any liquid to penetrate the unit.
- Protect the unit from strong impacts.
- Never touch the mains cable with wet hands, this can cause an electric shock.
- Do not pull the mains plug from the socket by the cord.
- Do not pinch or bend the mains cable, pull it over sharp objects, or allow it to hang freely, and protect it against sources of heat.
- We recommend that the power cable is completely unrolled to avoid dangerous overheating.
- If the mains connection cable of this device is damaged, it must be replaced by the manufacturer, its customer service department or a simi-larly qualified person to avoid any risk.
- Opening the unit carries a risk of electric shock. Disconnection from the mains supply is only guaranteed if the mains plug has been removed from the socket.
- Do not use the unit if it has fallen or been dropped on the floor, been exposed to extreme humidity or is otherwise damaged. In case of doubt, contact our customer service department or your local dealer.
- The IH 21 nebuliser must only be used with suitable atomisers and with the appropriate accessories. The use of other atomisers and accessories can impair therapeutic efficiency and may damage the device.

Important
- Power cuts, sudden faults or other unfavourable conditions may render the unit unusable. You are therefore recommended to keep a spare device or replacement medicine (as agreed with the doctor).
- If adapters or extensions are required, these must comply with the applicable safety regulations. The electrical capacity and the maximum capacity specified on the adapter must not be exceeded.
- The unit and the mains cable must be stored away from sources of heat.
- Do not use the unit in a room in which a spray has previously been used. If this is the case, ventilate the room before commencing treatment.
- Do not allow any objects to enter or obstruct the ventilation openings.
- Do not use the device if you notice an unusual noise.
- For hygiene reasons, each user must use his own accessories.
- Always disconnect from the mains after use.
- Store the device in a location protected against environmental influences. The device must be stored in the specified ambient conditions.
FUSE
- The device contains an overload protection fuse. This must only be changed by authorised specialist personnel.
General notes

Important
- Use the unit only:
-On humans, - for its intended purpose (aerosol inhalation) and in the manner described in these instructions for use.
- Any improper use can be dangerous!
- In acute emergencies, first aid takes priority.
- Apart from the medication, use only distilled water or a saline solution in the unit. Other liquids may cause defects to the nebuliser or atomiser.
- This unit is not intended for commercial or clinical use, but only for individual, private household use!
Before using the unit for the first time

Important
- Before you use the unit for the first time, remove all packaging materials.
- Protect the unit against dust, dirt and moisture. Never cover the unit during operation.
- Do not use the unit in very dusty environments.
- Switch the unit off immediately if it is defective or malfunctioning.
- The manufacturer shall not be held liable for damage or injuries caused by improper or incorrect use.
Repairs
Note
- Never open or attempt to repair the unit yourself, as otherwise proper function is no longer guaranteed. Failure to observe this regulation shall void the warranty.
- The device is maintenance-free.
- If you need to have the unit repaired, contact customer service or an authorised dealer.
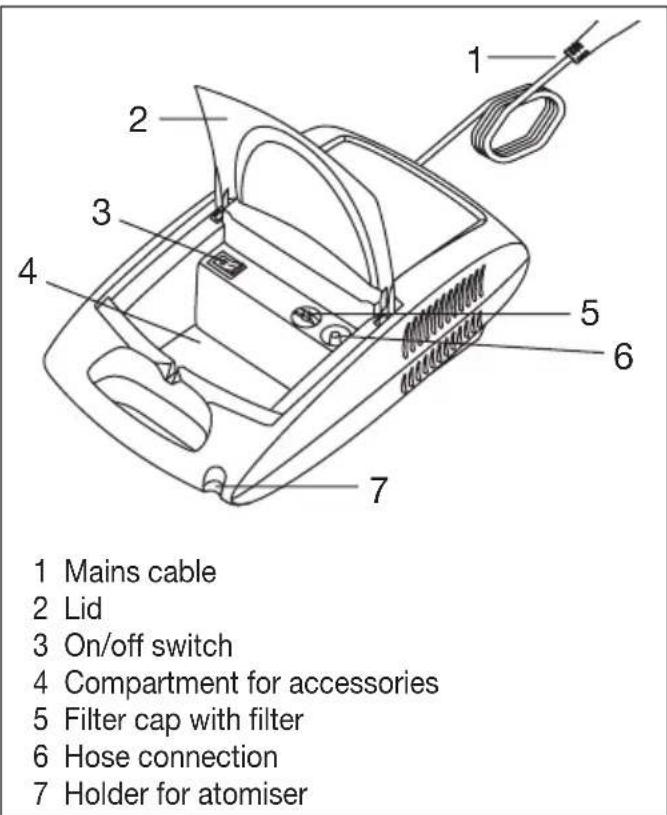
4. Description of device and accessories
Overview of the nebuliser
Overview of atomiser and accessories
5. Initial use
Setup
Remove the unit from the packaging. Place it on a flat surface.
Ensure that the ventilation slits are free.
Fold open the lid [2] to access the accessories.
Before using the unit for the first time

- The atomiser and accessories must be cleaned and disinfected before they are used for the first time. For information about this, see "Cleaning and disinfection" on page 16.
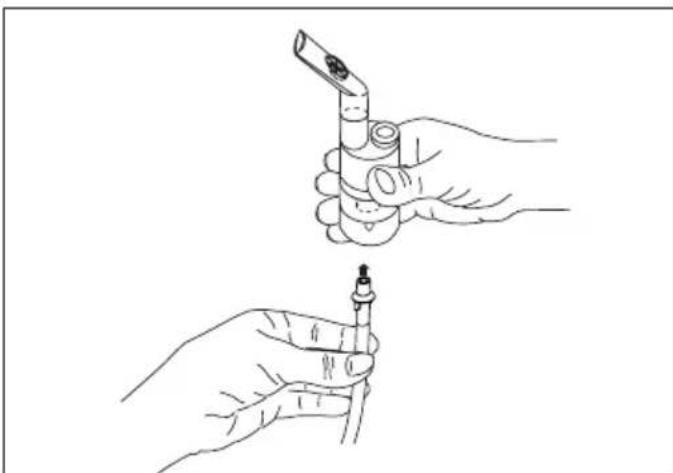
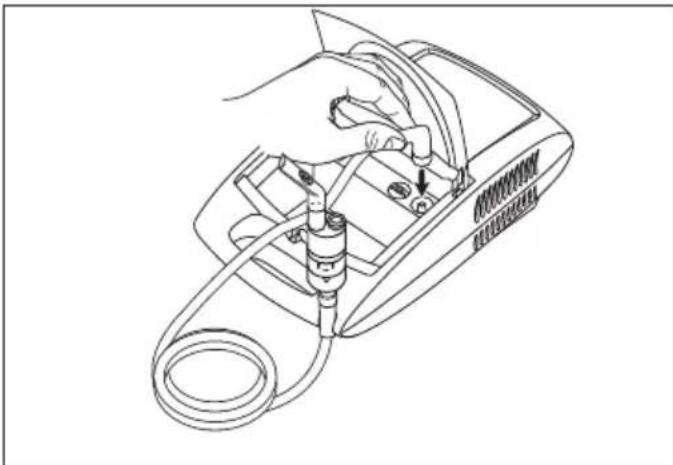
- Attach the compressed-air hose [8] at the bottom of the medicine container [12].
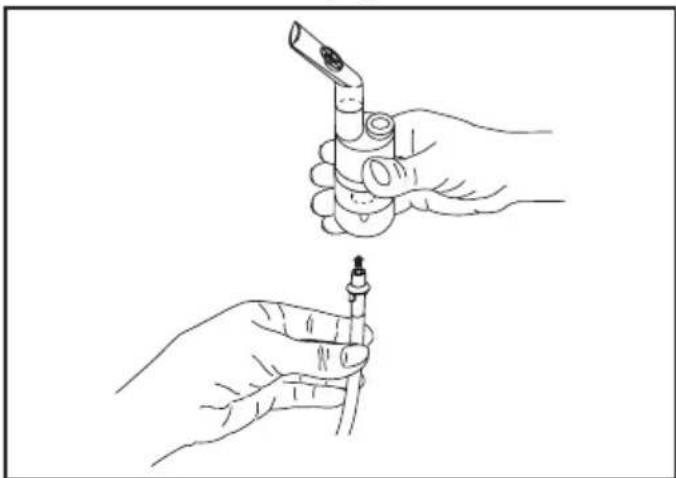
- Connect the other end of the hose [8] to the hose connector [6] on the nebuliser by lightly turning it.
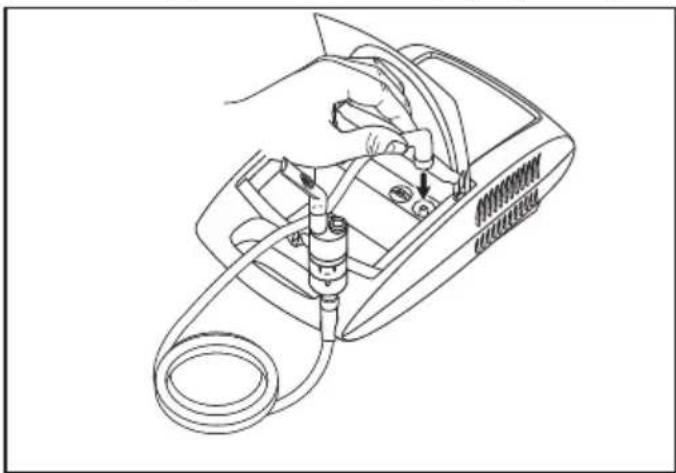
Mains connection
Connect the unit only to the mains voltage listed on the type plate.
- Insert the mains cable plug [1] into a suitable socket.
- Push the plug completely into the mains socket.

