IH 40 - Inhaler BEURER - Free user manual and instructions
Find the device manual for free IH 40 BEURER in PDF.

| Product type | Ultrasonic inhaler (nebulizer) |
| Brand | Beurer |
| Model | IH 40 |
| Dimensions (L × W × H) | 201 × 147 × 141 mm |
| Weight | 315 g |
| Power supply | Mains adapter 100-240 V~, 50-60 Hz, 0.15 A; Output DC 13.5 V, 1 A |
| Water tank capacity | Max. 20 ml |
| Medication container capacity | Max. 8 ml |
| Medication flow rate | Approx. 0.4 ml/min |
| Ultrasound oscillation frequency | 1.7 MHz |
| Particle size | 1 to 5 µm |
| Automatic shut-off | Yes, after 15 minutes or when medication is depleted |
| Airflow adjustment | Yes, via dial |
| Included accessories | Nebulizer with container, mouthpiece, adult mask, child mask, tube, mains adapter, storage bag, replacement filter, user manual |
| Housing material | ABS |
| Cleaning and disinfection | Clean with hot water; disinfect with 70% ethyl alcohol or boiling water (mouthpiece); replace container after each use |
| Filter replacement | Every 10 uses or at most every 6 months |
| Warranty | 5 years |
| Expected service life | 5 years |
| Operating conditions | Temperature: +5 °C to +40 °C; Relative humidity: < 85% |
| Storage and transport | Temperature: -20 °C to +70 °C; Relative humidity: < 95% |
| Safety class | BF type device, safety class II |
| Available spare parts | Medication container (REF 163.373), Year-Pack (REF 601.07) containing mouthpiece, masks, tube, containers, filters |
Frequently Asked Questions - IH 40 BEURER
User questions about IH 40 BEURER
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Inhaler in PDF format for free! Find your manual IH 40 - BEURER and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. IH 40 by BEURER.
USER MANUAL IH 40 BEURER
natural_image
White beurer device with clear glass cover and handle, no visible text or symbols on the device itselftext_image
Labeled diagram of various household air purifier components with numbered parts6. Inbetriebnahme
Aufstellen
natural_image
Line drawing of a beurer device with liquid pouring from a cylindrical container (no text or symbols)natural_image
Line drawing of a hairdryer with a handle and base device (no text or symbols)
Hinweis
natural_image
Line drawing of a hand holding a curved object, possibly a tool or component (no text or symbols present)- Getting to know your device ....13
- Signs and symbols....13
- Warnings and safety notes....14
- Useful information regarding the device.....15
- Description of nebuliser device and accessories ....16
- Initial use....16
- Operation....17
- Inhaling correctly .....18
- Changing the filter ..... 19
- Cleaning and disinfection....19
- Technical specifications ....21
- Replacement parts and wearing parts .....21
- Disposal....22
- Warranty / Service 22
Included in delivery
See Description of units and accessories, page 15.
- Ultrasonic atomiser with medication container
- Mouthpiece
- Adapter
- Adult mask
- Children's mask
- Replacement filter
- Mains part
- Storage pouch
• These instructions for use
1. Getting to know your device
Dear customer,
Thank you for choosing one of our products. Our name stands for high-quality, thoroughly tested products for applications in the areas of heat, weight, blood pressure, blood glucose, body temperature, pulse, gentle therapy, massage and air.
Please read these instructions for use carefully and keep them for later use, be sure to make them accessible to other users and observe the information they contain.
With kind regards,
Your Beurer team
Application area
This nebuliser is an ultrasonic atomiser for treating the upper and lower airways.
The aerosol generated by the high-frequency vibration has a predominant droplet size of 1 – 5 m and is therefore very well suited for transporting the nebulised active agents directly to the lower airways.
By nebulising and inhaling the medication prescribed/recommended by your doctor, you can prevent diseases affecting the airways, or in the case that you contract such an illness, you can alleviate symptoms and speed up your recovery. Ask your doctor or pharmacist for further information about the potential applications.
The device is suitable for inhalation at home and can also be used on multiple occasions in one day when on the move. Inhaler medication should only be used following instructions to do so from a doctor. Ensure you are calm and relaxed when inhaling the medication and breathe slowly and deeply to ensure that the medication reaches right down to the lower bronchi. Breathe out normally.
Once it has been properly prepared, the device can be used again. Preparation involves replacing all the accessories, including the air filter, and disinfecting the surface of the device using a standard disinfectant.
Please note that all accessories should be replaced if the device is used by more than one person.
We recommend that you replace the atomiser and other accessories after one year.
2. Signs and symbols
The following symbols appear in these instructions for use.

Warning
Warning instruction indicating a risk of injury or damage to health.

Important
Safety note indicating possible damage to the unit/accessory.

