IH 50 - Inhaler BEURER - Free user manual and instructions
Find the device manual for free IH 50 BEURER in PDF.

| Product type | Vibrating membrane inhaler (nebulizer) |
| Brand | Beurer |
| Model | IH 50 |
| Dimensions (L x W x H) | 72 x 59 x 143 mm |
| Weight (with batteries) | 238 g |
| Power supply | 4 batteries 1.5 V AA (LR6) or mains adapter 100-240 V~, 50-60 Hz |
| Reservoir capacity | Max. 8 ml |
| Medication flow rate | Approx. 0.25 ml/min |
| Vibration frequency | 100 kHz |
| Housing material | ABS/TPE |
| Expected service life | 3 years |
| Operating conditions | +5°C to +40°C, relative humidity < 80% |
| Storage conditions | -10°C to +45°C, relative humidity < 80% |
| Included accessories | Membrane nebulizer, mouthpiece, adult mask, child mask, connection piece, power adapter, 4 batteries, storage pouch |
| Main functions | Automatic shut-off, spill protection, low battery indicator, use angle up to 45° |
| Maintenance and cleaning | Clean with hot water, disinfect with ethyl alcohol (70-75%) or boiling water (except masks) |
| Safety | Automatic shut-off at low level, IP22 protection rating, immediate shut-off in case of malfunction |
| Spare parts and repairability | Membrane nebulizer (ref. 162.711), Yearpack (ref. 603.05), annual replacement recommended |
| General information | Domestic use, non-professional, compliant with CE standards (93/42/EEC), NE60601-1, NE13544-1 |
Frequently Asked Questions - IH 50 BEURER
User questions about IH 50 BEURER
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Inhaler in PDF format for free! Find your manual IH 50 - BEURER and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. IH 50 by BEURER.
USER MANUAL IH 50 BEURER
- Getting to know your instrument 12
- Signs and symbols 12
- Safety information and warnings.. 13
- Useful information on the device. 14
5.Description of device and accessories 14 - Initial use. 15
- Operation 16
- Cleaning and disinfection 16
- Technical specifications 19
- Replacement parts and wearing parts 19
- Disposal 20
Included in delivery
For details of the delivery scope, see description of the device and accessories on page 14.
- Nebuliser and accessories
- Mesh atomiser with medicine container
- Mouthpiece
- Connecting piece
- Adult mask
- Child mask
Power pack - 4 x AA 1.5 V batteries
These instructions for use - Storage bag
1. Getting to know your instrument
Dear customer
thank you for choosing one of our products. Our brand stands for well-engineered, quality made products; driven by innovation and safety. Our product range encompasses a wealth of products for health and well-being in the heat, weight, blood pressure, body temperature, pulse, gentle therapy, massage, beauty, baby and air sectors.
Please read these instructions for use carefully and keep them for later use, be sure to make them accessible to other users and observe the information they contain.
With kind regards
Your Beurer team
Area of application
This nebuliser is a mesh atomiser for treating the upper and lower respiratory tract.
The nebulisation and inhalation of medication prescribed or recommended by a doctor can prevent against diseases of the respiratory tract, treat the side effects of these disorders and accelerate healing.
For more information on possible applications, consult your doctor or pharmacist.
The device is suitable for inhalation at home. Medicines should only be inhaled when instructed by a doctor. Inhalation should be performed in a calm and relaxed atmosphere. Inhale slowly and deeply to enable the medicine to reach the small bronchi deep into the lungs. Exhale normally.
Once prepared, the device is suitable for reuse. Reconditioning includes the replacement of all accessories including the mesh atomiser, as well as the disinfection of the surface of the device using a standard disinfectant. Please note that all accessories should be replaced if the device is used by more than one person.
We therefore recommend that you replace the atomiser and other accessories after a year.
2. Signs and symbols
The following symbols appear in these instructions.

Warning
Warning instruction indicating a risk of injury or damage to health.

Important
Safety note indicating possible damage to the unit/accessory.

Note
Note on important information.
The following symbols are used on the packaging and on the nameplate of the device and accessories.
| Type BF applied part | |
| Follow instructions for use | |
| Manufacturer | |
| On/Off | |
| SN | Serial number |
| Device in protection class 2 | |
| Do not use outdoors | |
| IP 22 | Protected against foreign objects equal to or greater than 12.5 mm in diameter and against drops of water falling at up to 15° from vertical |
| CE 0483 | The CE labelling certifies that the product complies with the essential requirements of Directive 93/42/EEC on medical products. |
3. Safety information and warnings

Warning
- Before use, ensure that there is no visible damage to the unit or accessories. When in doubt, do not use the unit and contact your dealer or the customer service address provided.
- Using the unit is not a substitute for consultation with or treatment from a physician. Whenever you have any pain or illness, always contact your physician first.
If you have health concerns of any kind, contact your general practitioner! - When using the nebuliser, always follow the basic hygiene procedures.
- Always follow your doctor's instructions regarding the type of medicine to be used and the dose, frequency, and duration of inhalation.
- Only use medicines that have been prescribed or recommended by your doctor or pharmacist.
- If the unit does not function correctly or if you begin to feel unwell or feel pain, stop using the unit immediately.
- During use, hold the unit away from the eyes, as some nebulised medicines may cause damage to the eyes.
- Do not operate the unit in the presence of flammable gases.
- Do not use the device near electromagnetic transmitters.
- This device is not intended to be used by children or persons with restricted physical, sensory (e.g. insensitivity to pain) or mental abilities or persons lacking in the required experience or knowledge for safe operation of the device, unless supervised or instructed by a person responsible for their safety.
- Check in the medicine package leaflet for possible contraindications for use with common aerosol therapy systems.
- Keep children away from packaging materials (risk of suffocation).
- Do not use attachments that are not recommended by the manufacturer.
- Do not dip the unit into water and do not use it in wet rooms. Do not allow any liquid to penetrate the unit.
- Protect the unit from strong impacts.
- Do not use the unit if it has fallen or been dropped on the floor, been exposed to extreme humidity or is otherwise damaged. In case of doubt, contact our customer service department or your local dealer.
- The IH 50 must only be operated using the appropriate Beurer accessories. The use of other accessories can impair therapeutic efficiency and may damage the device.
- Remove the batteries from the device if you will not be using it for an extended period. Leaking batteries may damage the device.

