SBC 21 - Blood pressure monitor SANITAS - Free user manual and instructions
Find the device manual for free SBC 21 SANITAS in PDF.

User questions about SBC 21 SANITAS
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Blood pressure monitor in PDF format for free! Find your manual SBC 21 - SANITAS and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. SBC 21 by SANITAS.
USER MANUAL SBC 21 SANITAS
Quelle: WHO, 1999 (World Health Organization)
- Getting to know your device 12
- Important notes 12
- Unit description 15
4.Preparing the measurement 16 - Measuring blood pressure 17
Please read these instructions for use carefully and keep them for later use, be sure to make them accessible to other users and observe the information they contain.
1. Getting to know your device
The wrist blood pressure monitor is used to carry out noninvasive measurement and monitoring of arterial blood pressure values in human adults. This allows you to quickly and easily measure your blood pressure and to display the last recorded measurement.
A warning is issued for anyone suffering from cardiac arrhythmia.
The recorded values are classified and evaluated graphically.
2. Important notes

Signs and symbols
The following symbols are used in these instructions for use, on the packaging and on the type plate for the device and the accessories:
- Saving and displaying measurements 19
- Deleting stored values 19
- Error message/troubleshooting 19
- Cleaning and storing the device and cuff 19
- Technical specifications 19
| Attention | |
| Note Note on important information | |
| Observe the instructions for use | |
| Application part, type BF | |
| Direct current | |
| Dispose of the device in accordance with EC Directive – WEEE (Waste Electrical and Electronic Equipment). | |
| Manufacturer | |
| Storage -20°C RH 10-95% | Permissible storage temperature and humidity |
| Operating 10°C 40°C RH 30-85% | Permissible operating temperature and humidity |
| J | Protect from moisture |
| SN | Serial number |
| CE 0483 | The CE labelling certifies that the product complies with the essential requirements of Directive 93/42/EEC on medical products. |

Advice on use
- In order to ensure comparable values, always measure your blood pressure at the same time of day.
- Before every measurement, relax for about five minutes.
- If you want to perform several measurements on the same person, wait five minutes between each measurement.
- Do not take a measurement within 30 minutes after eating, drinking, smoking or exercising.
- Repeat the measurement if you are unsure of the measured value.
- The measurements taken by you are for your information only - they are not a substitute for a medical examination! Discuss the measurements with your doctor, and never base any medical decisions on them (e.g. medicines and their administration)!
-
Do not use the blood pressure monitor on newborns, pregnant women or patients with preeclampsia.
-
In the case of restricted circulation on the arm as a result of chronic or acute vascular diseases (including vascular constriction), the accuracy of the wrist measurement is limited. In this case you should avoid using an upper arm blood pressure monitor.
- This device is not intended for use by people (including children) with restricted physical, sensory or mental skills or a lack of experience and/or a lack of knowledge, unless they are supervised by a person who has responsibility for their safety or they receive instructions from this person on how to use the device. Supervise children around the device to ensure they do not play with it.
- Cardiovascular diseases may lead to incorrect measurements or have a detrimental effect on measurement accuracy. The same also applies to very low blood pressure, diabetes, circulatory disorders and arrhythmias as well as chills or shaking.
- The blood pressure monitor must not be used in connection with a high-frequency surgical unit.
- Only use the unit on people who have the specified wrist measurement for the device.
- Please note that when inflating, the functions of the limb in question may be impaired.
- During the blood pressure measurement, blood circulation must not be stopped for an unnecessarily long time. If the device malfunctions, remove the cuff from the arm.
- Do not allow sustained pressure in the cuff or frequent measurements. The resulting restriction of the blood flow may cause injury.
-
Ensure that the cuff is not placed on an arm in which the arteries or veins are undergoing medical treatment, e.g. intravascular access or therapy, or an arteriovenous (AV) shunt.
-
Do not use the cuff on people who have undergone a mastectomy.
- Do not place the cuff over wounds as this may cause further injury.
- Place the cuff on your wrist only. Do not place the cuff on other parts of the body.
- The blood pressure monitor can only be operated with batteries.
- To conserve the batteries, the monitor switches off automatically if no buttons are pressed for two minutes.
- The device is only intended for the purpose described in these instructions for use. The manufacturer is not liable for damage resulting from improper or careless use.

Instructions for storage and maintenance
-
The blood pressure monitor is made from precision and electronic components. The accuracy of the measurements and service life of the device depend on its careful handling:
-
Protect the device from impacts, humidity, dirt, marked temperature fluctuations and direct sunlight.
- Do not drop the device.