Note
Note on important information.
The following symbols are used on the packaging and on the type plate for the device and accessories.
| Applied part, type BF | |
| Observe the instructions for use | |
| Manufacturer | |
| On/off | |
| Serial number | |
| Protection class 2 device | |
| Do not use outdoors | |
| IP22 | Protected against foreign objects equal to or greater than 12.5 mm and against angular falling drops of water |
| CE0483 | CE labellingThis product satisfies the requirements of the applicable European and national directives. |
 PAP PAP | Dispose of packaging in an environmentally friendly manner |
3. Warnings and safety notes

Warning
- Before use, ensure that there is no visible damage to the device or accessories. If you have any doubts, do not use the device and contact your retailer or the specified Customer Services address.
- The device is not a substitute for medical consultation and treatment. Talk with your doctor first if you are experiencing any pain or are suffering from an illness.
- If you are pregnant or if you suffer from a chronic disease or other condition, consult your doctor before starting a course of inhalation therapy.
- If you have health concerns of any kind, consult your GP!
- Please note the general hygiene measures when using the nebuliser.
- For hygiene reasons, every user should use their own set of accessories (medication container, hose, mask, mouthpiece).
- You should always follow the instructions of your doctor regarding the type of medication to use, the dosage, and the frequency and duration of inhalation.
- Only use medication prescribed or recommended by your doctor or pharmacist.
- If the device does not work properly, or you feel unwell or experience pain, stop using it immediately.
- Keep the device away from your eyes when it is in use, as the nebulised medication could be harmful.
- Never leave the device unattended when it is switched on.
- Do not remove the upper cover while the device is in operation (when the blue light is illuminated).
- Do not operate the device in the presence of flammable gases.
- Do not operate the device near electromagnetic transmitters.
- This device is not intended for use by children or people with restricted physical, sensory (e.g. reduced sensitivity to pain) or mental skills or a lack of experience and/or lack of knowledge, unless they are supervised by a person who is responsible for their safety or are instructed by such a person in how to use the device.
- Check in the medicine package leaflet for possible contraindications for use with common aerosol therapy systems.
- Keep packaging material away from children (risk of suffocation).
- Do not use any additional parts that are not recommended by the manufacturer.
- Never submerge the device in water or other liquids and do not use it in the bathroom. Under no circumstances may liquid enter the device. However, if the device does fall into a liquid do not pull it out; instead, disconnect it from the power supply immediately.
- Protect the device from heavy impacts.
- If the device has been dropped, exposed to high levels of moisture or suffered any other damage, it must no longer be used. If in doubt, contact Customer Services or the retailer.
- The IH 40 may only be operated with the designated Beurer accessories. The use of accessories made by other companies may result in less efficient therapy and could damage the device.
- Do not pull the mains plug out of the socket using the mains cable.
- Do not crush or bend the mains cable, pull it over sharp-edged objects or leave it dangling down, and protect it from sources of heat.
- If the mains connection cable of this device is damaged, it must be disposed of. If it cannot be removed, the device must be disposed of.
- Ensure that you only insert and remove the mains part with dry hands and that you only press the ON/OFF button with dry hands.
- During operation, the mouthpiece or mask can reach a temperature of 43^ C degrees within 15 minutes.
- If the nebuliser motor is blocked, this does not pose a safety risk to the user.
- The product has no Essential Performance whose absence or degradation would result in an unacceptable risk.

Important
- Power cuts, sudden interferences or other unfavourable conditions could lead to the device becoming inoperable. We therefore recommend that you obtain a replacement device or medication (the latter should be agreed with your doctor).
- Do not use the device when the water tank or medication container is empty. Only add clean tap water or distilled water to the water tank. Other liquids could cause irreparable damage to the device.
- Do not store the device near to sources of heat.
- Do not use the device in a room in which a spray has previously been used. Air the room before carrying out the therapy.
- Store the device in a location protected against climatic influences. The device must be stored in the environmental conditions specified.
- Do not use any powdered medication (including in a solution).
- Do not shake the atomiser as this may cause liquid to leak out and damage the device.
Mains part

Important
- Only use the mains part supplied.
- Ensure that the power supply socket does not come into contact with any liquids.
- Should you require an adapter or extension lead, this must meet the applicable safety requirements. The power limit and the maximum output specified on the adapter must not be exceeded.
- Always unplug the mains part from the mains socket after use.
- The device must only be connected to the mains voltage that is specified on the type plate.
General notes

Important
• The device is only to be used:
- on people, - for the purpose for which it is designed (aerosol inhalation) and in the manner specified in these instructions for use.
- Any form of improper use can be dangerous.
- In the event of an acute emergency, the provision of first aid has top priority.
- Only use the medication with a saline solution.
- This device is not intended for commercial or clinical use; it is designed exclusively for self-treatment in a private home.
Prior to initial use