Important
- Power cuts, sudden faults or other unfavourable conditions may render the unit unusable. You are therefore recommended to keep a spare device or replacement medicine (as agreed with the doctor).
- The device must be stored away from sources of heat.
- Do not use the unit in a room in which a spray has previously been used. If this is the case, ventilate the room before commencing treatment.
- For reasons of hygiene, each user should use their own set of accessories (mesh atomiser, mask and mouthpiece).
- Store the device in a location protected against environmental influences. The device must be stored in the specified ambient conditions.
- Do not use any liquids that have a viscosity (thickness) greater than 3, as these may damage the mesh irreparably.
- Do not use any medicines in powder form (even if dissolved).
- Do not shake the atomiser, as this may cause liquid to leak, and the device may become damaged.
Power pack

Important
- Do not use any power pack other than the one supplied.
- Ensure that the mains socket cannot come into contact with liquids.
- If adapters or extensions are required, these must comply with the applicable safety regulations. The electrical capacity and the maximum capacity specified on the adapter must not be exceeded.
- After using the power pack, unplug it from the socket.
- Connect the unit only to the mains voltage listed on the type plate.
General notes

Important
- Use the unit only: - On humans,
for its intended purpose (aerosol inhalation) and in the manner described in these instructions for use. - Any improper use can be dangerous!
- In acute emergencies, first aid takes priority.
- Apart from medicines, use only a saline solution.
- This unit is not intended for commercial or clinical use, but only for individual, private household use!
Before using the unit for the first time

Important
- Before you use the unit for the first time, remove all packaging materials.
- Protect the unit against dust, dirt and moisture.
- Do not use the unit in very dusty environments.
- Before use, ensure the device and accessories are at room temperature.
- Switch the unit off immediately if it is defective or malfunctioning.
- The manufacturer shall not be held liable for damage or injuries caused by improper or incorrect use.

Notes on handling batteries
- If your skin or eyes come into contact with battery fluid, flush out the affected areas with water and seek medical assistance.
- Choking hazard! Small children may swallow and choke on batteries. Store the batteries out of the reach of small children.
- Observe the plus (+) and minus (-) polarity signs.
- If a battery has leaked, put on protective gloves and clean the battery compartment with a dry cloth.
- Protect the batteries from excessive heat.
- Risk of explosion! Never throw batteries into a fire.
- Do not charge or short-circuit batteries.
- If the device is not to be used for a long period, take the batteries out of the battery compartment.
- Use identical or equivalent battery types only.
Always replace all batteries at the same time. - Do not use rechargeable batteries.
- Do not disassemble, split or crush the batteries.

Warning
Leaking or damaged batteries can cause burns if they touch the skin. When removing such batteries, wear suitable protective gloves.
Repairs

Note
- Never open or attempt to repair the unit yourself, as otherwise proper function is no longer guaranteed. Failure to observe this regulation shall void the warranty.
- The device is maintenance-free.
- If you need to have the unit repaired, contact customer service or an authorised dealer.
4. Useful information on the device
Accessories
The safe functioning of the device can only be guaranteed if the accessories recommended by the manufacturer are used.

Important
Protecting against leaks
When filling the medicine container with medicine, ensure that you do not exceed the maximum mark (8 ml). The recommended filling amount is between 2 and 8 ml.
During use, you can tilt the device up to a maximum angle of 45^ in any direction, without impairing its nebulising function or the success of the treatment. However, spraying only takes place if the substance to be sprayed is in contact with the mesh. If it is not, spraying stops automatically.
However, please try to hold the device as upright as possible.
Automatic switch-off
The device has an automatic switch-off function. To prevent damage to the mesh, the device switches off automatically when the medicine/liquid has been almost entirely used up.
Do not use the device if the medicine container or water tank is empty.
The device switches off automatically if the substance to be sprayed is no longer in contact with the mesh.
5. Description of device and accessories
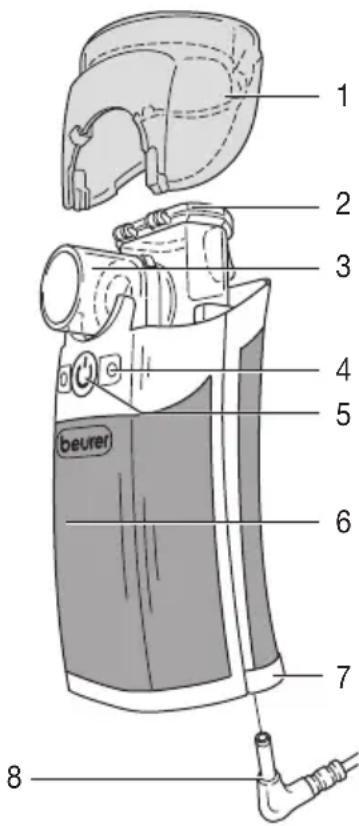
Overview
1 Cover
2 Medicine container
3 Mesh atomiser
4 Operation LEDs:
blue: device ready orange: change bat teries
5 On/off button
6 Casing
7 Battery compartment
8 Power pack connector
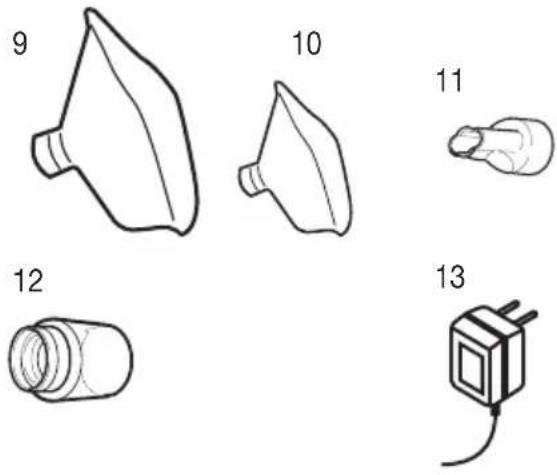
Overview of accessories
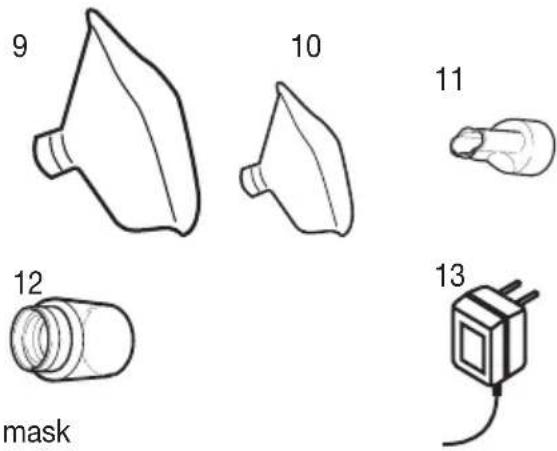
9 Adult mask
10 Child mask
11 Mouthpiece
12 Connecting piece
13 Power pack
Storage bag
6. Initial use
Before using the unit for the first time

