ComfortTENS Back - Pain relief device COMPEX - Free user manual and instructions
Find the device manual for free ComfortTENS Back COMPEX in PDF.

User questions about ComfortTENS Back COMPEX
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Pain relief device in PDF format for free! Find your manual ComfortTENS Back - COMPEX and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. ComfortTENS Back by COMPEX.
USER MANUAL ComfortTENS Back COMPEX
Indications for Use....4
Safety Warning....5
Contraindications....5
Warnings....5
Precautions....6
Adverse Reactions....8
Symbol and Title 8
Environmental Condition for Transport and Storage....9
Electromagnetic Compatibility 10
How the Device Works....14
Device Description....15
Operating Instruction 16
Performance Specifications....21
Cleaning and Maintenance 21
Trouble Shooting....22
Contact Information 23
INTRODUCTION
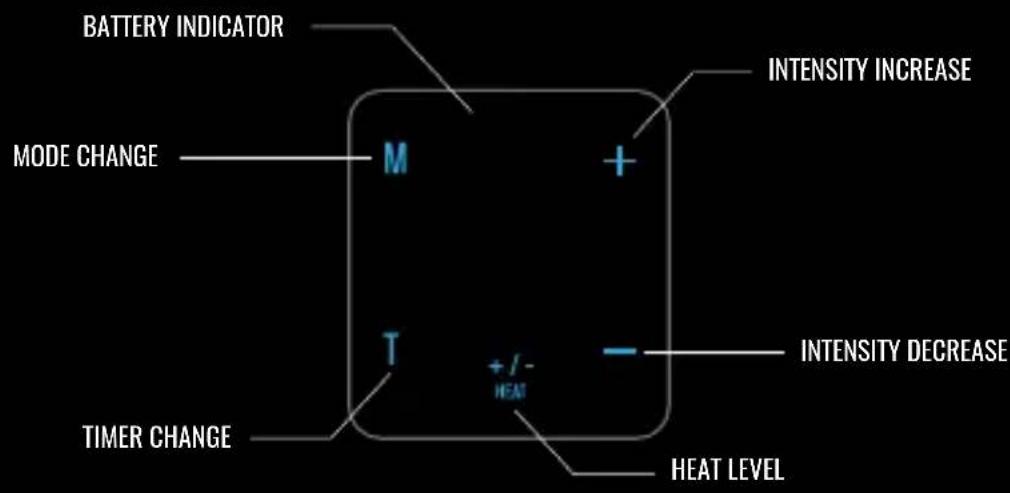
Compex Tens / Heat delivers electric pulses and heat to the user's body areas such as knee and back through the conductive silver pads. The portable and compact device has multiple modes of different pulse frequencies, covering Transcutaneous Electrical Nerve Stimulation (TENS) and Electrical Muscle Stimulation (EMS). It includes operating elements of ON/OFF button, intensity increase button, intensity decrease button, mode button, timer button, and heat/temperature button, and can be attached and detached to a wrap with conductive silver pads through magnetic connectors.
INDICATIONS FOR USE
To be used for temporary relief of pain associated with sore and aching muscles due to strain from exercise or normal household and work activities.
It is also intended for symptomatic relief and management of chronic, intractable pain and relief of pain associated with arthritis.
To stimulate healthy muscles in order to improve and facilitate muscle performance. To be used for the improvement of muscle tone and firmness, and for strengthening muscles. Not intended for use in any therapy or for the treatment of any medical conditions or diseases.
It is also intended to temporarily increase local blood circulation in the healthy muscles.
SAFETY WARNING
CONTRAINDICATIONS
Do not use this device on people who have a cardiac pacemaker, implanted defibrillator, or other implanted metallic or electronic device, because this may cause electric shock, burns, electrical interference, or death. Do not use this device on people whose pain syndromes are undiagnosed.
WARNINGS
- Use carefully. May cause serious burns. Do not use over sensitive skin areas or in the presence of poor circulation. The unattended use of this device by children or incapacitated persons may be dangerous. To reduce the risk of buns, electric shock, and fire, this device must be used in accordance with the instructions.
- Do not crush the device and the wrap and avoid sharp folds.