Important
- Remove all packaging material before using the device.
- Protect the device from dust, dirt and humidity.
-
Do not operate the device in a very dusty area.
-
Ensure the device and accessories are at room temperature before using them.
- Switch the device off immediately if it is faulty or not working properly.
- The manufacturer is not liable for damage resulting from improper or careless use.
Repairs
i Important
- Never open the device, modify it or repair it; otherwise proper operation can no longer be guaranteed. Failure to comply with this instruction can be dangerous, and will also invalidate the warranty.
- The device is maintenance-free.
- For repairs, please contact Customer Services or an authorised retailer.
4. Useful information regarding the device
Accessories
Only use the accessories recommended for use by the manufacturer; safe function is only guaranteed when this requirement is met.
Automatic switch-off
The device has an automatic switch-off function.
When there is only a very small amount of medication or fluid left, the device switches off automatically. The device switches off automatically after 15 minutes of operation. If you wish to continue the treatment, switch the device back on by pressing the ON/OFF button.
Do not operate the device when the medication container or water tank is empty.
5. Description of nebuliser device and accessories
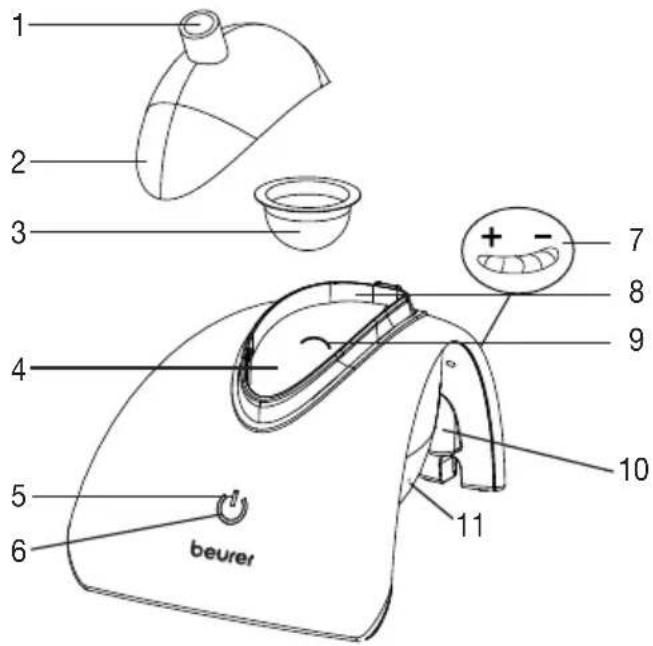
Overview of nebuliser
1 Accessory connector
2 Cover
3 Medication container
4 Water tank
5 ON/OFF button
6 LED indicator for displaying the opera - ting status
7 Control dial for airflow
8 Housing
9 Water level fill line
10 Connector socket for the mains part plug
11 Filter cap with filter
text_image
1 2 3 4 5 6 beurer 7 8 9 10 11Overview of atomiser and accessories
12 Connector for the masks
13 Adult mask
14 Children's mask
15 Mouthpiece
16 Hose
17 Mains part
18 Storage pouch
19 Replacement filter
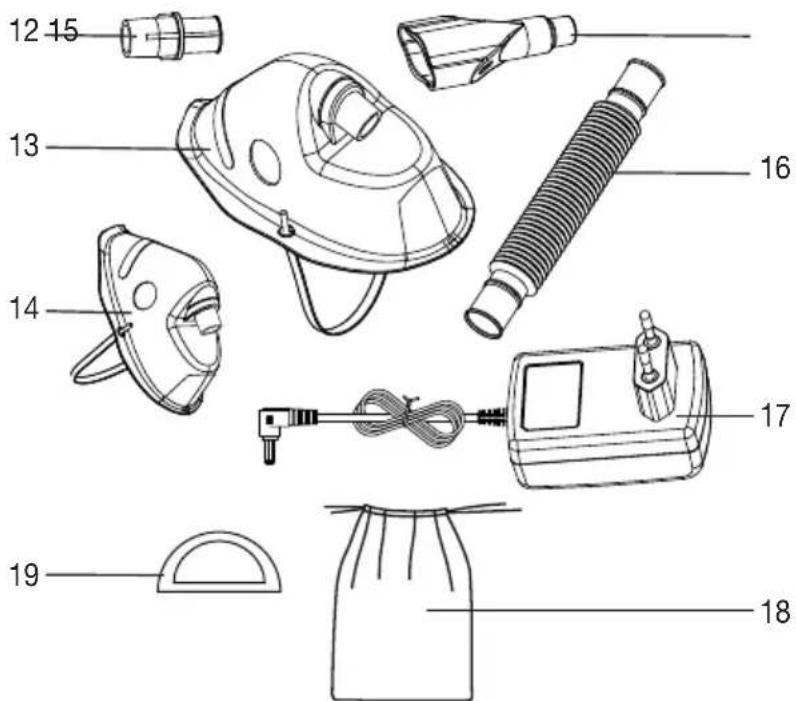
text_image
Labeled diagram of various household air purifier components with numbered parts6. Initial use
Positioning the device
Take the device out of the packaging. Place the device on a hard, even surface. Make sure that the vents are free from obstructions. Remove the accessories from the packaging.