Note
- The atomiser and accessories must be cleaned and disinfected before they are used for the first time. For information about this, see "Cleaning and disinfection" on page 16.
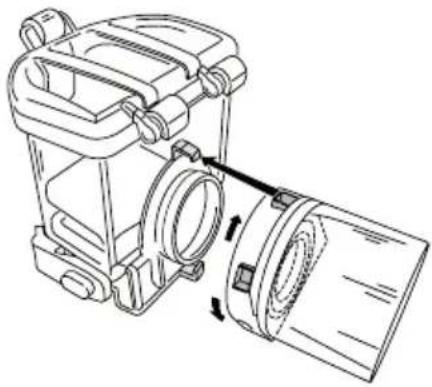
Assembly
Remove the unit from the packaging.
- If not already fitted, fit the mesh atomiser (3) onto the neck of the container (2), and lock into place by turning it clockwise until the silver-coloured contact electrodes are facing
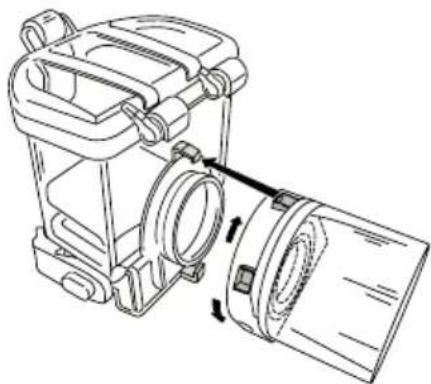
-
Place the container with fitted mesh firmly onto the housing (6) or base of the device, so that you hear it click into place on the left and right.
-
Insert the batteries.
Inserting the batteries
- To open the battery compartment (7) on the underside of the device, apply light pressure and pull the battery compartment cover in the direction of the arrows, then fold the lid up.
- Insert four batteries (type AA LR6).
- Ensure that the poles of the batteries are touching their respective poles in the device (see illustration on the bottom of the casing).
- Fold the battery compartment lid down again and push it back in place, in the opposite direction to the arrows.

Note
- When changing batteries, ensure that the medicine container is completely empty, otherwise there is a risk of leaks.
- When the orange LEDs (4) light up, replace all four batteries, because spraying becomes much less effective when the batteries are weak.
- You can operate the device for approx. 180 minutes with new alkaline batteries.
- This is sufficient for 5 - 9 uses of saline solution (depending on the dose and duration of each use).
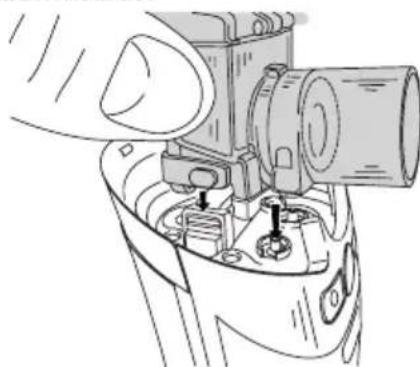
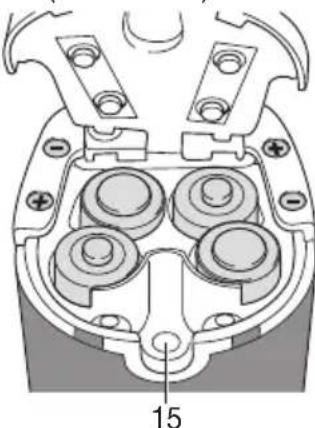
Operating the device with the power pack
Only connect the power pack (13) to the mains voltage listed on the type plate.
- Open the battery compartment lid to connect the power pack (13).
- Insert the power pack connector (8) into the connection socket provided (15), and the power pack (13) fully into a suitable socket.

Note
- Ensure that there is a socket near to where the unit will stand.
- Lay the mains cable in such a way that no one can trip over it.
- To disconnect the nebuliser from the mains after use, first switch off the device and then remove the plug from the socket.
- The batteries must not be removed from the mains part (13) during operation.
- The power pack (13) cannot be used for charging batteries.
7. Operation
1.Preparing the atomiser
- For reasons of hygiene, it is imperative that the atomiser and accompanying accessories are cleaned and disinfected after every use.
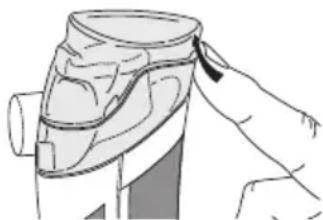
If the therapy requires that various different medicines be inhaled in succession, ensure that the atomiser is rinsed after every use. For details, see Cleaning and disinfection on page 16.
- Remove the cover (1) from the device.
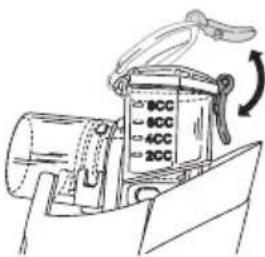
2. Filling the atomiser
- Open the medicine container (2) by opening the clip, and fill the medicine container with an isotonic saline solution or medicine. Do not overfill! The maximum recommended fill amount is 8ml .

- Use medication only on the advice of a physician, and check the appropriate inhalation duration and quantity for your needs.
- If the recommended dose of the medicine is less than 2ml , top up this quantity to at least 4ml using only isotonic saline solution. Viscous medicines may also require dilution. Always follow the instructions of your doctor.
3. Closing the atomiser
- Close the lid of the medicine container (2) and click the clip in place, and then refit the cover (1).
4. Fitting accessories
- When the required accessories (mouthpiece, adu It mask or child mask) have been attached to the device, attach the device firmly to the assembled medicine container (2).
- The adult mask and child mask must be fitted using the connecting piece (12) (adapter).
- Now place the device to your mouth and close your lips firmly around the mouthpiece. When using either of the masks, place it over the nose and mouth.
- Start the device using the on/off button (5).
- The spray mist coming from the device and the illuminated blue LEDs (4) show that the device is working perfectly.
5. Inhaling correctly
- Breathing technique
The right breathing technique is important to ensure that the particles are distributed as widely as possible into the air ways. So that they can enter the air ways and lungs, you must inhale slowly and deeply,
hold your breath briefly (5 to 10 seconds) and then exhale quickly.
- You should only use nebulisers to treat respiratory disorders if advised to do so by your doctor, who will recommend the medicine, dose and application for inhalation therapy.
- Certain medicines are only available on prescription.

Note
You should hold the device as upright as possible. Tilting it slightly will not, however, affect its function, since the container is leakproof. To ensure that the device functions correctly, make sure that you do not tilt it more than 45^ in any direction and that the medicine is in contact with the mesh while in use.