-
Ensure that there is a socket near to where the unit will stand.
-
Lay the mains cable in such a way that no one can trip over it.
- To disconnect the nebuliser from the mains after use, first switch off the device and then remove the plug from the socket.
6. Operation

Caution
- For hygiene reasons, the atomiser [9] and the accessories must be cleaned after each treatment and disinfected after the last treatment of the day.
The accessories may be used only by one person; use by several people is not recommended. - If several different medicines are to be inhaled in succession during therapeutic use, the atomiser [9] must be rinsed under warm tap water after each use. For information about this, see "Cleaning and disinfection" on page 16.
- Follow the instructions for changing the filter contained in these instructions.
- Before each use, check that the hose connections on the nebuliser [6] and the atomiser [9] are tight.
- Before use, check that the device is functioning correctly. To do this, switch on the nebuliser (including the connected atomiser, but without any medication) for a short time. If the atomiser [9] expels air, the device is functioning correctly.
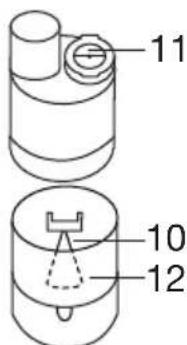
1. Inserting the nozzle attachment
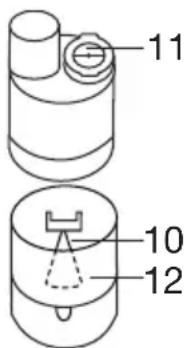
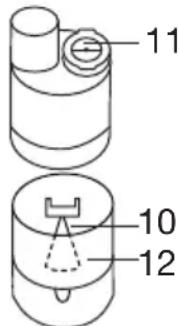
- To open the atomiser [9], twist the upper section anticlockwise against the medicine container [12]. Place the nozzle attachment [10] in the medicine container [12].
- Ensure that the cone for administering medication fits well on the cone for the air duct inside the atomiser [9].
2. Filling the atomiser
- Pour an isotonic saline solution or the medicine directly into the medicine container [12]. Do not overfill! The maximum recommended fill level is 8 ml !
- Use medication only on the advice of a physician, and check the appropriate inhalation duration and quantity for your needs.
If the required volume of medication is less than 2ml top up this quantity to at least 4ml using an isotonic saline solution. Viscous medicines may also require dilution. Always follow the instructions of your doctor.
3. Closing the atomiser
- To close the atomiser [9], twist the upper section clockwise against the medicine container [12]. Ensure the parts are connected correctly.
4. Inserting the valve
- Press the blue valve [11] into the opening intended for this purpose on the upper side of the atomiser [9].

Note
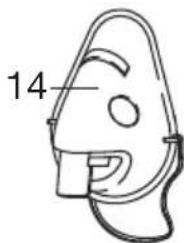
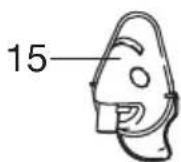
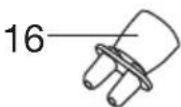
The valve system ensures better delivery of the medication to the lungs. Only use the valve [11] with the mouthpiece [13], never with the masks [14, 15] or the nosepiece [16].
5. Connecting the accessories to the atomiser
- Connect the required accessory part (mouthpiece [13], adult mask [14], child mask [15] or nosepiece [16]) to the atomiser [9].
In addition to the accompanying adult [14] and children's mask (from 2 years of age) [15], you also have the option of using baby mask (from ages 0-3 years) with your nebuliser. The baby mask is not included in delivery with the IH 21. You can find an overview of all replacement items in chapter "12. Replacement parts and wearing parts".

Note
The most effective form of nebulisation is by using the mouthpiece. Nebulisation using a mask is only recommended if it is not possible to use a mouthpiece (e.g. for children who are not yet able to inhale medication using a mouthpiece).
When using a mask to inhale, take care to ensure the mask fits well and the eyes are unobstructed.
- Before commencing treatment, pull the atomiser upwards out of its holder [7].
- Start the nebuliser using the on/off switch [3].
- A flow of spray from the atomiser indicates that the device is functioning properly.
6. Treatment
- When inhaling, sit upright and relaxed at a table and not in an armchair to avoid compressing the airways and therefore impairing the effectiveness of the treatment.
- Inhale the nebulised medicine deeply.

Important
The device is not suitable for continuous operation; after 30 minutes of operation it must be switched off for 30 minutes.

Note
During treatment, hold the atomiser straight (vertical), otherwise the atomisation does not work and correct function cannot be guaranteed.

Important
Essential oils, cough medicines, solutions designed for gargling, and drops for application to the skin or for use in steam baths are wholly unsuitable for inhalation using a nebuliser. These substances are often highly viscous and can impair the correct function of the device and hence affect the effectiveness of its application in the long term.
In the case of oversensitivity of the bronchial system, medicines containing essential oils can sometimes cause acute bronchiaspasm (a sudden, cramp-like tightening of the bronchi accompanied by breathlessness). Always ask your doctor or pharmacist for advice!
7. End of inhalation
If the spray is only output sporadically or if the noise changes during inhalation, you can end the treatment.
- Switch off the nebuliser after treatment using the on/off switch [3] and disconnect it from the mains.
After use, replace the atomiser [9] in its holder [7].
8. Nasal douche
The nasal douche is not included in delivery with the IH 21. You can order the nasal douche using the item number 601.37. You can find an overview of all replacement items on page 21.

You can use the nasal douche to clean your nasal cavities. The nasal douche creates a fine jet of liquid for treating respiratory tract problems or for moistening the nasal mucous membranes.
We recommend using the nasal douche for colds, dryness inside the nose, dust exposure, pollen allergies, chronic inflammation of the sinuses or upon the advice of your doctor.

Warning
- Do not use the nasal douche in combination with other medication or essential oils.

Important
- The nasal douche should only be used with isotonic saline solutions.
- Replace the nasal douche after a year.
The maximum volume of the nasal douche is 10ml . If you are unsure about the correct dilution of the saline solution, please consult your doctor.
9. Using the nasal douche
To use the nasal douche correctly, proceed as follows:
| 1. Twist the cover [19] 90° anticlockwise and remove it in an upward direction. 2. Lift the collection container [20] out. | |
| 3. Fill the saline container [21] with saline solution. | |
| 4. Place the collection container [20] into the saline container [21]. 5. Place the cover [19] on the saline container [21]. Twist the cover [19] in a clockwise direction until it clicks firmly into place. | |
| 6. Connect the compressed air hose [8] to the saline container [21] from below. | |
| 7. Position the cover [19] carefully inside one of your nostrils. Breathe through the nose. |
- Press the On/Off switch [2] on the back of the nebuliser. To start the application, place one finger over the cover hole [22] on the container.