Before using the device for the first time
i Note
- Clean and disinfect the atomiser and accessories before using them for the first time. See 10. "Cleaning and disinfection".
7. Operation
Preparing the atomiser
- Before using them, always clean and disinfect the water tank, the cover, the connector for the masks, the extension hose, the mouthpiece and the masks, according to the instructions under section 10. "Cleaning and disinfection".
If the therapy involves inhaling several different medications one after the other, please be aware that the atomiser must be rinsed under warm tap water following every usage. See section 10. “Cleaning and disinfection”.
- Place the device on an even surface.
- Remove the cover (image 1).
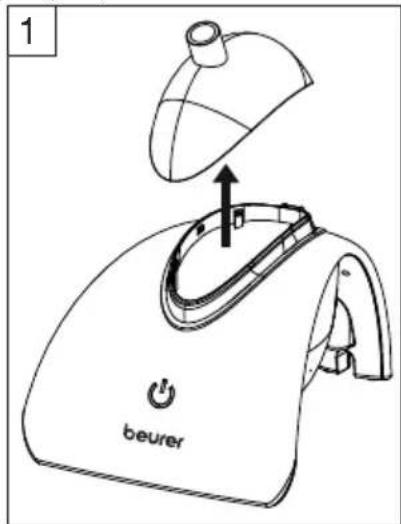
text_image
1 beurer
Note
- Ensure that there is a mains socket close to the set-up area.
- Arrange the mains cable so that no-one will trip over it.
- To disconnect the nebuliser from the mains after nebulisation, first switch off the device and then pull the mains plug out of the socket.
Operating the mains part
- Only connect the mains part [17] using the voltage specified on the type plate.
- Insert the mains part connector plug [17] into the provided connector socket [10] and fully insert the mains part [17] into a suitable mains socket.
Filling the nebulisation chamber
The tank capacity is 20 ml. Fill the water tank [4] with clean water (preferably distilled water) up to the water fill line [9]. This water is used as the fluid through which the ultrasonic waves are guided to the medication. Do not pour medication directly into the water tank.
Filling the medication container
Place the medication container in the water tank (image 2) and then fill it with the medication (image 3).
Ensure that the maximum capacity of 8 ml is not exceeded.
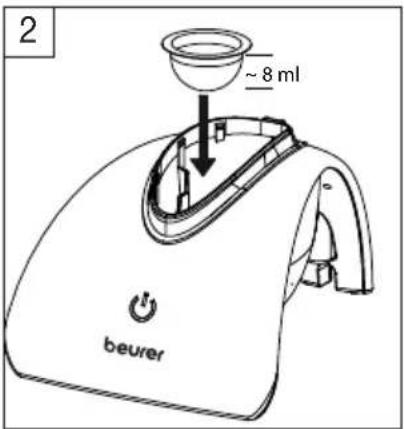
text_image
2 ~ 8 ml beurerFor this purpose, refer to the marker lines on the medication container (image 2.1).
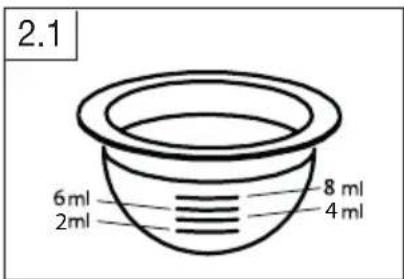
text_image
2.1 6ml 2ml 8ml 4mlAdhere to the instructions for use and dosage instructions for the medication to be used. If it is necessary to dilute a medication with an isotonic sodium chloride solution, first pour the solution into the medication container and then add the required dosage of