Important
Essential oils, cough medicines, solutions designed for gargling, and drops for application to the skin or for use in steam baths are wholly unsuitable for inhalation using a nebuliser. These substances are often highly viscous and can impair the correct function of the device and hence affect the effectiveness of its application in the long term.
In the case of oversensitivity of the bronchial system, medicines containing essential oils can sometimes cause acute bronchiaspasm (a sudden, cramp-like tightening of the bronchi accompanied by breathlessness). Always ask your doctor or pharmacist for advice!
6. End of inhalation
After treatment, switch off the device using the on/off button (5).
The LEDs (4) go out.
- Once the inhalant has been fully sprayed out, the device switches off automatically.
For technical reasons, a small amount may still remain in the medicine container (2).
- Do not use it.
- If applicable, disconnect the power pack (13) from the mains.
7. Clean the device
For information about this, see "Cleaning and disinfection" on page 16.
8. Cleaning and disinfection

Warning
Observe the following hygiene instructions in order to avoid any health risks.
- The nebuliser and accessories are intended for multiple use. Please note that cleaning and hygienic reconditioning requirements differ according to the different areas of application.

Note
- Do not clean the mesh or accessories with brushes or similar, as this may cause irreparable damage, and this will mean that the intended result of the treatment is no longer guaranteed.
- For additional requirements regarding the necessary hygienic preparations (hand washing, handling of medicines or inhalation solutions) in high-risk groups (e.g. cystic fibrosis patients), contact your doctor.
Cleaning
The mesh atomiser and container, as well as accessories such as mouthpiece, mask, connecting piece, etc. must be cleaned with hot water after each use. Carefully dry all components using a soft cloth. Once all components are fully dry, reassemble the atomiser and place the components in a dry, sealed container or carry out disinfection.
- Remove the mouthpiece and any accessories from the atomiser.
- Add 6 ml of clean water to the medicine container (2) and switch on the device to first remove any medicine residue from the mesh. (Please spray the entire liquid).
- Remove the batteries.
- Remove the detachable parts from the device, e.g. mesh atomiser or medicine container and cover.
Disassembly
To continue with cleaning its individual components, disassemble the atomiser (2, 3):
- Remove the medicine container (2) and mesh atomiser (3) by pressing both brackets inwards and drawing the unit out.


- Remove the mesh atomiser (3) from the medicine container (2) by turning it anticlockwise and drawing it out.
The nebuliser is later reassembled in the reverse order.
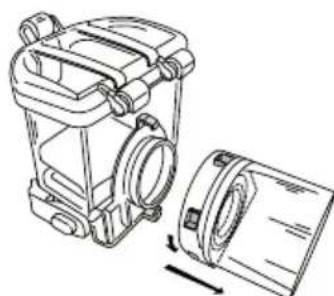

Important
Never hold the complete device under running water to clean it.
When cleaning, ensure that all residues are removed. Never use any substances that may be poisonous when in contact with the skin or mucous membranes, or when swallowed or inhaled.
If required, you can clean the device casing with a lightly moistened cloth that has been soaked in mild soapy water.
If there is any residue from medical solutions or contamination on the silver-coloured contacts of the device or atomiser, clean with a cotton bud moistened with ethyl alcohol.
- Clean the outside of the mesh and outside of the medicine container with a cotton bud dampened with ethyl alcohol.

Important
- Before you clean the unit, always switch it off, unplug it and let it cool off.
- Do not use abrasive detergents and never immerse the unit in water.
- Ensure that no water penetrates inside the unit!
- Do not put the device or accessories in a dishwasher.
- Do not touch the unit with wet hands while it is plugged in; do not allow any water to be sprayed onto the unit. The unit must be operated only when it is completely dry.
- Do not spray any liquids into the ventilation slots. Any liquid that penetrates into the unit can damage the electrical parts or other components of the nebuliser and impair the function of the device.

Important
We recommend carrying out inhalation without interruptions since the inhalant may clog the mesh atomiser during a long pause in treatment, and the effectiveness of the application may be impaired as a result.
Disinfection
Follow the steps listed below carefully in order to disinfect your atomiser and the accessories. It is advisable to disinfect the individual components after the last time they are used each day at the latest.
You can disinfect the IH 50 and its accessories in two different ways, either with ethyl alcohol or in boiling water.
First clean the atomiser and the accessories as described under "Cleaning". You can then continue with disinfection.

Important
Ensure that you do not touch the mesh (3), as this may destroy it.
Disinfection with ethyl alcohol (70-75%)
- Pour 8 ml ethyl alcohol into the medicine container (2). Close the container. Leave the alcohol in the container for at least 10 minutes.
- For better disinfection, shake the unit gently from time to time.
- Afterwards, pour the ethyl alcohol out of the container.
- Repeat this process, using water this time.
- Place the medicine container (2) in a position that allows you to apply a few drops of ethyl alcohol to the mesh (3). Allow the alcohol to work for 10 minutes.
- Finally, clean all parts again under running water.
Disinfection with boiling water
- The atomiser should be disassembled as described and, together with the mouthpiece, placed in boiling hot water for 15 minutes. Avoid the parts coming into contact with the hot base of the pot.
- You can also disinfect the atomiser and the mouthpiece with a standard vaporiser. Observe the manufacturer's instructions for use for the vaporizer when doing this.
- The atomiser must not be placed in the microwave.

Note
The face masks and air hose must not be placed in hot water!
- We recommend disinfecting the masks using a retail disinfectant.
Drying
- Carefully dry all components using a soft cloth.
- Shake the mesh atomiser (3) lightly backwards and forwards (5 to 10 times), so that the water inside the mesh is removed from the small holes.
- Place the individual components on a dry, clean and absorbent surface and allow to dry completely (for at least 4 hours).