- Carefully examine the device and the wrap, and do not use if they show any sign of deterioration.
- Do not tamper with this device and the wrap in any way. There are no user serviceable parts. If for any reason they do not function satisfactorily, return to the authorized service center at address given.
- The long-term effects of chronic electrical stimulation are unknown.
- Stimulation should not be applied over the carotid sinus nerves, particularly in patients with a known sensitivity to the carotid sinus reflex.
- Stimulation should not be applied over the neck or mouth. Severe spasm of the laryngeal and pharyngeal muscles may occur and the contractions may be strong enough to close the airway or cause difficulty in breathing.
- Stimulation should not be applied transthoracically in that the introduction of electrical current into the heart may cause cardiac arrhythmias.
- Stimulation should not be applied transcerebrally.
- Stimulation should not be applied over swollen, infected, or inflamed areas or skin eruptions, e.g., phlebitis, thrombophlebitis, varicose veins, etc.
• Stimulation should not be applied over, or in proximity to, cancerous lesions.
PRECAUTIONS
- Safety of stimulation use during pregnancy has not been established.
- Caution should be used for people with suspected or diagnosed heart problems.
- Caution should be used for people with suspected or diagnosed epilepsy.
- Caution should be used if you have any of the following:
- if you have a tendency to bleed internally following an injury;
- if you recently had surgery, or have ever had surgery on your back;
- if areas of skin lack normal sensations, such as skin that is numb.
- Consult with your physician before use over the menstrual uterus.
- Caution should be used in the presence of the following:
- When there is a tendency to hemorrhage following acute trauma or fracture;
- Following recent surgical procedures when muscle contraction may disrupt the healing process;
• Over the menstruating or pregnant uterus;
• Over areas of the skin which lack normal sensation. - You may experience skin irritation or hypersensitivity due to the electrical stimulation or electrical conductive medium. The irritation can usually be reduced by using an alternate conductive medium, or alternate electrode placement.
- Do not use this device while driving, operating machinery, or during any activity in which involuntary muscle contractions may put the user at undue risk of injury.
- Keep this device out of reach of children.
- Do not use this device while sleeping.
- Do not use this device in high humidity areas such as a bathroom.
- Stop using this device at once if you feel discomfort, dizziness or nausea, and consult your physician.
- Do not attempt to move the conductive silver pads while the device is operating.
- Do not apply stimulation of this device in the following conditions:
- across the chest because the introduction of electrical current into the chest may cause rhythm disturbances to the heart, which could be lethal;
• over open wounds or rashes, or over swollen, red, infected, or inflamed areas or skin eruptions (e.g., phlebitis, thrombophlebitis, varicose veins); - in the presence of electronic monitoring equipment (e.g., cardiac monitors, ECG alarms); on children or incapacitated persons.
- Be aware of the following.
- consult with your physician before using this device;
- this device is not effective for pain associated with Central Pain Syndromes, such as headaches;
- the device is not a substitute for pain medications and other pain management therapies;
- the device is a symptomatic treatment and, as such, suppresses the sensation of pain that would otherwise serve as a protective mechanism;
- stop using the device if the device does not provide pain relief; and,
- use this device only with the conductive silver pads, and accessories recommended for use by the manufacturer
- Keep the device away from high-temperature and direct-sunlight place.
- Do not share the use of the conductive silver pads with others.
- Do not use the device while it's charging.
- The device contains the lithium battery. If overheating of the device occurred during the charging, stop the charging or operation immediately and report to the seller.
- Dispose of the battery-containing device according to the local, state, or federal laws.
- Skin burns may occur, and check the skin under the conductive silver pads periodically.
ADVERSE REACTIONS
- You may experience skin irritation and burns beneath the conductive silver pads applied to the skin;
- You should stop using the device and should consult with your physicians if you experience adverse reactions from the device.