natural_image
Line drawing of a beurer device with liquid pouring from a cylindrical container (no text or symbols)Closing the nebulisation chamber
Close the nebulisation chamber using the cover provided [2] (image 4).
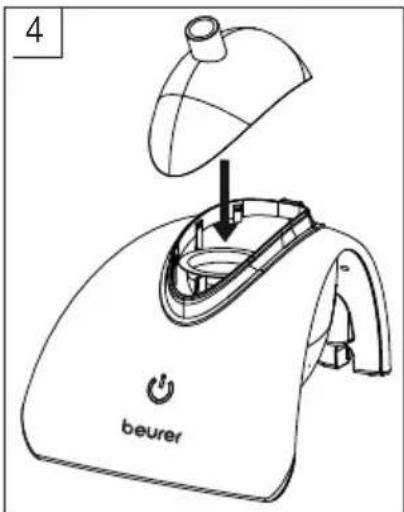
text_image
4 beurerConnecting an accessory
Now connect the hose to the accessory connector [1] on the nebulisation chamber (image 5).
Now connect the accessory you wish to use (mask with connector or mouthpiece) to the hose.
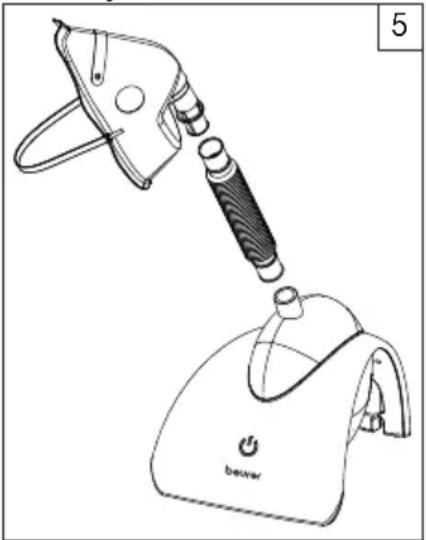
natural_image
Line drawing of a hairdryer with a handle and base device (no text or symbols)Note
The most effective form of nebulisation is by using the mouthpiece. Nebulisation using a mask is only recommended if it is not possible to use a mouthpiece (e.g. for children who are not yet able to inhale using a mouthpiece).
If you are using the adult mask [13] you can secure it to your head with the adult mask strap. There is also a strap for the children's mask [14].
When using a mask to inhale, take care to ensure the mask fits well and the eyes are unobstructed.
When using a mask to inhale, take care to ensure the mask fits well and the eyes are unobstructed.
Switch on the device by pressing the ON/OFF button. When the device is functioning correctly, a mist is then generated for inhalation.
Airflow control dial
You can adjust the airflow using the airflow control [7]. You can increase the airflow by turning the airflow control to the left and decrease it by turning it to the right.
Stopping inhalation
- When you have finished the treatment, switch off the device using the ON/OFF button [5].
- The LED indicator for the operating display [6] goes out.
- If the inhalant is nebulised, the device switches off automatically. For technical reasons, a small amount of medication remains in the medication container [3].
- Do not use this.
- Disconnect the mains part [17] from the power supply.
Carry out cleaning
• See 10. "Cleaning and disinfection".
8. Inhaling correctly
Important
- Do not tilt the device or position it at an angle.
- Essential oils, cough syrups, gargling solutions and drops to be used as a rub or in a steam bath are wholly unsuitable for inhalation using a nebuliser. Such additives are often viscous and can impair the correct functioning of the device and the effectiveness of the application in the long-term.
- For individuals with a hypersensitive bronchial system, medications containing essential oils may under certain conditions cause an acute bronchospasm (a sudden cramp-like restriction of the bronchi with shortness of breath). Consult your doctor or pharmacist in relation to this matter!
Breathing technique
- It is important to use the correct breathing technique to ensure that the particles are distributed as widely as possible across the airways. To ensure that the particles reach your airways and lungs, you must breathe in slowly and deeply, then hold your breath briefly (5 to 10 seconds) and then breathe out quickly.
- Nebulisers should only be used for the treatment of respiratory diseases after consultation with your doctor. He or she will advise you about the selection, dosage and use of medications suitable for inhalation therapy.
- Certain medications are only available with a prescription.
When inhaling through the mouthpiece
- Use the mouthpiece if you are suffering from a condition of the lower airways, such as a cough or a bronchial infection.
- Sit up straight.
- Connect the hose [16] to the accessory connector [1]. Then connect the mouthpiece [15] to the hose.
- Place your lips around the mouthpiece.
- Switch on the device by pressing the ON/OFF button.
- Breathe in slowly and deeply through your mouth and breathe out through your nose. After breathing in, hold your breath briefly to increase the effectiveness of the therapy. Remain calm and relaxed during the treatment. Take breaths at regular intervals and do not breathe too fast. If you want to pause the inhalation for a short time, switch off the device by pressing the ON/OFF button and take out the mouthpiece.
When inhaling using the mask
- The inhalation mask is suitable for the treatment of the nasopharynx.
- Sit up straight.
- Connect the hose [16] to the accessory connector [1]. Now connect the connector [12] to the hose and the mask to the connector.
- Hold the mask pressed gently against your nose; the mask must cover your mouth and nose without squashing them.
- Take deep and slow breaths in and out through your nose. After breathing in, hold your breath briefly to increase the effectiveness of the therapy. Remain calm and relaxed during the treatment. Take breaths at regular intervals and do not breathe too fast. If you want to pause the inhalation briefly, switch off the device by pressing the ON/OFF button and remove the mask from your face for a moment. To continue with the inhalation, place the mask back over your nose and mouth and switch on the device by pressing the ON/OFF button.
Replacing the medication container
The medication container should be replaced following usage every time inhalation treatment is carried out. This is to avoid deposits and chemical reactions due to medical products. In the package you will find 12 medication containers which can be used to replace used containers. Additional containers can be obtained from the Customer Services address provided.
9. Changing the filter
Check the filter regularly (e.g. after every tenth use). Replace the used filter if it is very dirty or blocked (but at the latest after 6 month). If the filter has become wet, it must also be replaced with a new one.