Note
During the application, breathe slowly and evenly through the nose. Tilt your head slightly in the opposite direction to the nostril being treated so that the saline solution can flow deep into the nose.
9. To pause or stop the application, take your finger off the cover hole [22] on the saline container [21].
10. Once your nose feels clear again, you can stop the application. The application should be stopped at the latest once the maximum filling quantity has been used.
10. Clean the device
For information about this, see "Cleaning and disinfection" on page 19.
7. Changing the filter
In normal operating conditions, the air filter must be replaced after approx. 500 operating hours or one year. Please check the air filter regularly (after 10-12 nebulisation procedures).
If the filter is very contaminated or blocked, replace the used filter. If the filter has become wet, it must also be replaced with a new one.

Caution
- Do not try to clean and re-use the used filter!
- Only use original filters supplied by the manufacturer. Otherwise, your nebuliser may become damaged or effective therapy can no longer be guaranteed.
- Do not repair or maintain the air filter while it is in use.
- Never operate the device without a filter.
To replace the filter, proceed as follows:

Caution
- First switch off the unit and unplug it.
-
Allow the unit to cool down.
-
Remove filter cap [5] upwards.

Note
If the filter remains in the unit when the cap is removed, remove the filter from the unit using tweezers or similar.
- Replace the filter cap [5] containing the new filter.
- Check that the filter cap is firmly in place.
8. Cleaning and disinfection
Atomiser and accessories

Warning
Observe the following hygiene instructions in order to avoid any health risks.
- The atomiser [9] and accessories are intended for multiple use. Please note that cleaning requirements differ according to the different application areas.
Notes:
- The atomiser and accessories must not be mechanically cleaned with brushes or similar as this can cause irreparable damage and the success of the treatment can no longer be guaranteed.
- For additional requirements regarding the necessary hygienic preparations (hand washing, handling of medicines or inhalation solutions) in high-risk groups (e.g. cystic fibrosis patients), contact your doctor.
- Ensure that all parts are properly dried following all cleaning and disinfection. Residual moisture or water droplets can result in an increased risk of bacterial growth.
Preparation
- Directly following each treatment, all parts of the atomiser [9] and any accessories used must be cleaned of medicine residues and contamination.
- To do this, dismantle the atomiser [9] into its component parts.
- Remove the mouthpiece [13], mask [14,15] or nosepiece [16] from the atomiser.
- Disassemble the nosepiece if you have used it with the comfort attachment.
- Dismantle the atomiser by twisting the upper section anti-clockwise against the medicine container [12].
- Remove the nozzle attachment [10] from the medicine container [12].
- Remove the valve [11] by pulling it out of the atomiser [9].
- The nebuliser is later reassembled in the reverse order.
Cleaning

Important
Before you clean the unit, always switch it off, unplug it and let it cool off.
The atomiser and the used accessories such as the mouthpiece, mask, nozzle attachment, nasal douche,
etc. must be cleaned with hot but not boiling water after each use.
Carefully dry all components using a soft cloth. Once all components are fully dry, reassemble the atomiser and place the components in a dry, sealed container or carry out disinfection.
When cleaning, ensure that all residues are removed.
Never use any substances that may be poisonous when in contact with the skin or mucous membranes, or when swallowed or inhaled.
Use a soft, dry cloth and non-abrasive cleaning products to clean the device.
Do not use abrasive detergents and never immerse the unit in water.

Important
- Ensure that no water penetrates inside the unit!
- Do not put the device or accessories in a dishwasher.
- Do not touch the unit with wet hands while it is plugged in; do not allow any water to be sprayed onto the unit. The unit must be operated only when it is completely dry.
- Do not spray any liquids into the ventilation slots. Any liquid that penetrates into the unit can damage the electrical parts or other components of the nebuliser and impair the function of the device.
Condensation, care of the hose
Depending on the environmental conditions, condensation may collect in the hose. To prevent against bacterial growth and to ensure effective therapeutic use, this moisture must always be removed. To remove moisture from the hose, proceed as follows:
- Remove the hose [8] from the atomiser [9].
- Leave the hose plugged into the nebuliser [6].
- Run the nebuliser until all the moisture has been displaced by the flow of air.
- In the event of significant contamination, replace the hose.
Disinfection
Follow the steps listed below carefully in order to disinfect your atomiser and the accessories. It is advisable to disinfect the individual components after the last time they are used each day at the latest.
(For disinfection, you will simply require a small amount of clear vinegar and distilled water).
- First clean the atomiser and the accessories as described under "Cleaning".
- Place the disassembled atomiser, nasal douche, mouthpiece and the disassembled nosepiece in boiling water for 5 minutes.
- For the remaining accessories, use a vinegar solution consisting of 14 vinegar and 34 distilled water. Make
sure that the volume is sufficient to fully submerge the parts in the solution.
- Leave the components in the vinegar solution for 30 minutes.
- Rinse the parts with water and dry carefully with a soft cloth.

Important
Do not boil or autoclave the masks and air hose.
- Once all components are fully dry, reassemble the atomiser and place the components in a dry, sealed container.

Note
Ensure that all parts are thoroughly dried during, and in particular after cleaning, otherwise there is an increased risk of bacterial growth.
Drying
- Place the individual components on a dry, clean and absorbent surface and allow to dry completely (for at least 4 hours).
Material resistance
- As with all plastic components, frequent use and cleaning of the atomiser and the accessories can lead to a certain amount of wear. Over time, this can alter the aerosol properties and may eventually affect the efficiency of therapy. We therefore recommend that you replace the atomiser once a year.
- When choosing a cleaning or disinfecting agent, note the following: Only use mild cleaning or disinfectant agents in the quantities recommended by the manufacturer.
Storage
- Do not store the nebuliser in a damp atmosphere (e.g. in the bathroom) or transport it together with damp objects.
- Store and transport the nebuliser away from direct sunlight.
- The accessories can be safely stored in the accessories compartment [4]. Store the device in a dry place, if possible in its original packaging.
9. Disposal
In the interest of environmental protection, do not dispose of the device in household waste.
Please dispose of the device in accordance with
EC Directive - WEEE (Waste Electrical and Elec
tronic Equipment).
If you have any queries, please contact the appropriate local authorities.

