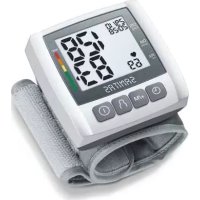
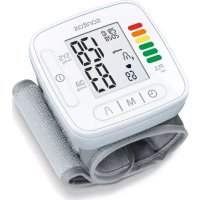
SBM 54 - Blood pressure monitor SANITAS - Free user manual and instructions
Find the device manual for free SBM 54 SANITAS in PDF.

User questions about SBM 54 SANITAS
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Blood pressure monitor in PDF format for free! Find your manual SBM 54 - SANITAS and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. SBM 54 by SANITAS.
USER MANUAL SBM 54 SANITAS
natural_image
Illustration of an open book with a folded corner and horizontal lines, no text or symbols presentGB Unfold page 3 before reading the instructions for use.
natural_image
Three technical line drawings of cylindrical devices with internal components, labeled A (no text or symbols on the devices themselves)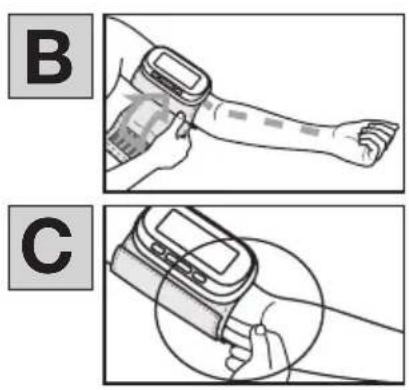
text_image
B C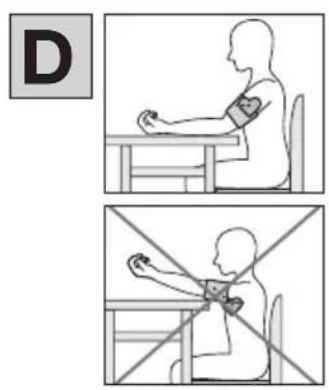
natural_image
Illustration showing two different positions of a person in arm and torso, with no visible text or symbols.

DEUTSCH

1 Quelle: WHO, 1999 (World Health Organization)
2 Quelle: National Health Service, 2023
Read these instructions for use carefully. Observe the warnings and safety notes. Keep these instructions for use for future reference. Make the instructions for use accessible to other users. If the device is passed on, provide the instructions for use to the next user as well.
CONTENTS
- Included in delivery 19
- Signs and symbols....19
- Intended purpose 21
- Warnings and safety notes....22
- Device description....24
- Usage 25
6.1 Initial use....25
6.2 Before the blood pressure measurement .....25
6.3 Taking a blood pressure measurement....26
6.4 Evaluating the results....27
6.5 Displaying and deleting measured values .....28 - Cleaning and maintenance....29
- Troubleshooting....29
- Disposal 29
- Technical specifications 30
- Guarantee/service 31
1. INCLUDED IN DELIVERY
Check that the exterior of the cardboard delivery packaging is intact and make sure that all contents are included in the delivery. Before use, ensure that there is no visible damage to the device and accessories and/or replacement parts, and that all packaging material has been removed. If you have any doubts, do not use the device and contact your retailer or the specified Customer Services address.
- Upper arm blood pressure monitor including upper arm cuff (22–42 cm)
- Batteries, see chapter "Technical specifications"
- Blood pressure pass
- Instructions for use
2. SIGNS AND SYMBOLS
The following symbols are used on the device, in these instructions for use, on the packaging and on the type plate for the device:

WARNING
Indicates a potentially impending danger. If it is not avoided, death or serious injury will occur.
| ⚠️ CAUTIONIndicates a potentially impending danger. If it is not avoided,slight or minor injuries may occur. | |
| NOTICEIndicates a potentially harmful situation. If it is not avoided,the system or something in its vicinity may be damaged. | |
| i | Product informationNote on important information |
| Observe the instructionsRead the instructions before starting work and/oroperating devices or machines | |
| The electronic device must not be disposed of withhousehold waste | |
| Do not dispose of batteries containing harmful sub-stances with household waste | |
| Manufacturer | |
| Date of manufacture | |
| CE labellingThis product satisfies the requirements of the applicableEuropean and national directives. | |
| Marking to identify the packaging material.A = material abbreviation, B = material number:1-7 = plastics, 20-22 = paper and cardboard | |
| Separate the product and packaging elements and dispose of them in accordance with local regulations. | |
| Protection class II device | |
| IP22 | Device protected against foreign objects ≥ 12.5 mm and against water dripping at an angle |
| Direct current | |
| Battery | |
| UDI | Unique device identifier (UDI)Identifier for unique product identification |
| LOT | Batch designation |
| REF | Item number |
| SN | Serial number |
| EU REP | Authorized representative in the European Community |
| CH REP | Swiss authorised representative |
| MD | Medical device |
| Type number | |
| Type BF applied part | |
| Temperature range |
| Humidity range |
| Atmospheric pressure limitation |
| Importer |
3. INTENDEDPURPOSE
Intended Use
The blood pressure monitor (hereinafter, device) is intended for the fully automatic, non-invasive measurement of arterial blood pressure and pulse values on the upper arm.
It is designed for self-measurement by adults in a domestic environment.
Intended Users
The blood pressure measurement is suitable for adult users whose upper arm circumference is within the range printed on the cuff.
Clinical benefits
The user can record their blood pressure and pulse values quickly and easily using the device. The recorded values are classified according to internationally applicable guidelines and evaluated graphically. Furthermore, the device can detect any irregular heart beats that occur during measurement and inform the user via a symbol in the display. The device saves the recorded measurements and can also output average values of previous measurements. The recorded data can provide healthcare service providers with support during the diagnosis and treatment of blood
pressure problems, and therefore it plays a part in the long-term monitoring of the user's health.
Indications
In the event of hypertension or hypotension, the user can independently monitor their blood pressure and pulse values at home. However, the user does not need to be suffering from hypertension or arrhythmia in order to use the device.
Contraindications
WARNING
- Do not use the blood pressure monitor on newborns, children or pets.
- This device is not approved for use with children weighing less than 10 kg.
- Persons with reduced physical, sensory or mental capabilities should be supervised by a person responsible for their safety and receive instructions from that person on how to use the device.
- Do not use the cuff on people who have undergone a mastectomy or who have had their lymph nodes removed.
- Do not place the cuff over wounds as this may cause further injury.
- Make sure that the cuff is not placed on an arm whose arteries or veins are undergoing medical treatment, e.g. intravascular access or intravascular therapy, or an arteriovenous (AV) shunt.
- Do not use the device on people with allergies or sensitive skin.
Undesirable side effects
- Skin irritation
- Negative influence on blood circulation
4. WARNINGS AND SAFETY NOTES
General warnings