Caution
- Do not try to clean and re-use the used filter!
- Only use original filters supplied by the manufacturer. Otherwise, your nebuliser may become damaged or effective therapy can no longer be guaranteed.
- Never operate the device without a filter.
To replace the filter, proceed as follows:

Caution
- First switch off the unit and unplug it.
- Allow the unit to cool
- Remove the filter cap [11] in a downwards motion.
natural_image
Line drawing of hands holding a mechanical component (no text or symbols)- After replacing the old filter with a new one, replace the filter cap.
- Ensure that it is securely in place.
10. Cleaning and disinfection

Warning
Adhere to the following hygiene instructions to avoid health risks.
- The atomiser and accessories are designed for multiple usage. Please note that different areas of application involve different requirements in terms of cleaning and hygienic preparation.
- Please consult your doctor about the additional requirements in terms of the hygienic preparation needed (hand care, handling of medication/inhalation solutions) for high-risk groups (e.g. patients with cystic fibrosis).

Important
- The device must be switched off and allowed to cool down each time before cleaning (the ultrasonic transducer heats up considerably during usage).
- Do not clean the ultrasonic transducer or the accessories mechanically using a brush or similar device, as this could cause irreparable damage and it will mean that the best treatment results can no longer be guaranteed.
- Ensure that no water gets inside the device!
- Do not clean the device or accessories in the dishwasher!
- Do not touch the device with wet hands when it is switched on and do not allow water to spray on the device. The device may only be operated when it is completely dry (this excludes the liquids that are required to be used in the water tank and the medication container).
- Do not spray any liquid into the vents! If liquid penetrates the device this could cause damage to the electronics or other nebuliser parts and lead to a malfunction.
Cleaning
When you have detached the device from the mains socket, remove the cover and if required remove any remaining residue from the medication container and the nebulisation chamber.
The used accessories such as the mouthpiece, mask, adapter, etc. must be washed with hot water after each use. Dry the parts carefully using a soft cloth and place the parts in a dry, sealed container or disinfect them.
- Clean the tank and the cover with a cloth soaked in water or vinegar.
- Clean the surface of the device using a damp cloth (water or a mild cleaning solution). Dry the device using a lint-free cloth..

Important
- Never hold the whole device under running water to clean it.
- When cleaning, ensure that any residue is removed and dry all parts carefully.
- Never use any substances for cleaning that could potentially be dangerous if they came into contact with the skin or mucous membranes, or if they were swallowed or inhaled.
Disinfection
Please carefully observe the points below when disinfecting your atomiser and accessories. We recommend disinfecting the individual parts on a daily basis after the last usage as a minimum measure.
You can disinfect the IH 40 and its accessories in two ways: using 70% ethyl alcohol or boiling water. First, clean the atomiser and accessories as described in the “Cleaning” section. Then continue with the disinfection.
- Drop a few drops of ethyl alcohol onto the ultrasonic transducer and leave this to work for 10 minutes.
- Place the used accessory parts in ethyl alcohol for 10 minutes or use an alcohol-based disinfection spray.
- Then clean all the parts again under running water.
Disinfection with boiling water

Important
The facial masks and the hose must not be submerged in hot water!
- We recommend disinfecting the masks and hose using commercially available alcohol-based disinfectant.