Note
Ensure that all parts are thoroughly dried after cleaning, otherwise there is an increased risk of bacterial growth. Once all components are fully dry, reassemble the nebuliser and place the components in a dry, sealed container.
Ensure that the mesh atomiser (3) has dried completely by shaking it. Otherwise, nebulisation may not function properly after reassembling the device.
If this is the case, disassemble the mesh atomiser (3) again and shake it so that the water can escape. Once
it has been reassembled, the nebuliser should function properly again as usual.
Material resistance
- When choosing a cleaning or disinfecting agent, note the following: Only use mild cleaning or disinfectant agents in the quantities recommended by the manufacturer.
- As with all plastic components, frequent use and cleaning of the atomiser and the accessories can lead to a certain amount of wear. Over time, this can alter the aerosol properties and may eventually affect the efficiency of therapy. We therefore recommend that you replace the atomiser and accessories at least once a year.
Storage
- Do not store the nebuliser in a damp atmosphere (e.g. in the bathroom) or transport it together with damp objects.
- Store and transport the nebuliser away from direct sunlight.
Troubleshooting
| Problem/Question | Possible Cause/Remedy |
| The nebuliser produces little or no aerosol. | 1. Insufficient medicine in the atomiser. |
| 2. The atomiser is not held in an upright position. | |
| 3. The medicine used is unsuitable for spraying (e.g. too thick, viscosity must be less than 3). The medicine solution should be specified by the doctor. | |
| Output is too low. | 1. The batteries are flat. Replace the batteries or connect the power supply unit and try again. |
| 2. There are air bubbles inside the medicine container which are preventing the medicine from coming into continuous contact with the mesh. Please check and remove any air bubbles. | |
| 3. Particles on the mesh are impeding output. To remove the particles, fill the nebuliser with 2 to 3 drops of vinegar and 3 to 6 ml of water, then nebulise this solution completely. Do not inhale this spray and ensure that you clean and disinfect the medicine container afterwards (see page 16). If the output does not improve, replace the mesh. | |
| 4. The mesh is worn out. | |
| Which medicines are suitable for inhalation? | Only the doctor can advise you which medicine to use to treat your condition.Consult your doctor.With the IH 50, you can atomise medicines with a viscosity lower than 3. |
| Some inhalation solution remains in the nebuliser. | This is normal and occurs for technical reasons. Stop inhaling as soon as you can hear a marked difference in the sound made by the atomiser, or when the device switches off automatically due to insufficient inhalant. |
| What should be taken into account when using the device with infants and children? | 1. In babies, the mask should cover the mouth and nose to guarantee effective inhalation. 2. In children, the mask should also cover the mouth and nose. Nebulisation next to a sleeping person is not suitable because insufficient medicine reaches the lungs. Note: Children should only use the device with help and under supervision of an adult. Never leave a child alone with the nebuliser. |
| Inhalation with the mask takes longer. | This is for technical reasons. Less medicine is inhaled per breath through the mask holes than using the mouthpiece. The aerosol is mixed with ambient air through the holes in the mask. |
| Does each user need their own accessories? | This is absolutely necessary for hygiene reasons. |
9. Technical specifications
Dimensions
(LxWxH) 72 × 59 × 143 ~mm
Weight 238 g incl. batteries
Battery operation 4 × 1.5 V type AA, Mignon (LR6)
Volume max. 8 ml
Medicine flow approx. 0.25ml / min
Oscillation
frequency 100 kHz
Housing material ABS / TPE
Mains
connection 100-240V\~;50-60Hz;0,15A
Expected ser
vice life 3 years
| Operating conditions | Temperature: +5 °C – +40 °C Relative humidity: <80 % non-con-densing |
| Storage and transport conditions | Temperature: -10 °C – +45 °C Relative humidity: <80 % non-con-densing |
The serial number is located on the device or in the battery compartment.
Subject to technical modifications.
Wearing parts are not covered by the guarantee.

All measurements were obtained with a sodium chloride solution using a laser diffraction method.
This diagram may not be applicable for suspensions or highly viscous medicines. More information can be obtained from the relevant medicine manufacturer.
10. Replacement parts and wearing parts
Replacement parts and wearing parts are available from the corresponding listed service address under the stated material number.
| Designation | Material | REF |
| Mesh atomiser with medicine container | PC | 162.711 |
| Yearpack (Mouthpiece, Adult mask, Child mask, Mesh atomiser, Connecting piece) | PP, PVC, EVA, PET | 603.05 |

Note
If the unit is used outside of the specifications, proper function is no longer guaranteed! We reserve the right to make technical changes to improve and further develop the product.
This device and its accessories comply with European standards EN60601-1 and EN60601-1-2, as well as EN13544-1, and is subject to special safety measures in terms of electromagnetic tolerance. Note that portable and mobile RF communication equipment can affect this unit. More details can be requested from the stated Customer Service address or found at the end of the instructions for use. The unit conforms to the requirements of the European Directive for Medical Products 93/42/EEC, the MPG (German Medical units Act).
ELECTROMAGNETIC COMPATIBILITY
- The device complies with current specifications with regard to electromagnetic compatibility and is suitable for use in all premises, including those designated for private residential purposes. The radio frequency emissions of the device are extremely low and in all probability do not cause any interference with other devices in the proximity.
- It is recommended that you do not place the device on top of or close to other devices. Should you notice any interference with other electrical devices, move the unit or connect it to a different socket.
- Radio equipment may affect the operation of this device.
11. Disposal
Battery disposal
-
The empty, completely flat batteries must be disposed of through specially designated collection boxes, recycling points or electronics retailers. You are legally required to dispose of the batteries.
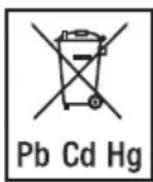
-
The codes below are printed on batteries containing harmful substances: Pb = Battery contains lead, Cd = Battery contains cadmium, Hg = Battery contains mercury.
General disposal
For environmental reasons, do not dispose of the device in the household waste at the end of its useful life. Dispose of the unit at a suit
able local collection or recycling point. Dispose of the device in accordance with EC Directive - WEEE (Waste Electrical and Electronic Equipment). If you have any questions, please contact the local authorities responsible for waste disposal.
FRANÇAIS
Sommaire
Cher cliente, cher client,
Isletime almadan once

Dikkat
PeKOMeHnyETc3aMeHHTb paCnblntelb n dpyrne npHaJIeXHOCTn He No3dHee, Yem Upe3 rOJ.
BHHMaHHe YKa3bIBaet Ha BO3MOxHbIe NOBpeXKeHnIa

Yka3aHne OTMeHaET BaXHyIO INOpMaunIO.
Ha ynapokBke n Ha 3aBODCKoTabnUKe npbopa n npHnAdJexKHOCTe NcNoJIb3yOTcN cJeDyUOuNe CmBOJIbl.
Ipeed Haayalom IcnoJb3ObaHna

BhimaHne
- Pered nCnoJb3ObaHnem npu6opa HxKHO ydaJIITb BeCb ynaKOBOUHbI MaTePnA1.
- 06eperaTe npnbop ot nblln, rpr3n n BnaR.
He nCnoB3yIe np6Op B ycNoBnx CunbHoi 3a- nblnEHHOCTn. - Pered nCpOJIb3OBAHnEM npocJeIte, UTo6bl npI6bp
- IN pRHaJNeKHOCTN IMeJI KOMHaTHyO TempePaTyP.
Bcnyuae HncnpabHocT npnbopa nnn HapyueHn B pa6oTe HEmeJeHNO BbIKJIOHTe np6Op. - IV3ROTOBNTeJIb He HecET OTBetCTBeHHOCTb 3a NOBpeKdEHN, BO3HKnUHe No npuHHe HnPaBnIb-HORo NcNoJIb3OBaHN.