- Do not use the device in the vicinity of strong electromagnetic fields and keep it away from radio systems or mobile telephones.
- Do not press the buttons before the cuff is placed on the arm.
- We recommend that the batteries be removed if the device will not be used for a prolonged period of time.

Notes on handling batteries
-
If your skin or eyes come into contact with battery fluid, flush out the affected areas with water and seek medical assistance.
-
Choking hazard! Small children may swallow and choke on batteries. Store the batteries out of the reach of small children.
- Observe the plus (+) and minus (-) polarity signs.
- If a battery has leaked, put on protective gloves and clean the battery compartment with a dry cloth.
- Protect the batteries from excessive heat.
Risk of explosion! Never throw batteries into a fire.
- Do not charge or short-circuit batteries.
- If the device is not to be used for a long period, take the batteries out of the battery compartment.
- Use identical or equivalent battery types only.
Always replace all batteries at the same time. - Do not use rechargeable batteries.
- Do not disassemble, split or crush the batteries.

Instructions for repairs and disposal
- Batteries do not belong in household waste. Please dispose of empty batteries at the collection points intended for this purpose.
- Do not open the device. Failure to comply will result in voiding of the warranty.
- Do not repair or adjust the device. Proper operation can no longer be guaranteed in this case.
- Repairs must only be carried out by Customer Services or authorised suppliers. Before making a claim, please check the batteries first and replace them if necessary.
- For environmental reasons, do not dispose of the device in the household waste at the end of its useful life. Dispose of
the device at a suitable local collection or recycling point. Dispos of the device in accordance with EC Directive - WEEE (Waste Electrical and Electronic Equipment). If you have any questions, please cont local authorities responsible for waste disposal.

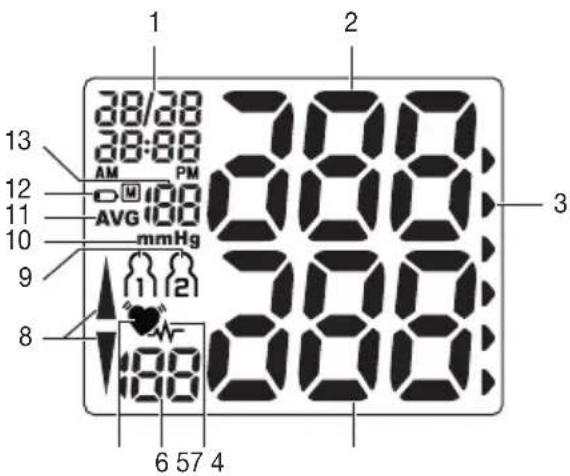
3. Unit description
- Display
- Scale for classifying the measurements
- Wrist cuff
- START/STOP button ①
- User selection button
- Memory button +/M
- Date/time setting button
- Battery compartment lid

Information on the display:
- Time and date
- Systolic pressure
- Classification of measurements
- Diastolic pressure
5.Cardiac arrhythmia symbol - Calculated pulse value
- Pulse symbol
- Pump up, release air (arrow)
- User memory
- Unit in mmHg
- Average value of the last three measurementsAVG
- Battery level indicator
- Memory space number
4. Preparing the measurement Insert the batteries
- Remove the battery compartment lid on the left side of the device.
- Insert two 1.5 V micro (alkaline type LR03) batteries.
Make sure that the batteries are inserted the correct way round. Do not use rechargeable batteries.
- Close the battery compartment lid again carefully.
If the battery change symbol is flashing and E appears, no more measurements are possible and you must replace all batteries.
After inserting the batteries, the year appears on the display and you can then set the date and time.

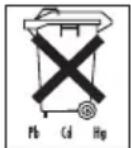
Battery disposal
- The empty, completely flat batteries must be disposed of through specially designated collection boxes, recycling points or electronics retailers. You are legally required to dispose of the batteries.
- The codes below are printed on batteries containing harmful substances:
Pb = Battery contains lead,
Cd = Battery contains cadmium,
Hg = Battery contains mercury.
Setting the date and time
You should set the date and time without fail. Otherwise, you will not be able to save your measured values correctly with a date and time and access them again later. The time is displayed in the 24-hour format.
To set the date and time, proceed as follows:
- After inserting the batteries:
- Following the display check, during which all segments appear, the year flashes on the display.
- Using the + / M button, you can increase the basic values to be set. Set the year and confirm the entry using the button.
- Now set the month using the + / M button. Confirm using the button.
- Set the day/hour/minute and confirm each using the button.
-
To switch off, press the START/STOP button ①
-
At any time:
-
Press the button to display the date/time and current user memory.
- Press and hold the button again for about 3 seconds to reach Date and time mode.