10. Troubleshooting
| Problem/Question | Possible Cause/Remedy |
| The atomiser produces little or no aerosol. | 1. Too much or too little medicine in the atomiser. Minimum: 2 ml. Maximum: 8 ml. |
| 2. Check that the nozzles are not blocked If necessary, clean the nozzle (e.g. by rinsing). Use the atomiser again. CAUTION: Only poke implements through the fine holes from the underside of the nozzle. | |
| 3. The atomiser is not held in an upright position. | |
| 4. The medicine solution is unsuitable for nebulising (e.g. too thick). The medicine solution should be specified by the doctor. | |
| Output is too low. | Hose kinked, filter blocked, too much inhalation solution. |
| Which medicines are suitable for inhalation? | Consult your doctor. Essentially all medicines that are suitable and approved for device-based inhalation can be inhaled. |
| Some inhalation solution remains in the atomiser. | This is normal and occurs for technical reasons. Stop inhalation if you hear a notable difference in the noise of the atomiser. |
| What special steps should be taken for babies and children? | 1. In babies, the mask should cover the mouth and nose to guarantee effective inhalation. 2. In children, the mask should also cover the mouth and nose. Nebulisation next to a sleeping person is not suitable because insufficient medicine reaches the lungs. Note: Children should only use the device with help and under supervision of an adult. Never leave a child alone with the nebuliser. |
| Inhalation with the mask takes longer. | This is for technical reasons. Less medicine is inhaled per breath through the mask holes than using the mouthpiece. The aerosol is mixed with ambient air through the holes in the mask. |
| Why do I need to replace the atomiser at regular intervals? | There are two reasons for this:1. To guarantee a therapeutically effective particle spectrum, the nozzle holes should not exceed a certain diameter.Due to the thermal and mechanical stresses, the plastic is subjected to a certain amount of wear. The nozzle attachment [10] is particularly susceptible to this wear. This can also cause changes to the droplet composition of the aerosol, which has a direct effect on the effectiveness of the treatment.2. You are also recommended to replace the atomiser on a regular basis for hygiene reasons. |
| Does each person need their own atomiser? | This is absolutely necessary for hygiene reasons. |
11. Technical specifications
| Model IH 21 | |
| Type IH 21/1 | |
| Dimensions (WxHxD) 300 x 180 x 100 mm | |
| Weight 1.65 kg | |
| Operating pressure approx. 0.8 - 1.45 bar | |
| Atomiser fill | max. 8 ml |
| volume | min. 2 ml |
| Medicine flow | approx. 0.3 ml/min |
| Sound pressure | approx. 52 dBA (acc. to DIN EN 13544-1 section 26) |
| Mains connection | 230 V~; 50 Hz; 230 VA UK: 240 V~; 50 Hz; 240 VA Saudi Arabia: 220 V~; 60 Hz; 220 VA |
| Expected service life | 1000 h |
| Operating conditions | Temperature: +10 °C to +40 °C Relative humidity: 10% to 95% Atmospheric pressure: 700 to 1060 hPa |
| Storage and transport conditions | Temperature: 0°C to +60°C Relative humidity: 10% to 95% Atmospheric pressure: 500 to 1060 hPa |
| Aerosol | 1) Flow: 5.3 l/min |
| Properties | 2) Aerosol delivery: 0.326 ml |
| 3) Aerosol delivery rate: 0.132 ml/min | |
| 4) Particle size (MMAD): 3.07 μm |
The serial number is located on the device or in the battery compartment.
Subject to technical modifications.
Particle size diagram
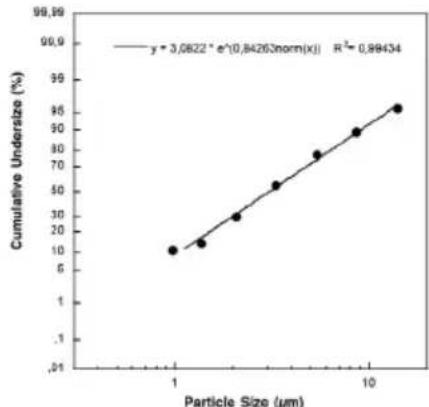
Measurements were performed using a sodium fluoride solution with a "Next Generation Impactor" (NGI). This diagram may not be applicable for suspensions or highly viscous medicines. More information can be obtained from the relevant medicine manufacturer.
12. Replacement parts and wearing parts
| Designation Material REF | ||
| Yearpack (contains Mouth-piece, Nosepiece, Adult mask, Child mask, Atomiser, Com-pressed air hose, Filter) | PP/PVC | 601.22 |
| Nasal douche | PP | 601.37 |
| Baby mask | PVC | 601.31 |

Note
If the unit is used outside of the specifications, proper function is no longer guaranteed!
We reserve the right to make technical changes to improve and further develop the product.
This device and its accessories comply with European standards EN60601-1 and EN60601-1-2 (CISPR 11, IEC61000-3-2, IEC61000-3-3, IEC61000-4-2, IEC61000-4-3, IEC61000-4-4, EC61000-4-5, IEC61000-4-6, IEC61000-4-7, IEC61000-4-8, IEC61000-4-11), as well as EN13544-1, and is subject to special safety measures in terms of electromagnetic tolerance. The unit conforms to the requirements of the European Directive for Medical Products 93/42/EEC, the MPG (German Medical units Act).
NOTES ON ELECTROMAGNETIC COMPATIBILITY
- The device is suitable for use in all environments listed in these instructions for use, including domestic environments.
- The use of the device may be limited in the presence of electromagnetic disturbances. This could result in issues such as error messages or the failure of the display/device.
- Avoid using this device directly next to other devices or stacked on top of other devices, as this could lead to faulty operation. If, however, it is necessary to use the device in the manner stated, this device as well as the other devices must be monitored to ensure they are working properly.
- The use of accessories other than those specified or provided by the manufacturer of this device can lead to an increase in electromagnetic emissions or a decrease in the device's electromagnetic immunity; this can result in faulty operation.
13.Warranty/service
Beurer GmbH, Söflinger Straße 218, 89077 Ulm, Germany (hereinafter referred to as "Beurer") provides a warranty for this product, subject to the requirements below and to the extent described as follows.
The warranty conditions below shall not affect the seller's statutory warranty obligations which ensue from the sales agreement with the buyer.
The warranty shall apply without prejudice to any mandatory statutory provisions on liability.
Beurer guarantees the perfect functionality and completeness of this product.
The worldwide warranty period is 5 years, commencing from the purchase of the new, unused product from the seller.
The warranty only applies to products purchased by the buyer as a consumer and used exclusively for personal purposes in the context of domestic use.
German law shall apply.
During the warranty period, should this product prove to be incomplete or defective in functionality in accordance with the following provisions, Beurer shall carry out a repair or a replacement delivery free of charge, in accordance with these warranty conditions.
If the buyer wishes to make a warranty claim, they should approach their local retailer in the first instance: see the attached "International Service" list of service addresses.
The buyer will then receive further information about the processing of the warranty claim, e.g. where they can send the product and what documentation is required.
A warranty claim shall only be considered if the buyer can provide Beurer, or an authorised Beurer partner, with
- a copy of the invoice/purchase receipt, and
- the original product.
The following are explicitly excluded from this warranty:
- deterioration due to normal use or consumption of the product;
- accessories supplied with this product which are worn out or used up through proper use (e.g. batteries, rechargeable batteries, cuffs, seals, electrodes, light sources, attachments and nebuliser accessories);
products that are used, cleaned, stored or maintained improperly and/or contrary to the provisions of the instructions for use, as well as products that have been opened, repaired or modified by the buyer or by a service centre not authorised by Beurer; - damage that arises during transport between manufacturer and customer, or between service centre and customer;
- products purchased as seconds or as used goods;
- consequential damage arising from a fault in this product (however, in this case, claims may exist arising from product liability or other compulsory statutory liability provisions).
Repairs or an exchange in full do not extend the warranty period under any circumstances.
FRANÇAIS
Sommaire
Cher cliente, cher client,
IJIyJeHnI NcNoB3OBaTb ToJbKO Te DeTaII, PpIMeHHeNKeKOTOpbIX 6bIIO peKOMeHDoBaHO BpaOMB COOTBETCTBn C COCTOHNm 3IOPOBBy.
BHNMaTeNbHO npouHaiTe Ha npuIarauOeMcK JekapCTBeHHOMy npenapaty ynaKOBOUHOM JnCTKe INΦOpMaUIO BO3MOXHbIX npOTnBOJOKa3AHJN K npIMeHeHIO C O6bIHbIMn CnCTeMaMn a3pO3OJIbHOJ TepaNN.
- Ecnn np6op pa6oTaet HeKOppeKTHO nn y Bac nOBnncb HeDOMOraHne nn 60n, cpa3y Je npekpataTe nCNoB3OBAHne np6opa.
Bo BpemnnoJb3OBAHn depKnte np6Op Ha doCTaTOHOM pacCToHn OT rna3, paCnblneHHbI nekapCTBeHHbI npenapaT MOKeT npNBecTu Knx NOBpeXDeHnM.
He nCnoJb3yIte npn60B nPomeueHnX c rOpOuHMn ra3amN npn BbICOKo KOnUeHTpaun NcCLopoJa.
JaHHbI np6op He npedHa3HueH dIy IcNoIb3OBA-HnA DeTbMn IIN JIODbMn C OrpaHnueHHbIMn fN3n-yeCKm, CEHCOPHbIMn (HaNPmep, C OTCyTCTBnEM 60JeBOu YyBCTBNTeJIbHOCTn) IIN yMCTBeHHbIMn CNOco6HOCTaMn, C HeIOCTaTOUHbIMn 3HaHnAIMn IIN ONbITOM. NcKIOUeHne COCTaBnIOT cnyan, KOrda 3a HMMN OcyuCETBnE TcR HaJnxKaun HaD3Op IIN ecIn OH nOnyuJIn OT Bac INHCTpyKUn No IcNoIb-3OBAHnIO np6opa.
-Пюоверп снстукцюВуразковеMeДИКаMuHTa HaPredMet BO3MOxHbIX ПOTиBOIOKa3aHINПрИ NcNoJIb3OBAHnCpacnpocTpapHeHHbIMNCCTeMaMn a3po30JbHoi TepaHn. - Ipeed BbIOnHeHnem JIO6bIX pa60 T NO OChTKe IIN TeXHueCKOMy 06cnyKuBaHNIO Heo6xOJMo BbIKIOHTb np6Op BblHyTb wTeKeP n3 po3eTKn.
He daBaIte ynaKOBOHbIM MaTePnAJI DeTAM (onacHOCTb ydyuSeHHa). - Ka6eIb n BO3dyxOBOdbl CJIeDyET XpaHnTb BHe DOcraemocTn DeTeMJaUwero BO3pacta BO N36exKaHne 3anyTbIBaHnN ydyuWeHn.
He nCnoJb3yIte DOnOpJHnTeJbHbIe DeTaII, He peKoMeHDoBaHHbIe IpnO3BOJnteJIem. - Ppi6op pa3peSeHo NODKJIIOuTaB TOJbKO K CeTeBOMy HaprrXeHnO, yKa3aHHOMy Ha 3aBOdCKoT a6JIuKe.
He norpykaite np6op B Body n He nCnoJb3yIte ero Bo BlaxKbIX nomeueHnx. He donyckaite no- naaHn JnkOcTn BHytbp np6opa. - Bepernte npnbop ot cnIbHbix yapob.
- He npikacaiTecb K Ka6JIIO MOKpbIM pyKaAMn - onachocTB nopaxHnna 3neKTpueeCKm TOKOM.
- He BBITACKNBAIte UTEKEp n3 po3eTKn, DEpkacb 3a ceTeBOI Ka6eIb.
He 3axmaTe n nepern6aTe KaebIb, He daBaTe emy cOpnKacatbC aOCTpbIM KpaMn IIN CBNcaTb, 3aUuauTe eTO OT BO3dEInCTBnB BBICOKo TMnepaTypbl. - PeKOMeHdyETcN NOHOCbIpa3MaTbIBaTb NITaOu 1 KaeBb IJI npEDoTbPaUeHn nepepeBa.
-
Пювов се teBOrO nHTaHnЯ пибopa He nOДнeжИЗамеHe. Пи NOВpeXdEHH nPoBoDA nPиБOP cIeDyET yTININ3uPoBaTb.
-
PnB BCKpbITnn PpN60pa BO3HnKaet ONaCHOCTb NopaxKeHn 3JKeKtpnueckm TokOM. Pnp6Op rapaHTnpoBaHHO OTcoeHNHe OT CeTN 3JKeKtpoCHa6KeHn TOIbKO B TOM Cnyae, ecNI UTeKeP BbIHyT n3 PO3eTKN.
- EcIn np6op yan, noDBeprCnBbHomy Bo3deiCTBnIO BnaI INI NOnyUIN INbIe NOBpeJDeHnI, erO daJIbHeIWeE nCNoIb3OBaHne 3aNpeuaeTc. Pn HAnuIN COMHeHNI ObaTnTeCb B cepBnCHyo CnyX6By INI K TOPROBOMy PpeCTaBnTeHIO.
- INHraJTop IH 21 pa3pe7aetc nCNoIb3ObaTb TObko Bmecte C noDxOJaUmm pacNbIInTeJMa Beurer n C COOTBETCTByIOUmm npHaIJeXHoCTAm. PnIMeHHe N pINHaIeXHOCTe CTOpOHnX IPOUN3BOdIteJe MoKeT npIBeCTN K CHNXeHnO 3ΦΦeKTUBHOCTn IueHnN n DaKe K NOBpeKDeHnO npIbopa.