O6paueHne c 3JeMeHTamn nITaHna
- Pn nonaHnn XnkOCTn n3 aKKymyIaTopa Ha KOnKy nn B rna Heo6xOIMo npomblc COOTBETCTBy
Iounn yuaTOK 60JIbIIM KOJIuYeCTBOM BOIbI N O6paTntbcr K BpaCy.
- OnachocTb nporlaTbHaHnmeJiknx yacTe! MaJIeHbKne Detn MOrTy nporIOTntb 6aTapeNn noDaBNTbcn NMI. PoTOMy 6aTapeNk Heo6xOIMO XpaHNTb B HeoCTynHom dJeTeN MeCte!
- 06paaainTe BnImaHne Ha o6o3HaueHne nolpHoCTn: pIoc (+) u MmHyc (-).
- Ecnn 6batapeiKa noteKna, ounctnte otdeJeHne dnn 6batapeek cyxoi caTkoi, naB 3aunTHbIe nepaTkn.
3aunuainTe 6aTapeKn ot ype3MepHoro Bo3deiCTBna Tnla. - OnachocbB3pbBa! He 6pocaTe 6aTapeKn B orOhb.
He 3apkaTe He 3aMbkaTe 6aTapeKn HakoPOTko. - Ecni npu6op dnteBhoe Bpemr He nCnoJb3yeTcra, n3BneKeNTe n3 Hero 6atapeiKn.
- IcnoIb3yIte 6aTapeiKu TOnbKO Ondoro Tnna nn paBHOeHHbIX TINOB.
- 3aMeHnTe Bce 6aTapeKn cpa3y.
He nCnoJIb3yIte nepe3apJkaemble aKKymJrTopbl!
He pa36npaTe, He OTKpbBaIte n He pa36nBaIte 6aTapeiKn.

PpeoctepexKeHne
BbTeKUne HnN NOBpeXdEHHbIe 6aTapeKu MOryT npu COnpNKoCHOBENu C KOKeB Bbl3BaTb XmMueCKNe OxOr.ДЯ n3bTaTHa 6aTapeek uCNoJb3ynte noXOJaIe 3aunTHbIe nepuaTK.
PemOHr

Yka3aHne
- Hn B Koem Cnyuae He OTKpbIbAitne Hr He pEmOHtpyuTe np6Op, TaK KaK B 3TOM Cnyuae He MoKeT 6bITb rapaHTnpoBaHO erO daNbHeiWee HaJIeXaUee yHKcIOHnPOBaHne. Pn Heco6JIIODeHN 3Tnx ycIOBn rapaHTn TepaET CBOIO cnly.
- Пибор He Tpe6yeT TexHnueeCKOrO 06cJyXnBaHnA.
-Дя поведеня ремонтных paBOT obpaTntecbВ cepвисную слжбу Или К уралночehHomу длелу.
3aunTa OT BbITEKaHnA
Pn 3aONHeHn EMKoCTn IJIeKapCTBeHHbIX CpeIcTB CneIte 3a TeM, YTO6bl O6bEM He IpeBbl-1JAMAKCImaJIbHyIO OTMeTKy 8 Ml. PekOMeHdyEmbl O6bEM 2-8 Ml.
Bo Bpem npimHeHn npnbopa Bbl moKeTe haknoHrTo ERO BO BCex HnpaBneHnx Do MaKcImMaJbHoRo yra 45° 6e3 yxudHnKaYeCTBa paCbIeHn I, COOTBeTCTBeHHo, pe3yIbTaTata leeHn. OHaKO paCbIeHn IpOUCXODIT TOJIbKO Do TEX nop, noka paCbIaEMoe BeUeCTBO copnKacaetc C cetKo. EcIn 3TOrO He npocXODIT, paCbIeHne ABTomTuyeckn PpeKpauaetc.
Ioxaanyuicta, cTapaaiTecb BcE Xe depkaTb npnbop nOB3MOxHocTu BepTuKaJIbHo.
ABTomatueckoe OTKJIOUeHne
Pnpbop nmeet fynkunabTOMaTHueCKOROTKIOHeH.N. EcnI JekapCTBeHHoro CpeCTBa Nn JxNkoCTN OCTaIOcB Oeyh MaNo, np6op aBTOMaTHueCKN BblKIOUOaETcBO N36eKaHne NOBpeXJDeHn CeTKN.
Helenb3nCnoB3OBaTbpnp6Op CnyctOJ EMKocTbIO IJIeKapCTBeHHbIX CpeIcTB!Pnp6Op ABTomTuYeCKN BbIKluOaETcA,ecPi pacblIReMoe BeIeCTBO 60JIbWe He cOnpNKacaETcC cetKoI.
TopaHKeBbIM CBETOM:3aMeHITb 6atapeKu
5.KhonkaBKJ/BbIKJ
6. Kopnyc
7. Otcek Дябата
8.CoeHnHtBnHbI nTeKepe

O630 npnHaJneKHocTei
- Macka Дя Взрочьх
- Macka dIra deTei
- MyHdWtYk
- CoeHHntbHbI 3JeMeHT
13.БLOKПИТAHNIA
YexonIyXpaHeHn
6. Побrotовka к paбote
Peped npBbIM npMHeHem
iYka3aHne
- Ipeed nepBbIM npimeHeHem pacbIInTeJb n npnHaIeXHOCTn Heo6xOdmo NOcHCTNb N De3uHcNcIPOBaTb. CM. «YncTk a N de3uHcEkua» Ha cTpaHnue 64.
MoHTax
BbInbTe np6op n3 ynaKOBKn.
Ecni cetyaTbIpaTbnITeJIb (3) C MeWMem6paHOH HeyCTaHOBHeN 3apahee, NOcOeINHTe eRO KHaCaJKe KOHTeINHepa (2) n3aФNKcpyu-Te, NOBEPHyB n
aCOBcTpeKe, noka cepebpcTbe KOHTaKTHbIe 3JIeKTPOdbI He 6ydt HappaBHeHbI BHN3.
- YCTAHOBITE EMKOCTb
ДлЯ Лекар-CTBEHHbIXCpeDCTB BmecTeCCeTcA TbIMpac-NblNTeIeMHaKOpNyc (6)ИОCHOBAHnE npIbopa TaKIM Obpa3OM,TO6bIOH CJIeBa nCnpaba 3aФИСКИСИРOBALCSO CJ

- BCTaBbTe 6aTapeKn.
YctaHOBka 6aTaapeek
- OTKpOIne OTcEK dIa 6aTapeek (7) Ha HnXHei CToPope npnbopa: IINr 3TOr0 Bam HyxHo cIerKa HaDaBnTB N ODHObPeMeHNO NOTaHyTb B HApBaJIeHmN CImBOJIOB CTpeIKN Ha KpbIuKE OTcEka.
BCTaBbTe YeTbIpe 6aTapeuKn (TIn AA LR6).
- Ośpaṭniete BHNMaHne Ha npaBnIbHoCTb pacnoIIOXeHn IOnIOcOB 6a-Tapeek (cM. pncyHOK ha dHnIe KOpnyca).
3akpoTe KpbuKy OTeKa dJa 6aTaapeek, a 3aTeM 3aФNKcUpyTe ee, NOBepHyB Do yNopa npOTnB HAnpaBHeHnY, Yka3aHHOrO CTpeKNKaMn.