- Set as described above.
5. Measuring blood pressure Attach the cuff
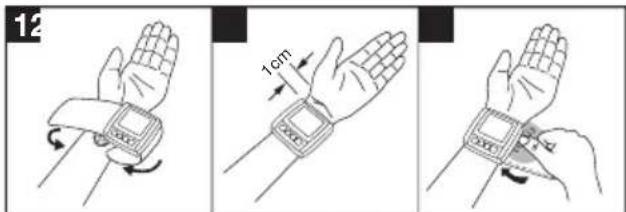
- Expose your left wrist. Ensure that the circulation of the arm is not hindered by tight clothing or similar.
- Place the cuff on the inside of your wrist.
- Fasten the cuff with the hook and loop fastener so that the upper edge of the monitor is positioned approx. 1 cm below the ball of your thumb.
- The cuff must be fitted tightly around the wrist, but must not constrict.
Adopt the correct posture
Before every measurement, relax for about five minutes. Otherwise deviations can occur.
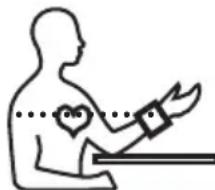
- You can perform the measurement while sitting or lying. To carry out a blood pressure measurement, make sure you are sitting comfortably with your arms and back leaning on something. Do not cross your legs. Place your feet flat on the ground. Make sure to rest your arm and move it. Always make sure that the cuff is at heart level.
Otherwise significant deviations can occur. Relax your arm and the palm of your hand.
- To avoid falsifying the measurement, it is important to remain still during the measurement and not to speak!
Selecting memory
Press the button to display the date/time and current user memory. Select the desired user memory by pressing the user selection button. Confirm your selection with the button.
You have 2 memories with 60 memory spaces each to store the measurements of 2 different people separately or to separate measurements taken in the morning and at night.
Performing the blood pressure measurement
- As described above, attach the cuff and adopt the posture in which you want to perform the measurement.
- Select the user memory for . Start the measurement by holding the button. Following the display check, during which all numbers appear, the cuff inflates automatically. Whilst the cuff is pumping up, the device already calculates measurements for estimating the necessary pump pressure. If this pressure is insufficient, the device automatically pumps more pressure.
- Then the pressure in the cuff is slowly released and the pulse is detected.
- When the measurement has been taken, the remaining air pressure dissipates very quickly. The pulse, the systolic and diastolic blood pressure are displayed.
- Measuring can be cancelled at any time by pressing (1)
The symbol E appears if the measurement could not be performed properly. Take note of the chapter on error messages/
trouble-shooting in these instructions for use and repeat the measurement.
The device switches off automatically after 2 minutes.
Wait at least 5 minutes before taking another measurement!
Evaluating results
Cardiac arrhythmias:
This unit can identify potential disruption of the heart rhythm when measuring and if necessary, indicates this after the measurement with the symbol
This can be an indicator for arrhythmia. Arrhythmia is an illness in which the heart rhythm is abnormal because of flaws in the bioelectrical system that regulates the heartbeat. The symptoms (skipped or premature heart beats, pulse being slow or too fast) can be caused by factors such as heart disease, age, physical make-up, excess stimulants, stress or lack of sleep. Arrhythmia can only be determined through an examination by your doctor. If the symbol is shown on the display after the measurement has been taken, it should be repeated. Please ensure that you rest for 5 minutes beforehand and do not speak or move during the measurement. If the symbol appears frequently, please consult your doctor. Self-diagnosis and treatment based on the measurements can be dangerous. Always follow your GP's instructions.
Classification of measurements:
The measurements can be classified and evaluated in accordance with the following table.
However, these standard values serve only as a general guideline, as the individual blood pressure varies in different people and different age groups etc.
It is important to consult your doctor regularly for advice. Your doctor will tell you your individual values for normal blood pressure as well as the value above which your blood pressure is classified as dangerous.
The classification on the display and the scale on the unit show which category the recorded blood pressure values fall into. If the values of systole and diastole fall into two different categories (e.g. systole in the ,High normal' category and diastole in the ,Normal' category), the graphical classification on the device always shows the higher category; for the example given this would be ,High normal'.
| Blood pressure value category | Systole (in mmHg) | Diastole (in mmHg) | Action |
| Setting 3: severe hyper-tension | ≥180≥110 | seek medical at- tention | |
| Setting 2: moderate hyper-tension | 160–179 | 00–109 | seek medical at- tention |
| Setting 1: mild hyperten-sion | 140–159 | 0–99 | regular monitoring by doctor |
| High normal 130– | 13985–89 | regular monitoring by doctor | |
| Normal 120–129 | 80–84 self-monitoring | ||
| Optimal <120<80 self-monitoring | |||
Source: WHO, 1999 (World Health Organization)
6. Saving and displaying measurements
- The results of every successful measurement are stored together with the date and time. If there are more than 60 measurements, the oldest measurements are lost.