BHHMaHne
C60B NOdaue 3eKtpo3Hepn, HeoxuHaHO B03- HNKwne HncnpaBHOCTN IN DpyrIe He6laRopnTt HbIe ycIOBm MOyT npBecTn K BbIXOy np6Opa n3 cTpo. IooTOMy peKomeHyetc IMeTb 3anachOH np6Op nn lekapCTBO (corlaocBaHHoe C Bpaqom).
- Pn Heo6xOIMOCt NcNoJb3OBaHn aAnTepeB uN yIINHTeJe OHI DOJIXHbI OTBeuATb DeIcTByIOUm npEiNCaHnM TexHnKn 6e3OpacHOCTn. HJIb3r npeBbIaTb npedeJIbHyIO MOHOCt b TOkA mAKCImaJIb-HyIO MOUHOCt, yKa3aHHyIO Ha aAnTepe.
- Pnp6op nntaioi Ka6eIb HeIb3x XpaHnTb B6n3n NCTOCHNkOB Tepla.
He noJIb3yIteCb npI6OpOM B nOmeUeHnX, B KOtOpbIX nepeD 3TmPacNbIJIaNcB aApO3oN. TaKe IOnMeUeHnHEo6xOdMn PpOBETpNBaTb nepeD npoBeDeHnEM TepaNN.
He onyckaIte nonaHaHna KaKx-Jn6o npedMeTOB B OXlaKaIoune OTBepCTn.
He nCnoJb3yIe npu6op, ecn OH n3daet Heo6bIHyBli IyM.
Buejxco6nOeHncaHHTapHO-rnHneHnuecknx Tpe6oBaHn KaKdbI NOb3OBaTeJIb DOJKeH IMEt b CBOINHmIBuDyAalbHbIe npHaNdxKHOCTn.
- После кадую Исторьаня Великийсяп рибор и розел.
- Доступа ведую.
- Роступа ведую.
- Ростotypical.
XpaHInTe npn6op B MeCTax, 3aUuIeHHbIX OT Bnna Hnra aTMocOpehBix BO3DeIcTBn. Heo6xOIMo oBeCneHTb COOTBeTCTByIOUne ycIOBnO kOpJkaIOSe n Cpebl Iy XpaHeHn npn6opa.
PPEOXPAHNTIEL
B npn6ope yctaHOBlen MaKcImMaJIbHbI npedoxpaHnteIb.Ero 3aMeHa DoJXHa npOn3BOuNTbc TOLbKO KBaJIuΦnUpOBAHHbIM NepCOHaIOM CO CneuaJIbHbIM DOnyCKOM.
06üne yka3aHna

BHHMaHne
- IcnoIb3yIte npI6Op nckJIIOHTeIbHO B cIeNyIOxuX ueJax:
-ДЯJIIODEI;
- B ueJx,ДЯ KOTopbIX OH 6bl pa3pa6oTaH (a3po30JIbHa INrAJaIyI),B COOTBeTCTBm C yKa3aHnIyMn,ПиВeDEHHbIMN B DaHHOINHCTpyKcIIN NO npImMeHeHIO.
- Любoe птуменине He no Ha3HaueHnIO MoKet 6bIb OnaChbIM!
B KpnTnuecknx cnTyauunx npexKe Bcero CneNyet OKa3aTb nepByIO NOMOuN NOcTaJaBAWeMy.
- IommoJekapCTBeHHbIX npenapaTOB pa3peWaeTcra NCIOJIb3OBaTb TOIbKO DnCTNJIIMPOBaHHyO BODY ININ Φn3NOJOrnueckn pactBOp. PpIMeHHe NpyrX KNDKOCTei npn ONpeDeJIeHHbIX ycNoBnIX MoKeT npNBecTI K NOBpeJdeHIu INHaJIrTopa INI paCnbIJIITeJI.
- Пибор He пedingанануен Ддя Испьзоваян Ha npoIN3BOIDCTBe nIn B KInHnuecknx yCIOBnIx. Ero npimHeHne pa3peSeHo INCKIIOHTeJIbHO B DomaHnIX yCIOBnIx!
IpeB BBODOM B 3KcnIyatauio