WARNING
- The measurements you take are for your information only – they are not a substitute for a medical examination! Discuss your measured values with your doctor and never make your own medical decisions based on them (e.g. regarding medicine doses).
- The device is only intended for the purpose described in these instructions for use. The manufacturer is not liable for damage resulting from improper or incorrect use.
- Using the blood pressure monitor outside your home environment or while on the move (e.g. while travelling in a car, ambulance or helicopter, or while undertaking physical activity such as playing sport) can influence the measurement accuracy and cause incorrect measurements.
- Cardiovascular diseases may lead to incorrect measurements or have a detrimental effect on measurement accuracy.
- If you have any of the following conditions, it is essential you consult your doctor before using the device: Cardiac arrhythmia, circulatory disorders, diabetes, pregnancy, pre-eclampsia, hypotension, chills, shivering.
-
Do not use the device at the same time as other medical electrical devices (ME equipment). This could cause the measuring device to malfunction and/or an inaccurate measurement.
-
Do not use the device outside of the specified storage and operating conditions. This could lead to incorrect measurements.
- Only use the cuffs included in delivery or described in these instructions for use with the device. Using a different cuff may lead to inaccurate measurements.
- Note that when inflating the cuff, the functions of the limb affected may be impaired.
- Do not perform measurements more frequently than necessary. Due to the restriction of blood flow, some bruising may occur.
- Blood circulation must not be stopped for an unnecessarily long time during the blood pressure measurement. If the device malfunctions, remove the cuff from the arm.
- Place the cuff on the upper arm only. Do not place the cuff on other parts of the body.
- Small parts may present a choking hazard for small children if swallowed. They should therefore always be supervised
- Keep packaging material away from children. There is a risk of suffocation.
- Keep away from children, pets and pests.
- The blood pressure monitor must not be used together with a high-frequency surgical unit.
- Do not use the device with a defibrillator.
- Do not use the device during an MRI scan.
- Do not expose the device to static electricity. Always make sure that you are free of static electricity before you use the device.
- Do not place the device in autoclaves or gas sterilisation devices.
-
Do not drop, step on or shake the device.
-
Do not disassemble the device as this may cause damage, faults and malfunctions.
- Do not modify the device.
- To rule out a difference between sides, the measurement should initially be taken on both arms.
- Never operate the device during maintenance work. Maintenance work includes maintenance, inspection and repair.
General precautions
CAUTION
- The blood pressure monitor is made from precision and electronic components. The accuracy of the measurements and service life of the device depend on its careful handling.
- Protect the device from impacts, moisture, dirt, drastic changes in temperature and direct sunlight.
- Ensure the device is at room temperature before taking a measurement. If the measuring device has been stored close to the maximum or minimum storage and transport temperatures and is placed in an environment with a temperature of 20 °C, it is recommended that you wait approx. 2 hours before using the measuring device.
- Do not use the device in the vicinity of strong electromagnetic fields and keep it away from radio systems or mobile telephones.
- We recommend removing the batteries if the device is not going to be used for a prolonged period of time.
Notes on handling batteries
WARNING
- Risk of explosion! Risk of fire! Failure to comply with the following points can result in personal injury or cause
overheating, leakage, venting, breakage, explosion, or fire on the battery.
- This device contains non-rechargeable batteries which must not be charged.
- Do not throw batteries into a fire.
- Never charge, forcibly discharge, heat, disassemble, open, crush, deform, encapsulate, or modify batteries.
- Never short-circuit batteries or battery compartment contacts.
- Protect the batteries from direct sunlight, rain, heat, and water.
- Exposure of batteries to an environment with extremely high temperatures or an extremely low air pressure may result in explosion or leakage of flammable liquids and gases.
- Dispose of defective and discharged batteries immediately and properly (see chapter on disposal).
- Do not use modified or damaged batteries.
- Always select the correct battery type.
- Always insert the batteries correctly, taking into account the polarity (+ / -).
- Never mix batteries of different manufacturers, capacities (new and used), size, or type within a device.
- If a battery has leaked, put on protective gloves and clean the battery compartment with a dry cloth.
- If fluid from a battery comes into contact with your skin or eyes, wash the affected areas with water and seek medical assistance.
- Choking hazard! Keep batteries out of the reach of children. Seek medical attention immediately if swallowed. Swallowing them may cause burns, severe internal injuries, and death.
- Never allow children to replace batteries without adult supervision.

CAUTION
- Store batteries in a well-ventilated, dry, and cool place in a non-conductive container in which the batteries cannot be short-circuited to each other or by other metal objects.
- Keep batteries clean and dry.
- Keep batteries away from water.
- If the device is not going to be used for a long period of time, remove the batteries from the battery compartment.
NOTICE
- Do not use rechargeable batteries.
Notes on electromagnetic compatibility

CAUTION
- The device is suitable for use in all environments listed in these instructions for use, including domestic environments.
- The device may not be fully usable in the presence of electromagnetic disturbances. This could result in issues such as error messages or the failure of the display/device.
- Avoid using this device directly next to other devices or stacked on top of other devices, as this could lead to faulty operation. If, however, it is necessary to use the device in the manner stated, this device as well as the other devices must be monitored to ensure they are working properly.
-
The use of accessories and/or replacement parts other than those specified or provided by the manufacturer of this device could lead to an increase in electromagnetic emissions or a decrease in the device's electromagnetic immunity; this can result in faulty operation.
-
Keep portable RF communication devices (including peripheral equipment, such as antenna cables or external antennas) at least 30 cm away from all device parts, including all cables included in delivery.
- Failure to comply with the above can impair the performance of the device.
5. DEVICE DESCRIPTION
The associated drawings are shown on page 3.
1 Display
3 Risk indicator
5 START/STOP button①
7 Upper arm cuff
2 Battery compartment lid
4 Memory button
6 Function button >
Information on the display
8 Time and date
10 Memory display: Average value AVG Morning evening
12 User memory 12
14 Measured pulse value
16 Cuff fit check OK
18 Risk indicator
9 Memory space number
11 Low battery indicator