- Use the button and then the button to select the desired user memory. Pressing the + / M button displays the average value of the 3 most recent measurements stored in the user memory. If you continue to press the memory button + / M , the most recent individual measured values are displayed in turn with the date and time.
7. Deleting stored values
To delete individual measurements, you must first select them. Press and hold the memory button + / M until appears in the display. Then confirm the deletion with the user selection button (and appear on the display).
To delete measurements from a user memory, you must first select them. Press the memory button + / M and the average value of the last 3 saved measurements is displayed. Then press and hold the memory button + / M again until appears in the display. Confirm the deletion with the user selection button (and appear on the display).
8. Error message/troubleshooting
In the event of errors, the error message E appears on the display.
Error messages may appear if:
- it was not possible to record the pulse: E 1
- you move or speak during the measurement: E2
-
the cuff is fastened too tightly or loosely: E3
-
errors occur during the measurement: £4
- the pump pressure is higher than 300mmHg : E5
the batteries are almost empty E6
In such cases, repeat the measurement. Ensure that you do not move or speak. If necessary, reinsert or replace the batteries.
9. Cleaning and storing the device and cuff
- Clean the device and cuff carefully using a slightly damp cloth only.
- Do not use any cleaning agents or solvents.
- Under no circumstances hold the device and cuff under water, as this can cause liquid to enter and damage the device and cuff.
- If you store the device and cuff, do not place heavy objects on the device and cuff. Remove the batteries.
10. Technical specifications
Model no. SBC 21
| Measurement method | Oscillometric, non-invasive blood pressure measurement on the wrist |
| Measurement range | Cuff pressure 0-300 mmHg, systolic 50-250 mmHg, diastolic 30-200 mmHg, pulse 40-180 beats /minute |
| Display accuracy | Systolic ± 3 mmHg, diastolic ± 3 mmHg, pulse ± 5 % of the value shown |
| Measurement inaccuracy | Max. permissible standard deviation according to clinical testing: systolic 8 mmHg /diastolic 8 mmHg |
| Memory 2 x 60 memory spaces | |
| Dimensions L 70 mm x W 72 mm x H 27.5 mm | |
| Weight Approximately 105 g (without batteries) | |
| Cuff size | 135 to 195 mm |
| Permissible operat-ing conditions | +10 °C to +40 °C, 30-85 % relative hu-midity (non-condensing) |
| Permissible storage conditions | -20 °C to +60 °C, 10-95 % relative hu-midity, 700-1050 hPa ambient pressure |
| Power supply 2x1.5V | === AAA batteries |
| Battery life For approx. 300 measurements, depending on levels of blood pressure and pump pressure | |
| Accessories Instructions for use, 2 x 1.5 V AAA ba-teries, storage box | |
| Classification Internal supply, IPX0, no AP or APG, continuous operation, application part type BF | |
- This unit is in line with European Standard EN 60601-1-2 and is subject to particular precautions with regard to electromagnetic compatibility (EMC). Please note that portable and mobile HF communication systems may interfere with this unit. More details can be requested from the stated Customer Services address or found at the end of the instructions for use.
- This device is in line with the EU Medical Devices Directive 93/42/EEC, the "Medizinproduktegesetz" (German Medical Devices Act) and the standards EN 1060-1 (non-inva
sive sphygmomanometers, Part 1: General requirements), EN 1060-3 (non-invasive sphygmomanometers, Part3: Supplementary requirements for electro-mechanical blood pressure measuring systems) and IEC 80601-2-30 (Medical electrical equipment - Part 2-30: Particular requirements for the safety and essential performance of automated non-invasive blood pressure monitors).
- The accuracy of this blood pressure monitor has been carefully checked and developed with regard to a long useful life. If using the device for commercial medical purposes, it must be regularly tested for accuracy by appropriate means. Precise instructions for checking accuracy may be requested from the service address.
FRANÇAIS
Sommaire
- Presentation 21
2.Conseils important 21
3.Description de I'appareil 24
4.Preparation de la mesure 25
5.Mesurer la tension 26
2. Consels important
Symboles utilisés
EcnMnMnraeT CmBON CMeHb6aTapeKu NnHa DnCnnee NOBnEeTcE 50 npOBeHeHne n3MepeHn HeBO3MOxHo, CJeDyET NOnHOCTbO 3aMeHHTb 6aTapeKn.