BhimaHne
- Pered nCnoB3OBAHnem np6opa cIeJyET ydaNTb BCE yNaKOBOuHbIe MaTePnaJIbI.
He donyckaTe 3aarp3HEnn np6opa, 3aunuaTe ero ot nbnn n BnaRn. Bo Bpem nCnoJb3ObaHn Hn B KOEM cIyae He HaKpbBaTe np6op.
He nCnoJIb3yIte npI6Op B cnJIbHO 3aIbJIeHHbIX NOMeUeHnX.
Bcnyae HeicnpaBHOCTe nn HenolaOK B pa6oTe HemeJeHHO OTKIOUHTe np6Op. - I3ROTOBtIeIb He HecET OTBETCTBEHHOCTn 3a yUeep6, Bbl3BaHHbIi HeKBAJIuΦuIupoBaHHbIM NIIi HeHaJNeJkaIiM NcNoIb3ObaHnem npIbopa.
Pemont npn6opa

Yka3aHne
- Hn B Koem clyuae He OTKpbIbAaTe npnbOp n He peMoHTpyuTE erO cAmocToTcIbHo, TaK KaK B DaHHOM clyuae HaedxHocTb erO pa60TbI 6oJIbUe He rapaHTpyetc. HecobJIOdHeNe 3tOrO Tpe6OBaHnB BeTe K Notepe rapaHTn.
- Пибор He Tpe6yeT TexHnueckOrO obcIyXnBaHnY.
- Ecni npnbop HxkdaetcB peMOHTe, o6paTntecb B cepBnCHyO cnjxK6y uN K aBTOpn3OBaHHOMy ToproBOMy npedCTaBNTeJIIO.
0630p KOMnoHcHTOB paCnblnteIy I npHaJdJIexKHOCTeI
- Tpy6ka-ydlnHnTeIb
- Pacnbintenb
- Hacaika pacnbilntela
- Klaanah
- Emkoctb dny lekapctBeHHbIX cpeIcTB
- MyHdUyK
- Macka Дяг Взрсьх
- Macka dIa ITei
- HacaKa Ira HOca CO cbeMHbIM 3JeMeHTOM Ira NOBbiIeHnKOMΦOpTa
- Cmehhbi nIbTp
5. Побrotовka к pa6ote
YctaHOBka
I3BneKeIte np6Op n3 ynaKOBKn.
YcTaHOBnTe np6Op Ha poBHyIO NobepxHocTb.
CneIte 3a TeM, TTo6bI npope3n IJRA BeHTNJraCIN OCTaBaJIncB OTKpbITbIMN.
YTo6bI DocTaB npHaJnEJXHOCTN,OTKINHbTe KpbILKy [2].
PepnepBbIM npmHeHneM
iYka3aHne
- Ipeed nepBbIM npimeHeHem CneJeT BbINOHNtB ouNCTky u De3nHΦeKuIO paCnblNTeJI N npHaJI JexHoCTe. CM. pa3dEJI «OuNCTKa n De3nHΦeKUIA» Ha cTp.72.
- BCTaBbTe Tpy6ky-ydnnHnTeIb [8] B eMKoCTb dIJI nekapcTba [12].
Cnerka nobepnyB erO, BCTaBbTe pyroK KOHeu Tpy6- Kn [8] B wTucep [6] nHraJrTopa.
IopKnIOueHne K cetn
IopKJIouaTe np6Op TOnbKO K CeTeBOMy HnprJKeHHIO, yka3aHHOMy Ha 3aBODcKo Ta6NIuKe.
BCTaBBTe WTEkep ceteBoro Ka6eIa [1] B noDxOJa- yuO po3eTKy.
- Дя подклioченя К сети StTekeр Должeh 6ытб
- РДнHOCTьО BCTaBЛЕнВ рОЗТКY.
iYka3aHne
- Y6eIntecb, yTO pAOM C MeCTOM yCTaHOBKn npi-6opa haxoNTcra po3etka.
Pa3mecntte Ka6eJb TaKIM o6pa3OM, Yo6bI O Hero HIKTO He MOr CNOTKHyTbcra.
-ДяOTKJIIOUeHnIHHraJIaTopaOTcTeNnocJeIHraJIaLzIu ChauJaBAbIKJIIOUHTe npI6Op,3aTeM N3BJIeKNTe ITeKeP U3 pO3ETKn.
6. 06cIyXnBaHne
BHHMaHne
Buejxco6IoudeHncaHTapHO-rnneHuecknx Tpe6oBaHn paCbInTeJb [9] n npHaJnxKHOCTn CneJyET OuNtB NocJe KaXdOrO uCNOJB3oBaHnA a B KOHcE Hr BbINOnHrTb De3HfKeLIO.
-ПинадлжноМогуТИСпОьБ3OBaTбСТОЛБКО OДнIM ЛцOM.ИспОьБ3OBaHNE HeCKОЛБКIMNЛI-цAMN He peKOMeHdyeTcR.
- EcIII IJIeHHeO6XoIMNo NocJeIOBaTeIbHO IIPOBOITb INrAJIaIIO HeCKOJIbKIMN BnJaMn JIeKapCTBeHHbIX IpeNaPatoB, To paCnblIITeJIb [9] HJxHOb IIPOMbIBaTb TEnIOBODOnpOBODHO BODOI NOcJIe KaJDoI OpiMeHHeH.N. CM. pa3dJIe «OuNCtKa n De3INHΦeKUry» Ha cTp. 72.
BHHMaTeIbHo cIeMyIte yKa3aHnM nO 3aMeHe hnIbTpA, coepXkaUIMcB daHHo INcTpyKuIN!
-Перед кадын Испьзовим пибора пюberяп почесь-coeINHeHЯ ТубкИ с ИнгалтopoM [6] npacblintelem [9].
- Ipeed nCNoB3OBAHnem npOBepbTe npaBnIbHoCTb pa60tbI np6opa.ДЯ 3TOro HeHaDoJIro BKJIOUHTe INrAIArTOp (BMecTe C NOdKJIIOUeHHbIM paCnblINTEJEM, Ho 6e3 JekapcTBeHHORo npenapaTa).EcIn npu 3Tom n3 paCnblINTEJRA [9] BbIXoDIT BO3dyx, To npu6Op pa6Otaet.
1. YctaHOBka paacnbJIInTeJbHoi HacaIkn
- OtkpoIte paCnblnteIb [9], ydepKuBaE mKOCTb dIJI nekapctBa [12] nOBepHyB BepXHIOU qactb Kopnyca npOTIB YacOBoi CTpeIKN. BcTaBBTe paCnblnteJIbHyo HacaIky [10] B EMKOCTb dJIy IekapCTBa [12].
- Y6eIITecb, yTO KOHyc dIy IODaHy IeKapCTBa XOpOio 3akpeIJIeH Ha KOHyCe dIy IIOBbOa BO3dyXa BHyTpN paCnblInTeJI [9].
2. HanolheHne paCnblnteJia
3aIeTe n3oToHnueckn fni3noIIOrueckn paCTBOp nn IekapCTBeHHbI npenapaT HenocpeICTBeHHo B EMKocTb Ira IekapCTBa [12]. He donyckaTe nepenolHeHna EMKOCTn! MakcmaIbHbI peKOMeHdyEmbI O6bem 3aONHeHncoCTabJIeT 8 m!
- IcnoIb3yIte JekapCTBeHHbI npenapaT TOnbKO nO Ha3HaueHIO Bpaa, nonTepeCyITecb y Hero, KaKaJ npoJOnKuTeJIbHOCTb INrAraUmN KAcoe KOJIInueCTBO MeDnKamEHTa dJa INrAraUm N ABnIOTcR Han6OJe NOxOJaUMM dJa Bac.
- Ecni Ha3HaueHHoe KOJIueCTBO JekapCTBeHHoro npenapata MeHbIe 2 Ml,do6aBbTe N3OTOnHueCKn fN3NOJOrnueCKn pactBOp,doBeIg O6bEm KNDKoCTn Do MNHMym 4 Ml. Heo6xOIMo TaKxe pa3-6abTb ryctIe JekapCTBeHHble npenapaTb.Ipn 3TOM TAKXe CNeDyET Co6HIOaTb yka3AHn Bpaua.
3. 3aKpbTne pacnbIInTeJra
3aKpOte paCnblnteIb [9], ydepxNBa eMKoCTb dIJIeKapcTbA [12] nIOBepHyB BepxHIOU qAcTb KOpnyCa no YacOBoi CTpeJIke. CJeIte 3a npabINbHOCTbIO COeINHeHInr!
4. YCTAHOBNTb KJanaH
- BctaBbTe cHnH KlaHn [11] B npEHa3HaueHHe OIy Hero OTBepCTne B BepxHe yactn paCnblnteIe [9].
iYka3aHne
CnCTema KJanaHOB oBecneuBaet yIyUweHHyO npoxoIMocTb MeiKamEHTa BJeRKe. KJanaH [11] npimHeTb TOnbKO C MyHdTuKOM [13], n HIKOrda c MaCKamN [14,15] nIIN HacaKDnI HoCa [16].
5.ПодсоeнHeHne npHaJNeXHoCTeK paCnblnteJIHO
CoeHInHe pacbIInTeIb [9] c Heo6xOIMbIMn npHaJIeXHOCTaMn (MyHdUrk [13], macka Ira B3pocIbIX [14], macka Ira DeTei [15] nIIu HacaKa dIra Hoca [16]).
Hapnyc npunaeraemom macko nB3pocnbix [14] macko dna deten (c 2 let) [15] Bby moxete nCnoIb-3OBaTb c nHraJrTOPOM macky dna MnaJeHceB (OT O do 3 let).Macka dna MnaJeHceB He BXOHT B KOMnIeKT noCTabKn IH 21.Ob3op BCEx npnaJnxHoCTe, npno6peTaembix DOONHITeJIbHO, COepXNTcB rIabe 12, «3anachbie yactn i 6bICTpon3HaunBaemble DeTaN»
iYka3aHne
HrAunuyeep3 MyHdTuK RaBnEeTc HAn6Oone 3ΦΦeKTHBHOfopMOI TepaNN. HrAunuyeep3 MACKy peKOMeHNyETc TObKO B TOM Clyue, ecN NTOlb3OBAHne MYHdTuKa HeBO3MOxHO (HaPmEp, DnA DeTei, KOtOpBie eUe He MOryT BdixaTb Yeep3 MyHdTuK).
Pn INrAun Yepe3 Macky CneIte 3a Tem, YTO6bl Macka XopoO Io PrnIeRana I He 3aKpbBaIra rla3a.
-
Pered npoceDpyoN n3BnKeNte paCnblnteNb N3 KpeIeHnra [7], NotaHyB BBePx.
-
BkIIOUHTe INHraJrTOp npn NOMOuN BBKJIHOaTeJr [3].
- Повленье aэрозоля ИЗ пасьителя OзначаET, до Oh pa6oTaET KорpeKTHO.
6.Лechенe
Bo Bpemn Hraan pekomhnyetc cndeTb npmo n paccna6JeHNO 3a cToJOM, a He B KpeCne InpedOTBpaueHn CdaBnBaHn DbIXaTeNbHbIX NyTeN CHNXeHn 3oFKeKTHBHOCTN neHn.
- Iny60ko BdIxaIte JekapCTBeHHbI npenapaT.