Yka3aHne
- Pn 3aMeHe 6aTapeek CneIte 3a TeM, YTO6bI EMKoCTb DnIeKapCTBeHHbIX CpeIcTB 6blNa NOLHOCTbIO NyCTa, T. K. B IpOTUBHOM Cnyae BO3HnKaET ONaCHOCTb NOBJIeHn OTDeIbHbIX KaneJIb.
- EcJn KOHTpOJIbHbIe INHdNKaTOpbl (4) 3aropaOTcApaHXeBbIM CBeTom, CJeIyET 3aMeHITb BCE YeTbIpe 6aTaapeKu, TAK KAK 3ΦΦeKTUBHOCTb paCblJeHnra Co Cna6bIM6aTaapeKamN CInbHO yMeHbShaetcR.
C HOBbIMU ⅢeIOnHbIMN 6aTapeKamn npnbop pa-6oTaET npm.180 MNHyT.
-3TOrO doCTaToHNo 5-9 nHraJauCn CnCnB3oBaHnEM paCTbopa NOBaPeHHoCOn (B 3aBnCMoCTN OT DO3nPOBKn IN PpOdoJIxHTeJIbHOCTN NcPONb3OBaHnA).
3KcNlyataaC 6JOKOM NITaHn
Блok nHTaHn (13) MoXeT NOdCoEINrTbCra TOnbKO K CETn C yKa3aHHbIM Ha 3aBODcKo T6JIuHKe HAnpRxKeHNEM.
- Дя подкючени 6лoka птань (13) оTKpoite Крblшky OTсЕКа Дя 6atapeek.
BOTKHNTe coeHNHTeJIbHbI WTeKep 6JOKa NITAHN (8) B npeducmOTpeHHoe rHe3do (15) nNoDKIIOHTe 6JOK NITAHN (13) K NOxOJauei po3ETke.

Yka3aHne
- 06paTte BnMaHne Ha To, qTo6bI po3eTKa HaxoJnJaCb B6In3m MeCTa yCTaHOBKn np6opa.
- YIIOXHTe CeTeBOH UHP TaKIM O6pa3OM, YTO6bl 06 HrO HeBO3MOxHO 6bINO CNOTKHyTbcra.
- OTKIIOUeHn INrAJIaTopa OT cETn NocJe INrAJIaCm ChauJaBa BbIKIOuHTe Pnp6Op n 3aTeM BbIHbTe BUNKy n3 PO3eTKN.
BoBpempa6oTbIOT6noka nHTaHn8(13)HeNb3a n3BJIeKaTb 6aTapeKn.
-Блokпитаня(13)нельзиспобьотьдяЗардкakkymлторов.
7.Управleness
1. Podrotobka pacnbilnteJra
- IIO TINHEHNUeCKM COO6paXeHNr HEO6xOaIMoNocIe KaXDoI PPOeUpybI YnCTITb N De3nHcNUPOBaTb PaCbIInTeJIbN pINHaJnxHocT.N.EcNI npINpOBeHm Jue6

HONIHraIyIINCNOJIb3yOTcN OOepeIHNecKOB Ko pa3nUHbIX JekapCTBeHHbIX CpeCTB,Heo6XoIMo nponolackBaTpaCblntelN oocLe KaKDoR npiMeHnIOI CTpyE TEPNoBODOnpOBoDHO BObI. CM. «YnctKa n Ie3nHfekuIaHa cTpaHnce 64.
- CHIMITE c npi6opa KpbliKy (1).
2.3aNoJIHeHne paCnblInTeJra
- OTKpoIte EMKoCTb IJRA JekapCTBeHHbIX CpeIcTB (2): 1JRA 3TOrO OTKpoIte 3axm, a 3aTeM 3aONHInTe EMKoCTb N3OTOHueCKnPacTBOpOM NOBapeHHoN CoN INn HeNoCpeIcTBeHHO JekapCTBeHHbIM CpeIcTBOM. He dony
ckaiTe nepenolHeHn! MakcimalbHoe peKOMeHdyemoe KOJIueCTBO 3aIOnJIHOUeRO BeUeCTBa COCTABJET 8 Ml!
-ПрименяпгларсбгьдсрсдвТОькпн
-НаЗнayehиВрачa,Co6ЛюддуАлььнь
-Уka3aHЯ NO pOdoJXHTeJIbHOCTN INHДИБИdYALьнь
-КоLynchecTBy JIeKapcTBeHHORo CpeDCTBa!
- EcJn Ha3HaueHHe KOJIyEcTBo JIeKApCTBeHHOrO cpeCTBa COCTaBIAET MeHee 2 Ml, IOBeIte DaHbI ObEhem He MeHee Yem Do 4 Ml, IcNoIb3yJ
TolbKO n30ToHnueckn pactBop NOBaepHHo CoJI. B83Kne IekapCTBeHHbIe cpeIcTBa TaKKe Tpe6yIoT pa36abHeHn. Co6JIouaTe COOTBeTcTByIOuNe yKa-3aHnBpa4a.
3. 3aKpbBaHne pacbIInTeJra
- 3aKpOte KpbIuKy EMKoCTn IJRA JIeKapCTBeHHbIX cpeIcTB (2)ИЗаФИКсUpyIte 3aXIMOM,ОЯТь Ha-DeHbTe KpbIuKy npu6opa (1).
cpeCTBa OK.0,25 Ml/MnH
Yactota kone6aHn 100Kt
MaTePnAknOpnycaABS/TPE
IopKJIIOueHHe K cetn 100-240 B-;50-60 rU;0,15A
PpeJnoJaraembI
cpoK cnjxkbI 3 roda
YcNoBnTeMnepaTypa: +5 10 +40^
ФКСПЛУАТAUИ OTHOCINTEЛьнаг ВлжHOCТБ BO3-уxa: <80%, He KoHdEHCnpo-BaHHbI
YcnoBna xpaHnna Tempepatya: ot-10do+45°C
TpaHCnOpTnPOBKn OTHOCnTeJIbHaN BJIaXHoCTb BO3- Dyxa: < 80% ,He KOHdEHCnpoBaHHbl
CepinHbI HOMep HaxoNTcHa npnbope nIn B ot- deJeHHn dJa 6aTaapeek.
OCTaBJIeM 3a co6oI npaBO Ha BHeceHne TexHnuecknx I3MeHeHn.
Ha n3HaunBaIOUneCyaTeaIg rapaHTnHe pacnpoctpaHreTc.