Korda 6atapeyn BCTabneHb,Ha dncnnee noaBnaretc ro, Tepeb Moxho HAcTpntb DaTy n Bpemr.

Ytuiin3aun6atapeek
BbI6paCbBaIte ICNoJIb3OBAHHbIe, NOJIHOCTbIO pa3pJxHbIe 6aTapeKn B CneUmaJIbHbIe KOHTeINHePbI, CdAbaTe B NHyKThI IprEma CNEcOTXODOB INIM B MaIa3INbI 3JIeKTPO-OBOpyIDoBAHn. 3aKOH O6BaET NOJIb3OBATeNe OBeCtHuTb yTuIN3aQuIO 6aTapeek.
CneyuOuIe 3HaKn npEynpeKdaIoT O HAnuHn B 6aTaapeKax TOKcHbIX BeueCTB: Pb = cBnHeU, Cd = KaMn, Hg = pTyTb.
HactpoKa DaTbI IN BpeMeHN
B 063aTeIbHOM NopJKe Heo6xOIMO yCTaHOBt TaTy n Bpem. 3TO NO3BOJNT npabINbHO COXpaHrTb B nAMrTI pe3yJIbTaTb I3MepeHn Cnx DaToN BpemHeM, a 3aTeM BblBOJtB nx Ha ekpaH. Bpem OTo6paKaetcB 24-ycobom fOpMaTe.
HactpoKn DaTbI N BpeMeHN DeiCTByTe CneDyUcIM 06pa3OM:
- He 3aHMaTecb aKTHBHOJ DeTeJIbHOCTbIO npIMepHO B TeHeHne 5 MInHT nepeI n3MepeHem! B IpOTNBHom cIyUaYe BO3MOXHbI OTKIOHEHnpe3YbTaTOB N3MepeHn.
BbMOKeTe npoBOuNTb 3MepeHne BIOJIOXeHN CNJa NIN
Jexka.ДяИЗмеренЯ apTeprnaIbHOro DaBHeHra 3aIMnTe yO6HoeNoIoxKeHne cnJa.CnHa n pyKINoJIxHbI IMTe onOpy.HeCKpeuBaIte HOrn.IocTaBbTeCTynHnPoBHO hon.Heo6XoDmONoIpePbpy Ky n corHyTbe ee B JOKTe
063aTeJIbHO CNEIHTe 3a TEM, YTO6bl MaHKeTa HaxOJIaIacb Ha yPoBHe cepIa. B npOTnBHOM cIyae BO3MOXHbI 3HaHTeJIbHbIe OKIOHEHnpe3yJbTAToB n3MepeHn. PaccIa6bTe pyKu I naDoHn.
- UTO6bI N36ExKaTb NCKaXeHnpe3yJbTaTOB N3MepeHn, BO BpEMr N3MepeHn CJeNyET BcTeN Ce6r CnOKoHn H He pa3- roBaPbBaTb.
Bb6op namrtn
Ipn HaxkTIN KHOJKN OTo6pa3ntc DaTa/BpeMn TEkyuaa NOnb3OBaTeNbcka NaMBy. Bb6epnte HyxHyIO NOnb3OBaTeNbckyIO NaMBy, HaxkAB KHOJKy BB6opaa NOnb3OBaTeNl IOnTBepnTe Bb6Op HaxkTneM KHOJKn 0Meetc DBe NaMnC 60 RaHKeAMN B KaKdoi, YTO6bl MOXHO 6blpo pa3dENo COxpaHrB pe3yNbTaTb N3MepeHnN DnAByX yeNoBek nnn yTpEHnne n BeuePHeNn N3MepeHnN.
I3mepenhe apTepnabHoro daBJeHna
- HanoXnTe MaXkeTy, KaK OINcAHO BblSe, IN 3aIMnTe yDIO6Hoe. IJRA N3MepeHnI NOLOXKeHne.
BbI6epnte noJIb3ObaTeTn HnHTo6bl HaaytBu 3MepeHne daBHeHHaXMMte KhoNky TocJe npOBepKn DcPiJle, BO BpemKOTOpOn DoJnxHbI rOpTe Bce UΦpbbl,
MaHxeta aBtOMaTnueckn HakaunBaetc. Yke BO BpeMHaKaunBaHn npnbop onpeJeIeT 3NaueHn N3MepeHn, KOTOpbIe CnyKAT nO uChENk Tpe6yEmo r daBLeHn HaKaunBaHn. Ecn 3TOrO daBHeHn HeDocTaToUHO, np6bp aBtOMaTnueckn yBeJIuHT erO.