Bhimahne
Pnp6op He npedHa3HaeH dna HnpepbBHO rnpMeHenH, nocne 30 mHyT paobToI erO cneDyET otKlouatb Ha 30 mHyT.

Yka3aHne
Bo Bpem npoueybpI depKInTe pacbIINTEIb nprmo (BepTKaJIbHO), TaK KaK B IpOTNBOM Clyuae He 6ydet npOn3BOIDTbcra pacbIJIeHne H He rapaHTnpyeTc 6e3- ynpueHoe FyHKIOHIpOBaHne npi6opa.

BhimaHne
3nHbIe Macna JekapCTBeHHbIX paCTeHIn, MUKCTyPbI OT Kaun, pactBopbl IINIOCKAHNIG ROPla, KanJIN DnIpaTnpAHHN INAPOBbIX 6aHb B pINHUNIE He NODXoYr Tn INHARLcN C NOMUbIO INHARLTOpOB.3TN Do6ABKn YACTO 6bIBaOT B8KIMN INMOT OPNUATEBHO cKa3aTbCn HA pa6ote np6opa, a TAKKe Ha 3ΦΦEKTNBHOCTn erO npimeHn. Pn IOBbIweHNOyBCTBNTeJIbHOCTn 6pOHxMaJIbHOIN CnCTEmbl MeINKA-MeHTbl C 3ΦnPBbIMN MACJamM MOY TBb3BaTb OCTpbII 6pOHXocNa3M (pe3Koe n cydopoxHoe cyxHeHne 6pOHXOB C ydyubem). IpOKOHcyJIbTnpuTEcB 6tOM CJeauWM Brayom INI φapMaueBTOM!
7. 3abepweHne nHraJauznn
Ecn paCnblraMoe BeIeCTBO BbIXoNT HepaBHomepHo IIN N3MeHReTc 3ByK npu INrAJIaUN, IpOceDpyMOxHO 3aBepWntb.
- Tocne 3aBepueHn npoceDpybI OTKIOHTe INHraJTop C NOMOuBO BbIKIOUaTeJIa [3] n OTCoeHNHTe eROOT CeTI.
- CHOBA BCTaBbTe paCnblnteIb [9] B KpenJIeHne [7].
8.Hocobon dyu
Hocoboi dyu He BXoHT B KOMnJIeKT NOCTaBKn IH 21. Bbl mokeTe 3aKa3aTb Hocoboi dyu, yKa3aB apTuKyn 601.37. O63Op BCex npedMeTOB, pnpO6peTaembIX Do- noHNtIeBHO, haoDITcHa cTpaHnue 76.
He nCnoB3yIe eKne yNCTaIe cpeCTBa Hn B Koem clyuae He norpykaIte np6Op B Body.