I3MepeHnI npOn3BODnIcB C nCNoJIb3OBAHNem paCTBOPa XLOpUda HaTpNIA MeToDOM Ja3ePHoI dIΦpaKuN.
Bo3MOxHo, dnaarpammy He cIeNyET npImeHb B OTHoWeHn CycneH3n nn OueHb Bra3Knx IekapCTBeHHbIX cpeiCTB. Bolee noDpo6HyIO nHΦopMaunio NO 3TOMy BOpocy MOxHO nOlyuHTb y n3rOToBnteJRAkapCTBeHHoro cpeiCTBa.
10. 3aIacHbIe qactn I DeTaJI, NOdBepXeHHbIe 6bICTpOMy n3-Hocy
3aIacHbIe YAcTNI DeTaII, NIOBBePKeHHbIe 6bICTpOMy I3HOCy, MOXHO pNIOBpeCTN B COOT BEcTBByUOxNCEPBUCHBIX CEHTpax, yKa3aB HOMep DeTaII IN KaTAlore.
Table 1
For all ME EQUIPMENT and ME SYSTEMS
Table 2
| Guidance and manufacturer's declaration - electromagnetic emissions | ||
| The IH 50 is intended for use in the electromagnetic environment specified below.The customer or the user of the IH 50 should assure that it is used in such an environment. | ||
| Emissions Compliance Electromagnetic environment - guidance | ||
| RF emissions CISPR 11 | Group 1 | The IH 50 uses RF energy only for its internal function. Therefore, its RF emissi-ons are very low and are not likely to cause any interference in nearby electronic equipment. |
| RF emissions CISPR 11 | Class B | The IH 50 is suitable for use in all establishments, including domestic establish-ments and those directly connected to the public low-voltage power supply net-work that supplies buildings used for domestic purposes. |
| Harmonic emissions IEC 61000-3-2 Class C | ||
| Voltage fluctuations/flicker emissions IEC 61000-3-3 | Complies | |
For all ME EQUIPMENT and ME SYSTEMS
Table 3
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The IH 50 is intended for use in the electromagnetic environment specified below.The customer or the user of the IH 50 should assure that it is used in such an environment. | |||
| Immunity test | EN 60601 test level | Compliance level | Electromagnetic environment - guidance |
| Electrostatic discharge (ESD)IEC 61000-4-2 | ± 6 kV contact± 8 kV air | ± 6 kV contact± 8 kV air | Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30%. |
| Electrical fast transient/burstIEC 61000-4-4 | ± 2 kV for power supply lines ± | 2 kV for power supply lines | Mains power quality should be that of a typic -cal commercial or hospital environment. |
| SurgeIEC 61000-4-5 | ± 1 kV line(s) and neutral ± 1 kV | line(s) and neutral | Mains power quality should be that of a typic -cal commercial or hospital environment. |
| Voltage dips, short interrupt -tions and voltage variations on power supply input linesIEC 61000-4-11 | <5% U_T (>95 % dip in U_T)for 0.5 cycle40% U_T (60 % dip in U_T)for 5 cycles70% U_T (30 % dip in U_T)for 25 cycles<5% U_T (>95 % dip in U_T)for 5 s | <5% U_T (>95 % dip in U_T)for 0.5 cycle40% U_T (60 % dip in U_T)for 5 cycles70% U_T (30 % dip in U_T)for 25 cycles<5% U_T (>95 % dip in U_T)for 5 s | Mains power quality should be that of a typic -cal commercial or hospital environment.If the user of the IH 50 requires continued operation during power mains interruptions, it is recommended that the IH 50 be powe -red from an uninterruptible power supply or a battery. |
| Power frequency(50/60 Hz)magnetic fieldIEC 61000-4-8 | 3 A/m | Not applicable | Not applicable |
| NOTE: U_T is the a.c. mains voltage prior to application of the test level. | |||
For ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The IH 50 is intended for use in the electromagnetic environment specified below.The customer or the user of the IH 50 should assure that it is used in such an environment. | |||
| Immunity test | IEC 60601test level | Compliancelevel | Electromagnetic environment - guidance |
| Conducted RFIEC 61000-4-6Radiated RFIEC 61000-4-3 | \( 3V_{rms} \)150 kHz to80 MHz3V/m80 MHz to2,5 GHz | \( 3V_{rms} \)3V/m | Portable and mobile RF communications equipment should be used no closer to any part of the IH 50, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter.Recommended separation distance:\( d = 1,2\sqrt{P} \)\( d = 1,2\sqrt{P} \) 80 MHz to 800 MHz\( d = 2,3\sqrt{P} \) 800 MHz to 2,5 GHzWhere \( P \) is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and \( d \) is the recommended separation distance in metres (m).Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey,\(^a\)should be less than the compliance level in each frequency range.\(^b\)Interference may occur in the vicinity of equipment marked with the following symbol: |
| NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies.NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||
a Field strengths from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the IH 50 is used exceeds the applicable RF compliance level above, the IH 50 should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as re-orienting or relocating the IH 50. b Over the frequency range 150kHz to 80MHz , field strengths should be less than 3V / m .
Table 4 For ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
| Recommended separation distances between portable and mobile RF communications equipment and the IH 50 | |||
| The IH 50 is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the IH 50 can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and the IH 50 as recommended below, according to the maximum output power of the communications equipment. | |||
| Rated maximum output power of transmitter(W) | Separation distance according to frequency of transmitter(m) | ||
| 150 kHz to 80 MHzd=1,2√P | 80 MHz to 800 MHzd=1,2√P | 800 MHz to 2,5 GHzd=2,3√P | |
| 0,01 0,12 0,12 0,23 | |||
| 0,1 0,38 0,38 0,73 | |||
| 1 1,2 1,2 2,3 | |||
| 10 3,8 3,8 7,3 | |||
| 100 12 12 23 | |||
| For transmitters rated at a maximum output power not listed above, the recommended separation distance d in meters (m) can be determined using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer.NOTE 1 At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies.NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||