13 Cardiac arrhythmia symbol / pulse symbol
15 Diastolic pressure
17 Systolic pressure
6. USAGE
6.1 Initial use
Inserting the batteries
- Remove the battery compartment cover on the back of the device A.
- Insert the batteries (see chapter “Technical specifications”). Insert the batteries, making sure the polarity is correct according to the label A.
- Close the battery compartment cover.
If the ☐ symbol is displayed and does not disappear, measurement is no longer possible. Replace all the batteries. Once the batteries have been removed from the device, the date and time must be set again. Any saved measured values are retained.
Adjusting the settings
Make sure that the device's settings have been set correctly so you can make full use of all its functions. Otherwise you will not be able to save your measured values with the date and time and access them later.
There are two different ways to access the settings menu:
- Before initial use and after each time you replace the battery: When inserting batteries into the device, you will be taken to the relevant menu automatically.
- If the batteries have already been inserted: With the device switched on, press and hold 📄 for approximately 5 seconds.
Set these settings in the order shown below:

Press Ⓐ to confirm your selection each time.
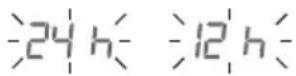
Time format
Time format flashes:
- Press > to select the time format.
Date
The year flashes:
- Press > to select the year.
The month flashes:
- Press > to select the month.
The day flashes:
- Press > to select the day.
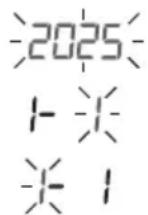
If the time format is set to the 12-hour format, the order in which the day and month are displayed is reversed.
Time
The hour flashes:
- Press > to select the hour.
The minutes flash:
- Press > to select the minutes.