BHHMaHHe
CneiTe 3a TEM, YTo6bI BHyTpB npn6opa He nonana BOda!
He MoTe np6Op n npHaJnEeKHOCTN B NOcyDOMOeHOn MaunHe!
HeIb3 npIkaTbcra K NOdkHIOyeHHOMy K cTeN npIb6opy BlaXHbIMn pyKam; Ha Hero TaKKe He DOJXHa nonaTaB BOa. Pnp6op pa3peWaeTc nCNoIb3OBaTb TOnbKO a6COJIOTHO cyXM.
He paCnblIaIte XnIDKOCTb B BeHTnlaIauHOHbIe OTBepCTnI! IonaDaHne XnIDKoCTe MoXeT npNBecTN K NOBpeXdEHNIO 3NeK- TpUYeCKNX N DpyrNx DeTaJIeN INHraJIaTOPa, a TaKKe K C6OaM B erOp pa6ote.
KoHdEHCaT, yXoD 3a Tpy6ko
B 3aBnCmOCTOn OT yCNoBn Okpy
XaIOUeI CpeBb I Tpy6Ke MOxET OcaKDaTbcra KOHDeHCat. YTO6bI InpenrTCTBOBaTb POCTy MUNKPOOpraHN3MOBn rapaHTnPoBaTb 63ynpeuHoe JueHHe, Bnary Heo6xOJIMO ydaJIaTb. IInr 3tOrO Heo6xOJIMO cdeJaTb cJeDyOJee:
- N3BneKeTe Tpy6ky [8] n3 pacnbijnteIer [9].
Co ctopohbI INHraJIaTIopa [6] Tpy6ky He OTCoeDnHnTe. - Bклочи Te nHraJIaTOp n octaBbTe erO pa6oTaTb do tex nop, noka TOK BO3dUxHa He ydaIIT BCIO Bnary.
- Ppi CnIbHOM 3aIrpA3HeHm 3aMeHInTe Tpy6Ky.
De3nHΦeKuzy
Pn npoBeHnn De3nHΦeKuHn pacnbIInTeJn npnHaJIeXHOCTe TuaTeJbHo Co6IIOaIte CJeDyUOuNe npabNa. PekomeHdyETcA De3nHΦuIpOBaTb DeTaII npi6opa He pexe Yem NocJe kaxdOro NocJeHero npimHeHna 3a DeHb.
(Дя STORO Bam notpe6yetcHemHoro 6ecuBETHORO yKcyca n ductnnpoBaHHa BOa.)
- ChaçaIa oUcHtIe paCnblntelb n npHaJNeJxHOCTN, kA KOnIcaHb B pa3dene «OuIcTka».
- IomeCTIte pa3o6paHHbI paCnblNTeIb HOCOBOn dyu, MyHdUyK n pa3o6paHHyO HacaKy dIra Ho-ca Ha 5 MInHyT B KInPaUyO BDOy.
-Дя npoqunx npnHaJIeXHocTe IcNoIb3yIte paCTBOp,coCTOaIu Ha 1/3 uKcUca n Ha 3/4 uN DCtIINIpOBaHHo BODbl. Y6eIITecb, yTO xNIOKOCTN DOCTaTOUHO, yTO6bl MOxHO 6blNO NOLHO TIOrpy3ntB HHe pa3LIuHbIe DeTaN (HaNPmep, paNblNTeJb, MaCKy I MyHdTuK). - OctabBe DeTaJI Na Ha 30 MInHyT ByKcyCHOM pAcTBope.
-ПомоуTe DeТалиВОди n pa3лoxкnte n3 nIpyocuSKnHa MЯrkOM NOJOTeHc.

BHHMaHHe
MaCKn I BO3dUshbI uHaHr He KInpTntb H He CTePnJIIN30BaTb B aBTOKnaBe.
- Pocne noHoro BbIcIxHaNna co6epnte deTaNN n noJoxnTe INB cyxHyrepeMeTuHyo emKoCTb.
i yka3aHne
UyTnte, yTO nocIe OuNCTKN Heo6xOdmo NOHOCbIO BbICyWHTb DeTaII, TaK KaK B IpOTnBHom Clyuae BO3- pAcTaET pNCK pa3MHOKeHn8 6aKTeprn.
Cyuoka
-ПОLOЖNTe OTdJIbHbIe DeTaIIHa cyxuO,чИCTyIO IN BnITbIBaIOU OBy BlaIg NOcTnIKy N OCTaBBTe Do NOJHOro BbICbIXaHnI (He MeHee 4 YacOB).
YCTOUIBOCTb MaTePnJa
- KaN Bce DeTaN N3 PJIaCTnKa, paCnblIITeNb I npHnHaJIeXHOCTN N3HaUNBaOTcR npu YAcTOM nCNOJb3OBAHN I rIINHeHueCKo O6pa60TKe. Co BpeMeHem 3TO MOKET pPnBEcTN K N3MeHeHIO COCTaBAa3pO3OJN I, KaK CNECDTBne, CHN3NTb 3ΦΦeKTNBHOCTb JleHnra. IOnTOMy peKOMeHdyETcR exKeOrdHO3aMeHrTb paCnblIITeNb.
-Пи Вьборе срсдва ДЯ OчNTКИ De3HФЕкцИ Нeo6xODIMO yHTbIbTb CLEdYIOоe: ИСпОЛБ3уЛТe TOЛБКО МягКо УСТЯооe CрсДТВО ИЛ Nde3HФИ ЦрУIOооe CрсДТВО B KОЛЧEcTBe, yka3aHHom ПОНЗВОДИТЕМ.
XpaHeHne
- HeIb3xpaHnTb np6Op BO BJIaXhBIX NOMEueHnax (HaIpUmEp, B BaHHoKOMHaTe) npepeBO3ntb erO BMeCTe C BJIaXhBIMN ppeMetAm.
XpaHnTe n nepeBO3nTe npu6Op B MeCTe, 3aunueHNOM OT dIInTeJIbHO BO3dEINCTBnA INPmBX COJHeuHbIX Jyuei.
-Пинадлжно мохно xpaHntbВ cneuaJIbHo npedHa3NaeHHOMДЯЗTOROOTceKe[4].XpaHnte np6opBcYxOMMeCTe,nyuWe Bcero,BynaKOBKe.
9.Утлизацnia
B cIeJx 3aunTbI OkpykaIOuei cpebl np6op cIeDyET yTNIN3NPOBaT OTdJIbHO OT 6bITOBORo Mycopa. Pnp6Op cIeDyET yTNIN3NPOBaT corlacho DnpeKTNBe
EC no OTxOJaM 3JIeKTpUcEsCKOro n 3JIeKTpOnHoro o6OpyDoBaHnra - WEEE (Waste Electrical and Electronic Equipment).
Pn BO3HKnHOBeHm BOpocOB, oBaTecb
B KOMMyHaJIbHyIOpraHn3aLIO, 3aHIMaIOUyOcR BO- nPoCaMn yTnIn3aCmN.
10. Peшене npo6jem
| ПюбLEMы/Воросы | Возможнaya р孥ни/устраенье |
| РасьліntельHe пОНЗВОДNT aЗрозольИли пОНЗВОДNT erо В He-ДостаTOHOM KOLINCHECTBE. | 1.В расьліnteile с liabilities KMORIJIcSLIIKOM MaLo Jelkap-CTBENHOrO npenapata.MinHMaJIbHoe KOJIYecTBo le-KapCTBENHOrO npenapata: 2 Ml,МakCSMJIbHoe KOJIYecTBO le-KapCTBENHOrO npenapata: 8 Ml |
| 2.Пюверыт, He засориLOSBЛICONLO.PriN Heo6xOIMOCSTN OUHCTNITe cOnLO (HaNPIMeP, npombl erO).ПосLE 3ToRr BHOBb BKJIOChte pасьліntelь.BHIMAHINE! MeJIKne OTBepCTNIAOCTOPOXHnO npotbIkaIte TOnbKOc HIXKHeI CTOpOHbI cOnla. | |
| 3.РасьліnteIL He B VeRTNKaJIb-HOM ПОLOXKeHNI. | |
| 4.ИсpoNsIb3yEmbl JelkapCTBENHbIйnpenapat He noDxODHT ДЯ paC-пылЕнna (HaNPIMeP, XmIDKOCTb CLIshKOM ructa).ЛекарSTBOdoJXHnO 6bITb Ha3NaYe-HO BpauOM. | |
| ОьемпolyuaemOTOaЗроЗлЯ сLIIshKOM mAl. | Шланг поERHyT, ФильТр засорен, сLIIshKOM bOllySObE KOJIYecTBO pactBOPa Дя ИнгаляцIII. |
| КашиMe-DnIkaMeHTbIMOxHNo Ic-пOLь3OBaTbДЯ INHgAIIaIzII? | ПюронсультуprуITEСь ob 3TOMc BpauOM.B prinuzne, moxHNo primeHЯтБлЮБbie lekapCTBa, noDxOJaIzneIdoJpySeHHbIe K IcNOLb3OBaHInOДЯ INHgAIIaTPOB. |
| В расьліnte-Île octaETcRaPCTBOPдЯ INHgAIIaIzII. | Это HopMaJIbHо и рюисхODIT NOTEXHnueCKIM prinuHam.Если bblycIbIshTe,чTO 3Byk,ИЗдаваe-Mый расьліntelem рпri pa6Ote,сИльно ИЗМeHnIcSа,срAZу ќeпpeKpaTITe ИнгаляцIII. |