SYMBOL AND TITLE
• Information essential for proper use shall be indicated by using the corresponding symbols. The following symbols may be seen on the device and labeling.
| Consult instructions for use | |
| Caution, consult accompanying documents | |
| Use by date Type BF applied | |
| Date of manufacture Manufa | |
| Batch code Temperature lim | |
| Unrecyclable |
| Catalogue number Humidity | |
| Serial number Non-sterile | |
| mitation | |
| CE 1639 | CE mark shows the conformity to the Medical Device Directive |
ENVIRONMENTAL CONDITION FOR TRANSPORT AND STORAGE
40 °C 5 °CTEMPERATURE LIMITATION 5 °CTEMPERATURE LIMITATION | Normal working ambient temperature: 5~40°C (41~104°F) |
90% 15 % 15 % | Normal working ambient humidity: 15~90% |
70 °C TEMPERATURE LIMITATION TEMPERATURE LIMITATION | Store and transport ambient temperature: -25~70°C (-13~158°F) |
90% 0 % 0 % | Store and transport ambient humidity: 0~90% |
| Fragile; handle with care | |
 | Keep away from rain |
 | Non-sterile |
 | Product packaging is able to be recycled |
| Atmospheric pressure: 70~106kPa |
ELECTROMAGNETIC COMPATIBILITY
- This product needs special precautions regarding electromagnetic compatibility (EMC) and needs to be installed and put into service according to the EMC information provided. This unit can be affected by portable and mobile radio frequency (RF) communications equipment.
- Do not use a mobile phone or other devices that emit electromagnetic fields, near the unit. This may result in incorrect operation of the unit.
- Caution: This unit has been thoroughly tested and inspected to assure proper performance and operation.
- Caution: This unit should not be used adjacent to or stacked with other equipment and that if adjacent or stacked use is necessary, this unit should be observed to verify normal operation in the configuration in which it will be used.
| GUIDANCE AND MANUFACTURE'S DECLARATION – ELECTROMAGNETIC EMISSION | ||
| The device is intended for use in the electromagnetic environment specified below. The customer of the user of the device should assure that it is used in such an environment. | ||
| Emission test Compliance Electromagnetic environment – guidance | ||
| RF emissionsCISPR 11 | Group 1 | The device uses RF energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. |
| RF emissionCISPR 11 | Class B | |
| Harmonic emissionsIEC 61000-3-2 | Class A | The device is suitable for use in all establishments, including domestic establish public low-voltage power supply network that supplies buildings used for domestic purposes. |
| Voltage fluctuations/ flicker emissionsIEC 61000-3-3 | Complies | |
| GUIDANCE AND MANUFACTURE'S DECLARATION - ELECTROMAGNETIC EMISSION | |||
| The device is intended for use in the electromagnetic environment specified below. The customer of the user of the device should assure that it is used in such an environment. | |||
| Immunity test | IEC 60601-1-2test level | Compliance level Electromagnetic environment - guidance | |
| Electrostatic discharge (ESD)IEC 61000-4-2 | ±8 kV contact±2 kV, ±4kV, ±8 kV,±15 kV air | ±8 kV contact±2 kV, ±4kV, ±8 kV,±15 kV air | Floors should be wood, concrete or ceramic tile. If floor are covered with synthetic material, the relative humidity should be at least 30%. |
| Electrical fast transient/ burstIEC 61000-4-4 | ±2 kV for power supply lines±1 kV for input/output lines | ±2 kV for power supply lines±1 kV for input/output lines | Main power quality should be that of a typical commercial or hospital environment. |
| SurgeIEC 61000-4-5 | ±1 kV line(s) to line(s)±2 kV line(s) to earth | ±1 kV line(s) to line(s)±2 kV line(s) to earth | Main power quality should be that of a typical commercial or hospital environment. |