- Pocne 3Toro DaBHeHne B MaHKeTe HaHHaET MeDHeHNo ONyCKaTbCn INPOIN3BOIDTCr NOCDcET NylbCa.
- Iocne OKOHuaHnI N3MepeHnI OCTaTOHoe BO3dyuHoe daBJIeHne OueHb 6bICTpo CHnxKaETcR. Ha nucnnee OTObpaXaIOTcR nyIbC, CNTOJIuYeCKoe IN dnaCTOJIuYeCKoe KPOBraHoe DaBJIeHne.
BbMOKeTe BJIIO6OIMOMeHT npeBaTb N3MepeHne,HaXaB KHOKNy ①
CIMBOL E NOBRAETCA, ECIN IN3MepeHne He ydaIocb BblNoHNHTb npabnIbHO. IpOHTaIe rIaby «Co06uHHe 6Ow6Ke/ycTpaHHe HncnPabHOCTeB» B daHHo INCTpyKcUN NIO pPmEHeHIO NOBTOPTE IN3MepeHne.
-Пибор abTomatnueckn BbIKNoyaeTcR uee3 2 MInHytbl.
Ipeed oupeepdHbIM n3MepeHHem BbIXDHTe He MeHee 5 MnHyT!
OueHka pe3yIbTaTob
HapyeHnA cepdeHoro pHTMa:
DaHbI npu6op MoKe T nDeHTnФmUPOBaTb BO3MOXHbIe HApUWeHn cepJeHOro pITMa BO BpEmn3MepeHn. Pnp INx HAnuHn NocLe n3MepeHn OTo6pa3NTcR CMBON
3TO MOKET yka3bBaTb Ha apuTMIO. ApuTMn - 3TO 60Je3Nb, pni KOTopoB BO3HnKaet aHOMaJIbHbI CEpDeHbI pNTM, Bbl3BaHHbI OUn6kAmN B 6No3JIeKTPnueCKOcN CnCTEme, peryIInpyUoJeuE bneHne cepda. CmMTombI (HepaBHomepHoe IIN npexKeBpeMeHHoe cepdue6neHne, MeIeHHbI
NIN CINIKOM 6bICTpbIyJbc) MOrY 6bITb Bbl3BaHbI 3a6oJIeBAHnA M cepUa, BO3pactOM, INpePacNOJoxEHHOCbIO K 3a6oJIeBAHnA, Ype3MePbHM yNOTpe6JIeHnEM KoFe, HNKOTUNHa n aIKOROJI, CTpeccOM NII HeIOCTaTKOM CHa. ApNTMn MoXeT 6bITb BblBHeHa TOLbKO B pe3yJbTaTe o6cIeNoBaHnY bpaA.
Ecn nocne nepBoro n3MepenHa ducnnee OTo6paKaetc
CNMBONOBTOPNE n3MepenHe.Obpatne BHHMaHne
Ha To, YTO B TeueHne 5 MInyT nepei n3MepenHem HeIb3a
3aHIMatbcra kTNBHOJ DeTeJIbHOCTbIO,a BO BpEmn3MepenH
HeIb3ra roBOpNTb NIN DBIRATbcr.Ecn CNMBONoRBAeTc
acto, o6patntecb K BPay.CamodnaHoctnka HcamoneeuHne
Ha oChObe pe3yNbTaTO n3MepenHnMOryt 6bITb OnacHbl.
O63aTeJIbHO cLeyuTe peKOMEnDaunm CBOero Bpaua.
OueHka pe3yIbTaTOB n3MepeHH:
OueHky pe3yIbTaTOB n3MepeHn MOxHO npOBecTn
c NOMOuBTo Tablncbl, npedCTabLeHHo Hnxke.
Ondako 3TN cTAHdapThbIe 3NaueHn CnyXkAT TOnbKO O6UIM
OpneHTnpom, TaK KaK INDINBuaJIbHbIe 3NaueHn KPOBraHOrO
DaBJeHry y pa3HbIX JIODe BApbpyOTcB 3aBNCUMOCTN OT
pPnHaJNeXHoCTN K ToI IIN INHO BO3pactHO rpynnE n T. P.
BaxHo peryJrpho KOHCyIbTnPoBaTbcS C BpaOH. Bpa
OnPdeJNT BaUn INDNBuaJIbHbIe 3NaueHn HopMaJIbHOrO
KPOBraHOrO DaBHeHnA, a TaKKe 3NaueHn, BblSe KOToPbIX
KPOBraHoe daBJeHne CJIeNyET KlaaccnФuipOBaTb KaK OnaCHOe.