Note
- The mouthpiece can be disinfected in boiling water for 10 minutes.
- Avoid touching the hot base of the pan in this case.
- You can also disinfect the mouthpiece using a commercially available vaporiser. For more information, please see the instructions for use of the vaporiser manufacturer.
Drying
- Dry the parts carefully using a soft cloth.
- Place the individual parts on a clean, dry and absorbent surface and leave them to dry completely (at least 4 hours).
i Note
Please ensure that the parts are completely dried after cleaning and disinfection before they are used again or stored; otherwise the risk of bacterial growth is increased.
Put the parts together again if these are completely dry and place the parts in a dry, sealed container.
Durability of materials
- The following point should be observed when selecting the cleaning product or disinfectant: only use a mild cleaning product or disinfectant in the concentration and volume prescribed by the manufacturer.
- As with any plastic parts, atomisers and their accessories are affected by a certain amount of wear and tear when used and hygienically prepared on a frequent basis. Over time, this can lead to a change in the aerosol, which can have a negative effect on the efficiency of the therapy.
We therefore recommend that you replace the accessories after no more than a year.
Storage
- Do not store in a room with a high level of dampness (such as a bathroom) and do not transport with any damp items.
- When storing and transporting, protect from prolonged direct sunlight.
Troubleshooting
| Problem/question | Possible cause/remedy |
| The atom-iser does not produce any aerosol or produces too little aerosol. | 1. No water in the water tank. |
| 2. Insufficient amount of medication in the atomiser. | |
| 3. Do not hold the atomiser vertically. | |
| 4. Unsuitable medication fluid filled for nebulisation (e.g. too viscous – viscosity must be less than 3). The medication fluid should be prescribed by the doctor. | |
| The output is too low. | Check whether the control dial for the airflow is set correctly. |
| What medications are suitable for inhaling? | Naturally, only your doctor can decide which medication should be used for treating your condition. Please consult your doctor in relation to this matter.With the IH 40 you can nebulise medications with a viscosity of less than 3. |
| There is inhalation solution residue in the medication container. | This is normal and is necessary for technical reasons. Stop the inhalation as soon as you can no longer detect nebulisation or the device switches off automatically due to a lack of inhalant. |
| What should be taken into account when using the device with infants and children? | 1. On babies, the mask should cover the mouth and nose to ensure effective inhalation.2. On children, the mask should also cover both the nose and mouth. It is not a good idea to carry out nebulisation on someone who is sleeping, as in this case not enough of the medication will reach the lungs.Note: Inhalation should only be carried out under the supervision of an adult and with their assistance and the child should not be left alone. |
| Does inhalation using the mask take longer? | This is necessary for technical reasons. You breathe less medication per breath through the holes of the mask than via the mouthpiece. The aerosol is mixed with ambient air via the holes. |
| Should each person have their own atomiser? | Yes, this is absolutely essential. |
11. Technical specifications
Dimensions
(LxWxH) 201 x 147 x 141 mm
Weight 315 g
Filling volume Medication container max. 8 ml Water tank max. 20 ml
Medication flow rate approx. 0.4 ml/min
Ultrasonic transducer frequency 1.7 MHz
Housing material ABS
Power supply 100–240 V\~; 50–60 Hz; 0.15 A DC output: 13.5 V; 1 A
Expected service life 5 years
Operating conditions Temperature: +5 °C to +40 °C Relative humidity: < 85% non-condensing
Storage and transportation conditions Temperature: -20 °C to +70 °C Relative humidity: < 95% non-condensing
The serial number is located on the device or in the battery compartment.
Subject to technical changes.
Wearing parts are excluded from the guarantee.
Particle size diagram
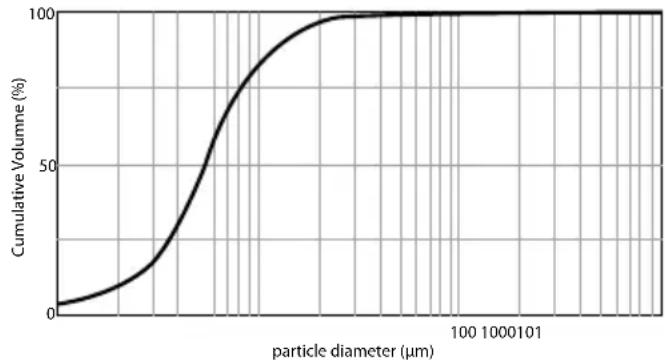
line
| particle diameter (μm) | Cumulative Volume (%) | | ---------------------- | --------------------- | | 0 | 0 | | 100 | 50 | | 1000 | 95 | | 10000 | 100 | | 100000 | 100 |The measurements were carried out with a sodium chloride solution using a laser diffraction method.
The diagram may therefore not be applicable to suspensions or highly viscous medications. You can obtain more detailed information from the manufacturer of your medication.
12. Replacement parts and wea-ring parts
Replacement parts and wearing parts are available from the corresponding listed service address under the stated material number.
| Name | Material | REF |
| Medication container | PET | 163.373 |
| Year pack (contains mouthpiece, adult mask, children's mask, connector for masks, 12 medication containers, hose, 2 replacement filters) | PP, PVC, EVA, PET | 601.07 |
i Note
If the device is not used according to the instructions specified, perfect functionality cannot be guaranteed! We reserve the right to make technical changes to improve and develop the product.
This device and its accessories comply with the European standards EN60601-1, EN60601-1-2 (In accordance with IEC61000-3-2, IEC61000-3-3, IEC61000-4-2, IEC61000-4-3, IEC61000-4-4, IEC61000-4-5, IEC61000-4-6, IEC61000-4-11) and EN13544-1 and is subject to particular precautions with regard to electromagnetic compatibility. This device meets the requirements of European Directive 93/42/EEC for medical products, as well as those of the Medizinproduktegesetz (German Medical Devices Act).