6.2 Before the blood pressure measurement
General rules when measuring your own blood pressure
- In order to generate an informative profile of changes in your blood pressure that can be used for comparisons, you should measure your blood pressure regularly and always at the same time of day. Measure your blood pressure twice a day: once in the morning after getting up and once in the evening.
- Always perform the measurement when you are sufficiently physically rested. Avoid taking measurements at stressful times.
- Do not take a measurement within 30 minutes of eating, drinking, smoking or exercising.
- Before the initial blood pressure measurement, make sure always to rest for about 5 minutes.
- If you want to take several measurements in succession, always make sure that you leave 1 minute between each measurement.
- Repeat the measurement if you have doubts about the measured value.
Attaching the cuff
You can measure your blood pressure on either arm. Some deviations between the values in the right and left arm are perfectly normal. Always perform the measurement on the arm with the higher blood pressure values. Consult your doctor about this before starting self-measurement.
• Always measure your blood pressure on the same arm.
- The blood pressure measurement is suitable for adult users with an upper arm circumference within the range printed on the cuff (22 – 42 cm).
- Expose your upper arm. The circulation of the arm must not be hindered by tight clothing or similar.
- The cuff must be placed on the upper arm so that the bottom edge is positioned 2-3 cm above the elbow and over the artery [B].
The cuff should be fastened so that two fingers fit under the cuff when it is closed C.
Adopting the correct posture
- Sit in a comfortable upright position when taking the blood pressure measurement. Lean back so that your back is supported.
- Place your arm on a surface D.
- Place your feet flat on the ground next to one another.
- The cuff must be level with your heart.
- Stay as still as possible during the measurement and do not talk.
Selecting the user
This device has 2 user options with 120 memory spaces each, so you can separately store measurements from 2 different people. If multiple people are using the device, make sure that the correct user has been selected before each measurement:
- Use > on the start screen to select the desired user.
6.3 Taking a blood pressure measurement
You must ensure the following: Cuff applied, device switched on, user selected.
Measurement
- Press Ⓐ. The cuff inflates itself automatically. The measurement process starts. ♥ displayed as soon as a pulse is detected.
To cancel the measurement, press Ⓞ - The systolic pressure, diastolic pressure and pulse measurements are displayed.
E_r is displayed if the measurement could not be performed properly. In this case, please refer to the “Troubleshooting” section.
If necessary, re-attach the cuff after 1 minute.
The device switches off automatically after approx. 30 seconds. The value is saved to the selected or most recently used user.
6.4 Evaluating the results
General information about blood pressure
- Blood pressure is the force with which the bloodstream presses against the arterial walls. Arterial blood pressure constantly changes in the course of a cardiac cycle.
• Blood pressure is always stated in the form of two values:
- The highest pressure is the systolic blood pressure. This occurs when the heart muscle contracts and blood is pumped into the blood vessels.
- The lowest pressure is the diastolic blood pressure. This occurs when the heart muscle has completely relaxed again and the heart is filling with blood.
- Fluctuations in blood pressure are normal. Even during repeat measurements, there may be considerable differences between the measured values. One-off or irregular measurements therefore do not provide reliable information about the actual blood pressure. Reliable assessment is only possible when you perform the measurement regularly under comparable conditions.
Cardiac arrhythmia
The device can identify heart rhythm abnormalities during the blood pressure measurement. If 🚫_ is displayed after the measurement, this indicates that an irregularity has been detected in your pulse.
Repeat the measurement if 🤒 is displayed.
When assessing your blood pressure, only use the results that have been recorded without any irregularities in your pulse.
Consult your doctor if 🤨 is displayed frequently. Only they can determine, through an examination, whether there is an abnormality.
Risk indicator
| Measured blood pressure value range | Classification | Risk indicator colour | |
| Systolic (in mmHg) | Diastolic (in mmHg) | ||
| ≥ 180 ≥ 110 | Stage 3 high blood pressure (severe) _1 | Red | |
| 160 – 179 100 – 109 | Stage 2 high blood pressure (moderate) _1 | Orange | |
| 140 – 159 90 – 99 | Stage 1 high blood pressure (mild) _1 | Yellow | |
| 130 – 139 85 – 89 High normal _1 | Green | ||
| 120 – 129 80 – 84 Normal _1 | Green | ||
| < 120 < 80 Optimal _1 | Green | ||
| < 90 < 60 Low blood pressure _2 | Orange | ||
1 Source: WHO, 1999 (World Health Organization)
2 Source: National Health Service, 2023
The risk indicator 3/18 indicates which category the recorded blood pressure values fall into. If the measured values are in two different categories (e.g. systolic pressure in the “high normal” range and diastolic pressure in the “normal” range), the risk indicator always indicates the higher range – “high normal” in the example described.
Note that these default values are for general guidance only, as individual blood pressures may vary.
Please note that self-measurement at home usually results in values lower than those recorded at a doctor's surgery. Consult your doctor at regular intervals. Only they are able to give you personal target values for controlled blood pressure, particularly if you are receiving medical therapy.
Low blood pressure