FpaJaun Ha DInCnlee n Shkana Ha np6ope Noka3bIAIoT,
B KaKOM dInaIa3OHe hAxODITcR n3MepeHno DaBJeHne.
EcJIn 3NaueHn CNCTOJIuYeCKoro n DnaCTOJIuYeCKoro
DaBJeHry hAXoJrTcB Pa3HbIX dInaIa3OHax (HaPmMep,
CnCTOJIuYeCKoe daBJeHne - BICOKOE B DOpyCTMbIX npedeJax,
a dnaactolnueckoe - HopmaIbHoe),To rpaufneckoe neleHne Ha npnbope Bcerda 6ydt OTo6paKaTb 6Oee BbICOKne npedebl, KaK B ONICAHHOM npIMpe: BBICOKe B DoNyCTMlbIX npedeJax.
IcToHnK:BO3,1999(World Health Organization)
6. CoxpaHeHne n Bbl3OB n3MepeHHbIX 3HaueHn
- Pe3yNbTaTbI KaKdOrO ycNeUHOro n3MepEnn CoXpaHrOTcC yKa3aHHeM DaTbI IN BpeMeHn n3MepEnn. Korda konuYeCTBO COxpaHeHHbIX pe3yNbTaTOB n3MepEnn ppeBbIaet 60, 6oJee paHHne daHHbIe ABTomATnueCKn ydaJIaOTcR.
HaXMMTE KONky 3aTEM KONky, ATo6bl Bb6paTb HyxHyIO NOIb3OBATeNbCkyo NaMAYb. EcNI HaxKaTb KONky +/M, OTo6pa3NTcpeDHee 3NaueHne N3 Tpex NocJeHNx COxpaHeHHbIX B daHHOIN b3OBATeNbCKoN pMaRTu N3MepeHHbIX 3NaueHm. Pn DaJIbHeNwem HaxaTN N KONKn CoXPAHeHn +/M 6byyTO6paKATbCn NocJeHnpe3yNbTaBtO TdJIbHbIX N3MepeHn C yka3AHm EM DaTb IN BpeMeHn.
7. YdaJIeHHe coXpaHeHHbIX daHHbIX
YTo6bI ydaNTb OTdJIbHbI pe3yIbTaT n3MepeHn, ChaJaBbI6epnte erO. HaxMnte N ydepXnBaIte HaxaToN KHOKcycoxpanHe +/M, do Tex nop noka Ha dncnlee He NOBnTCsCMBoN CBaTeM NoTBePnTe ydaJeHne HaKaTneM KHONKnBbI6opa noJIb3OBaTeJHa dncnlee NoRBArTCs CMBoJIb I
YTO6bI ydaNTb pe3yNbTaTb N3MepeHn B NOlb3OBaTeNbCKo nAMrTn, Chayana Bbi6epnte erO. HaxMMte KONKy COxpaHeHn +/M. NoBHTcra CpeHHe 3HaueHne no Tpem NocJeHm COxpaHeHHbIM pe3yNbTaTAM N3MepeHn. 3aTeM CHOba HaxMMte u YdepXNBaTe HaxaToN KHONky COxpaHeHn +/M, Noka Ha dncnlee He noBHTcra IIOdTBePnte ydaJeHne HaxaTneM KHONKn BBi6opa NOnb3OBaTeJn (Ha DInCnnee NoBBA TcR cMb0JI bL00
8. Coo6ueHne 06 oun6ke/yctpaHeHne oun6ok
Pn BO3HNKHOBeHn OuIN6OK Ha DnCnJee NOBJIeTc coo6ueHne 06 OuIN6Ke E.
Co06eHn8o6OwN6kaxIPOBnHOCTc,eCNI
He ydaocb n3mepntb nybc: E1
- BO Bpemn 3mpeHnBbI DnBraTeCb NnroBopnte: E2
MaHKeTa npuIeRaET CnUKOM CnIbHO NJIu CnUKOM Cna6o: E3; - PpOu3oUJN OUs6Kn BO BpeM u3MepeHn: E4
-Давлиеные НакачиBaHЯняпpeБыIaeТ 300 MM pt. CT.:E,S - 6aTapeiKn noTu n pa3pXeHbI: E6
B TaKnx Cnyaax BbIIOJHnTe NOBTOpHOe N3MepeHne. NOMHnTe, yTo BbI He DoJIxHbI DBNrA TbCBs IIN ROBOpNTB B nPoUecCe N3MepeHn. PIn Heo6XoJIMOCtN yCTaHOBnTe 6aTapeKn Ha MeCTO 3aHOBO NII 3aMeHnTe NX.