Notes on electromagnetic compatibility
- The device is suitable for use in all environments listed in these instructions for use, including domestic environments.
- The use of the device may be limited in the presence of electromagnetic disturbances. This could result in issues such as error messages or the failure of the display/device.
- Avoid using this device directly next to other devices or stacked on top of other devices, as this could lead to faulty operation. If, however, it is necessary to use the device in the manner stated, this device as well as the other devices must be monitored to ensure they are working properly.
- The use of accessories other than those specified or provided by the manufacturer of this device can lead to an increase in electromagnetic emissions or a decrease in the device's electromagnetic immunity; this can result in faulty operation.
- Failure to comply with the above can impair the performance of the device.

Warning
The use of accessories, transducers and cables other than those specified with the exception of transducers and cables sold by the manufacturer of IH 40 as replacement parts for internal components may result in increased emissions or decreased immunity of IH 40.
13. Disposal
For environmental reasons, do not dispose of the device in the household waste at the end of its useful life. Dispose of the unit at a suitable local collection or recycling point. Dispose of the device in accordance with EC Directive – WEEE (Waste Electrical and Electronic Equipment). If you have any questions, please contact the local authorities responsible for waste disposal.

14. Warranty / Service
Beurer GmbH, Söflinger Straße 218, 89077 Ulm, Germany (hereinafter referred to as "Beurer") provides a warranty for this product, subject to the requirements below and to the extent described as follows.
The warranty conditions below shall not affect the seller's statutory warranty obligations which ensue from the sales agreement with the buyer. The warranty shall apply without prejudice to any mandatory statutory provisions on liability.
Beurer guarantees the perfect functionality and completeness of this product. The worldwide warranty period is 5 years, commencing from the purchase of the new, unused product from the seller.
The warranty only applies to products purchased by the buyer as a consumer and used exclusively for personal purposes in the context of domestic use. German law shall apply.
During the warranty period, should this product prove to be incomplete or defective in functionality in accordance with the following provisions, Beurer shall carry out a repair or a replacement delivery free of charge, in accordance with these warranty conditions.
If the buyer wishes to make a warranty claim, they should approach their local retailer in the first instance: see the attached “International Service” list of service addresses.
The buyer will then receive further information about the processing of the warranty claim, e.g. where they can send the product and what documentation is required.
A warranty claim shall only be considered if the buyer can provide Beurer, or an authorised Beurer partner, with
- a copy of the invoice/purchase receipt, and
- the original product.
The following are explicitly excluded from this warranty:
- deterioration due to normal use or consumption of the product;
- accessories supplied with this product which are worn out or used up through proper use (e.g. batteries, rechargeable batteries, cuffs, seals, electrodes, light sources, attachments and nebuliser accessories);
- products that are used, cleaned, stored or maintained improperly and/or contrary to the provisions of the instructions for use, as well as products that have been opened, repaired or modified by the buyer or by a service centre not authorised by Beurer;
- damage that arises during transport between manufacturer and customer, or between service centre and customer;
- products purchased as seconds or as used goods;
- consequential damage arising from a fault in this product (however, in this case, claims may exist arising from product liability or other compulsory statutory liability provisions).
Repairs or an exchange in full do not extend the warranty period under any circumstances.
FRANÇAIS
Sommaire
text_image
Labeled diagram of household air purifier components with numbered parts for identification6. Mise en service
Installation
text_image
2.1 6ml 2ml 8ml 4mlnatural_image
Line drawing of a beurer device with liquid being poured into it (no text or symbols)natural_image
Line drawing of a hair dryer with a brush and accessories, no text or symbols presenti Remarque
natural_image
Line drawing of a hand holding a circular object with a finger, no text or symbols presenttext_image
Labeled diagram of household air purifier components with numbered parts for identificationtext_image
2.1 6ml 2ml 8 ml 4 mlnatural_image
Line drawing of a beurer device with liquid pouring from a cylindrical container (no text or symbols)natural_image
Line drawing of a hairdryer with a handle and base device (no text or symbols)
Nota
natural_image
Line drawing of a hand holding a ring, no text or symbols presenttext_image
Labeled diagram of various household air purifiers and household items with numbered partstext_image
2.1 6ml 2ml 8ml 4mlnatural_image
Line drawing of a beurer device with liquid pouring from a cylindrical container (no text or symbols)natural_image
Line drawing of a hairdryer with a handle and base device, no text or symbols present
Nota
natural_image
Line drawing of hands holding a curved object, possibly a tool or component (no text or symbols)text_image
Labeled diagram of various household cleaning and cleaning items, including a vacuum cleaner, hose, and bag6. Çalıştırma
Kurulum
text_image
2.1 6ml 2ml 8ml 4mlnatural_image
Line drawing of a beurer device with liquid pouring from a cylindrical container (no text or symbols)natural_image
Line drawing of a hairdryer with a handle and base device (no text or symbols)