WARNING
Low blood pressure (hypotension) can be a health hazard and cause dizziness or fainting. Blood pressure is considered low if systolic and diastolic pressure are below 90/60 mmHg (source: National Health Service, 2023).
Seek medical attention if you suddenly suffer from low blood pressure.
6.5 Displaying and deleting measured values
User
The results of every successful measurement are saved with the date and time. When the number of measurements exceeds 120, the oldest measurements are deleted.
Average value
AVG lights up:
The average value of all this user's saved measured values is displayed.
- Press >.
AVG lights up:
The average value of the morning measurements for the last 7 days is displayed (morning: 5:00–9:00).
- Press >.
AVG lights up:
The average value of the evening measurements for the last 7 days is displayed (evening: 18:00–20:00).
Individual measured values
- If you press > again, the last individual measurement is displayed.
- If you press > again, you can view the individual measured values.
- To switch the device off again, press and hold ① for more than 2 seconds. To exit the menu, briefly press ①
Deleting measured values
- To delete all of one user's saved measurements, go into the relevant user memory.
AVG will light up on the display, and the average value of all this user's saved measured values will be shown. - Press and hold = and > for approximately 5 seconds.
The display shows CLO for for All this user's values are deleted.
The device switches off automatically.
Reset the device to factory settings
- To delete all saved measured values and settings, select the user memory. Press >. The display shows AVG
- Press and hold = and > for approximately 15 seconds. [CL ALL] appears on the display. All data stored on the device is deleted; the device has been reset to factory settings. The device switches off automatically.
7. CLEANING AND MAINTENANCE
- Clean the device and cuff carefully using only a slightly damp cloth.
- Do not use any cleaning solutions or solvents.
- Under no circumstances hold the device or cuff under water, as this can cause liquid to enter and damage the device and cuff.
- If you store the device and cuff, do not place heavy objects on the device and cuff.
- Remove the batteries if the device is not going to be used for a long period of time.
8. TROUBLESHOOTING
| Error message | Possible cause Solution | |
| Er1 | Unable to record a pulse. | Please wait one minute and repeat the measurement.Ensure that you do not speak or move during the measurement. |
| Er2 | The measured blood pressure is outside the measurement range. | |
| Er3 | There is a pneumatic system error. | Repeat the measurement.Ensure that the cuff is correctly positioned and that you do not move or speak. |
| Error message | Possible cause Solution | |
| Er4 | An error occurred during the measurement. | Please wait one minute and repeat the measurement.Ensure that you do not speak or move during the measurement. |
| Er5 | The inflation pressure is higher than 300 mmHg. | Please take another measurement to check whether the cuff can be correctly inflated. |
| Er6 | There is a system error. | If this error message appears, please contact Customer Services. |
| ☐ L0 | The batteries are nearly flat. | Insert new batteries into the device. |
9. DISPOSAL
Repairing and disposing of the device
For environmental reasons, do not dispose of the device in household waste at the end of its service life. Dispose of the device at a suitable local collection or recycling point. Observe the local regulations for material disposal. Dispose of the device in accordance with EC Directive – WEEE (Waste Electrical and Electronic
Equipment). If you have any questions, please contact the local authorities responsible for waste disposal. You can obtain the location of collection points for old devices from the local authorities.