9. OuInCTka n xpaHHeHne npIn60pa n MaHXKeTbI
- PpOIN3BOIDTe OYNCTKy pnp6opu mMaHKeTbI COCTOPOXHOCTbIO. NcPONb3yInTe TOJIbKO cIeRka yBnaxKHeHHyO canPeTky.
He nCnoIb3yIte YnCTaIe CpeDCTBa IIN pACTBOPNTeJI. - Hn B KOem Cnyae He onyckaTne np6op n MaHKeTy B Ody, TAK KAK NOnaHaHHe BObl npBedeT K NOBpeXdEHNIO np60pa N MaHKeTbl.
- Ppi XpaHEnn Ha npnbOpE n MaHXeTe He DoJIKHbI CToRbT TaKeJbIe IpeMtebl. N3BJeKte 6aTaapeKn.
10. TexHnueckne daHHbIe
NMOeJI SBC 21
For all ME EQUIPMENT and ME SYSTEMS
| Guidance and manufacture's declaration - electromagnetic emissions | ||
| The SBC 21 is intended for use in the electromagnetic environment specified below. The customer of the user of the SBC 21 should assure that it is used in such and environment. | ||
| Emission test Compliance | Electromagnetic environment | guidance |
| RF emissions CISPR 11 Group 1 | The SBC 21 uses RF energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. | |
| RF emission CISPR 11 Class B The | SBC 21 is suitable for use in all establishments, including domestic establishments and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes. | |
Table 2
For all ME EQUIPMENT and ME SYSTEMS
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The SBC 21 is intended for use in the electromagnetic environment specified below. The customer or the user of SBC 21 should assure that it is used in such an environment. | |||
| Immunity test | IEC 60601 test level | Compliance level | Electromagnetic environment - guidance |
| Electrostatic discharge (ESD) IEC 61000-4-2 | ±6 kV contact ±8 kV air | ±6 kV contact ±8 kV air | Floors should be wood, concrete or ceramic tile. If floor are covered with synthetic material, the relative humidity should be at least 30%. |
| Power frequency (50Hz) magnetic field IEC 61000-4-8 | 3 A/m 3 A/m Power frequency magnetic fields should be at | magnetic fields should be at | levels characteristic of a typical location in a typical commercial or hospital environment. |
Table 3
For ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The SBC 21 is intended for use in the electromagnetic environment specified below. The customer or the user of SBC 21 should assure that it is used in such an environment. | |||
| Immunity test IEC | 60601 test level | Compliance level | Electromagnetic environment - guidance |
| Conducted RFIEC 61000-4-6Radiated RFIEC 61000-4-3 | 3 Vrms150 kHz to80 MHz3 V/m80 MHz to2.5 GHz | 3 Vrms3 Vrms | Portable and mobile RF communications equipment should be used no closer to any part of the SBC 21, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter.Recommended separation distance:d=[3.5/V]√Pd=[3.5/E1]√80 MHz to 800 MHzd=[7/E1]√80 MHz to 2.5 GHzWhere P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and d is the recommended separation distance in meters (m).Field strengths from fixed RF transmitters, as determined by an electromagnetic site surveya, should be less than the compliance level in each frequency rangeb. Interference may occur in the vicinity of equipment marked with the following symbol: |
| NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies.NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||
^a Field strengths from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the SBC 21 is used exceeds the applicable RF compliance level above, the SBC 21 should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as re-orienting or relocating the SBC 21.
b Over the frequency range 150kHz to 80MHz , field strengths should be less than 3V / m .
Table 4 For ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
| Recommended separation distances between portable and mobile RF communications equipment and the SBC 21 | |||
| The SBC 21 is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the SBC 21 can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and the SBC 21 as recommended below, according to the maximum output power of the communications equipment. | |||
| Rated maximum output power of transmitter W | Separation distance according to frequency of transmitter (m) | ||
| 150 kHz to 80 MHzd = [3.5/V1] √P | 80 MHz to 800 MHzd = [3.5/E1] √P | 800 MHz to 2.5 GHzd = [7/E1] √P | |
| 0,01 0.117 0.1167 0.2334 | |||
| 0,1 0.370 0.3689 0.7378 | |||
| 1 1.170 1.1667 2.3334 | |||
| 10 3.700 3.6893 7.3786 | |||
| 100 11.7 11.6667 23.3334 | |||
| For transmitters rated at a maximum output power not listed above, the recommended separation distance d in meters (m) can be determined using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer. NOTE 1 At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies. NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||

Hans Dinslage GmbH, Söflinger Str. 28, 88524 Uttenweiler, GERMANY