Disposal of the batteries
Used, completely discharged batteries must not be disposed of with household waste. Dispose of the batteries in specially designated collection boxes, at recycling points or at electronics retailers. You are legally required to dispose of the batteries correctly.
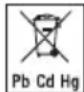
The codes below are printed on batteries containing harmful substances: Pb = battery contains lead, Cd = battery contains cadmium, Hg = battery contains mercury
10. TECHNICAL SPECIFICATIONS
Type DBP-61D6
| Model SBM 54 | |
| Measurement method | Oscillometric, non-invasive blood pressure measurement on the upper arm |
| Measurement range | Cuff pressure 299 mmHg, systolic pressure 50 – 260 mmHg, diastolic pressure 40 – 200 mmHg, pulse 30 –180 beats/minute |
| Display accuracy | Systolic pressure ± 3 mmHg, diastolic pressure ± 3 mmHg, pulse ± 5% of the displayed value |
| Measurement uncertainty | Max. permissible standard deviation according to clinical testing: systolic pressure 8 mmHg, diastolic pressure 8 mmHg |
| Memory 2 x 120 memory spaces | |
| Dimensions L 130 mm x W 66 mm x H 30 mm | |
| Weight Approx. 229 g (without batteries, with cuff) | |
| Cuff size 22 to 42 cm upper arm circumference | |
| Operating conditions | + 10 °C to + 40 °C, 15% – 93% relative humidity, 800 –1060 hPa ambient pressure |
| Storage and transport conditions | - 25 °C to + 55 °C, ≤ 93% relative humidity (non-condensing) |
| Power supply | 4x1.5V — — LR03 AAA batteries |
| Battery life For approx. 400 measurements, depending on blood pressure and inflation pressure levels | |
| Product life cycle to be expected | Information on the life cycle of the product can be found at www.sanitas-online.de |
| Classification Internal power supply, IP 22 no AP or APG, continuous operationBlood pressure: Application part, type BF | |
The serial number is located on the device or in the battery compartment.
We reserve the right to make technical changes to improve and develop the product.
- The device conforms with the respective national regulations and European standard EN 60601-1-2 (Group 1, Class B, in accordance with CISPR-11, IEC 61000-3-2, IEC 61000-3-3, IEC 61000-4-2, IEC 61000-4-3, IEC 61000-4-4, IEC 61000-4-5, IEC 61000-4-6, IEC 61000-4-7, IEC 61000-4-8, IEC 61000-4-11) and is subject to particular precautions with regard to electromagnetic compatibility.
- The device has been clinically tested in accordance with the requirements of ISO 81060-2.
- The accuracy of this device has been carefully checked and it has been developed with a long service life in mind. If the device is used for commercial medical purposes, the applicable
national regulations determine whether it must be tested for accuracy by appropriate means.
11. GUARANTEE/SERVICE
Hans Dinslage GmbH, Riedlinger Straße 28, 88524 Uttenweiler (hereinafter referred to as "HaDi"), provides a guarantee for this product, subject to the requirements below and to the extent described as follows.
Claiming the guarantee shall not affect the Purchaser's statutory guarantee rights arising from the purchase agreement with the Seller in the event of defects. The Purchaser may exercise these statutory guarantee rights free of charge. The guarantee shall apply without prejudice to any mandatory statutory provisions on liability.
HaDi guarantees the perfect functionality and completeness of this product.
The worldwide guarantee period is 3 years, commencing from the purchase of the new, unused product from the seller.
The guarantee only applies to products purchased by the buyer as a consumer and used exclusively for personal purposes in the context of domestic use. German law shall apply.
During the guarantee period, should this product prove to be incomplete or defective in functionality in accordance with the following provisions, HaDi shall carry out a repair or a replacement delivery free of charge, in accordance with these guarantee conditions.
Should the buyer wish to file a guarantee claim, they shall first contact the HaDi Customer Service team:
Service hotline (free): E-mail:
GB Tel.: 0800 931 0319 service-uk@sanitas-online.de
The buyer will then receive further information about the processing of the guarantee claim, e.g. where they can send the product and what documentation is required.
If we request the buyer to send the defective product, the product must be sent to the following address:
NU Service GmbH
Lessingstraße 10 b
89231 Neu-Ulm
Germany
A guarantee claim shall only be considered if the buyer can provide HaDi, or an authorised HaDi partner, with
- a copy of the invoice/purchase receipt, and
- the original product.
The following are explicitly excluded from this guarantee:
- deterioration due to normal use or consumption of the product;
- accessories supplied with this product which are worn out or used up through proper use (e.g. batteries, rechargeable batteries, cuffs, seals, electrodes, light sources, attachments and nebuliser accessories);
- products that are used, cleaned, stored or maintained improperly and/or contrary to the provisions of the instructions for use, as well as products that have been opened, repaired or modified by the buyer or by a service centre not authorised by HaDi;
-
damage that arises during transport between manufacturer and customer, or between service centre and customer;
-
products purchased as seconds or as used goods;
- consequential damage arising from a fault in this product (however, in this case, claims may exist arising from product liability or other compulsory statutory liability provisions).
Repairs or an exchange in full do not extend the guarantee period under any circumstances.
Notification of incidents
For users/patients in the European Union and identical regulation systems, the following applies: If a major incident occurs during or through use of the product, notify the manufacturer and/or their representative of this as well as the respective national authority of the member state in which the user/patient is located.
FR BE FRANÇAIS

Instructions relatives aux piles
AVERTISSEMENT
1Source: WHO, 1999 (World Health Organization)
_2 Source: National Health Service, 2023
The Ground Truth image displays a single, solid horizontal line. According to Rule 2 (UNDERSCORE & LINE RULES), this is a stylistic or background line, not a placeholder underscore. Therefore, the OCR result must ignore it and output nothing or only meaningful text. The provided OCR content is "____", which consists of four underscores. This is an incorrect interpretation of the line as a placeholder, violating the rule that stylistic lines must be ignored. The OCR has hallucinated underscores where none should exist based on the GT's visual context. Hence, the OCR result is inconsistent with the Ground Truth.

5. BESCHRIJVING VAN HET APPARAAT
1 Bron: WHO, 1999 (World Health Organization)
2 Bron: National Health Service, 2023
11. GWARANCJA/SERWIS
Manufacturer: Joytech Healthcare Co., Ltd.,
No. 365, Wuzhou Road, 311100 Hangzhou, Zhejiang, Province, PEOPLE'S REPUBLIC OF CHINA

Shanghai International Holding Corp. GmbH (Europe), Eiffestrasse 80, 20537 Hamburg, Germany

Imported by: EU: Hans Dinslage GmbH, Riedlinger Strase 28, 88524 Uttenweiler, www.sanitas-online.de