| Voltage dips, short interruptions and voltage variations on power supply input linesIEC 61000-4-11 | 0 % UT; 0.5 cycleat 0°,45°,90°, 135°,180°, 225°, 270°,315°0 % UT ; 1 cycle70 % UT; 25/30 cycle0% UT; 250/300cycle | 0 % UT; 0.5 cycleat 0°,45°,90°, 135°,180°, 225°, 270°,315°0 % UT ; 1 cycle70 % UT; 25/30 cycle0% UT; 250/300 cycle | Main power quality should be that of a typical commercial or hospital environment. If the user of the device requires continued operation during power interruptions, it is recommended that the device be powered from an uninterruptible power supply or a battery. |
| Power frequency (50Hz/60Hz)magnetic fieldIEC 61000-4-8 | 30 A/m 50Hz/60Hz 30 A/m 50Hz/60Hz | Main power quality should be that of a typical commercial or hospital environment. If the user of the device requires continued operation during power interruptions, it is recommended that the device be powered from an uninterruptible power supply or a battery. | |
| NOTE UT is the a.c. mains voltage prior to application of the test level. | |||
| GUIDANCE AND MANUFACTURE'S DECLARATION - ELECTROMAGNETIC IMMUNITY | |||
| The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that it is used in such an environment. | |||
| Immunity test IEC 60601 test level Compliance | level Electromagnetic environment - guidance | ||
| Conducted RFIEC 61000-4-6Radiated RFIEC 61000-4-3 | 3 Vrms150 kHz to 80 MHz3 V RMS outside the ISM band, 6 V RMS in the ISM and amateur bands10 V/m80 MHz °C 2,7 GHz 80 % AM at 1 kHz | 3 Vrms150 kHz to 80 MHz3 V RMS outside the ISM band, 6 VRMS in the ISM and amateur bands10 V/m80 MHz °C 2,7 GHz80 % AM at 1 kHz | Portable and mobile RF communications equipment should not be used near any part of the device, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter.Recommended separation distance d = 1,2 d = 1,2 80 MHz to 800 MHz d = 2,3 80 MHz to 2,5 GHzWhere P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and d is the recommended separation distance in metres (m).Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey, a) should be less than the compliance level in each frequency range. b)Interference may occur in the vicinity of equipment marked with the following symbol: |
| NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies.NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||
| a Field strengths from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the device is used exceeds the applicable RF compliance level. | |||
observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as re-orienting or relocating the device.
b Over the frequency range 150 kHz to 80 MHz, field strengths should be less than 3 V/m.
THE DEVICE IS INTENDED FOR USE IN THE ELECTROMAGNETIC ENVIRONMENT SPECIFIED BELOW. THE CUSTOMER OR THE USER OF THE DEVICE, SHOULD ASSURE THAT IT IS USED IN SUCH AN ENVIRONMENT.
| Test Frequency (MHz) | Band^a | Service^a | Modulation^a | Maximum Power (W) | Distance (M) | Immunity Test Level (V/m) |
| 385 380-390 TETRA 400. Pulse Modulation | b18Hz | 1.8 0.3 27 | ||||
| 450 430-470 GMRS 460. FRS 460. FM | c+5kHz sine 2 0.3 28 | |||||
| 710 | 704-787 LTE Band 13. 17. | Pulse Modulation^b 217 Hz | 0.2 0.3 9745 | |||
| 780 | ||||||
| 810 | 800-960 | GSM 800/900.TETRA 800. iDEN 820.CDMA 850. LTE.Nand 5. | Pulse Modulation^b 18 Hz | 2 0.3 28 | ||
| 870 | ||||||
| 930 | ||||||
| 1720 | 1700-1990 | GSM 1800. CDMA.1900.GSM 1900. DECT.LTE Nand 1, 3.4, 25; UTMS. | Pulse Modulation^b 217 Hz | 2 0.3 28 | ||
| 1845 | ||||||
| 1970 | ||||||
| 2450 2400-2570 | Bluetooth WLAN.802.11 b/g/n.RFID 2450.LTE Band 7. | Pulse Modulation^b 217 Hz | 2 0.3 28 | |||
| 5240 | 5100-5800 WLAN 802. | 11 a/n. | Pulse Modulation^b 217 Hz | 0.2 0.3 95500 | ||
| 5785 | ||||||
| NOTE If necessary to achieve the IMMUNITY TEST LEVEL, the distance between the transmitting antenna and the ME EQUIPMENT or ME SYSTEM may be reduced to 1m. The 1m test distance is permitted by IEC 61000-4-3. | ||||||
| a) For some services, only the uplink frequencies are includedb) the carrier shall be modulated using a 50% duty cycle square wave signal.c) As an alternative to FM modulation at 18 Hz may be used because while it does not represent actual modulation, it would be worst case. | ||||||
HOW THE DEVICE WORKS
The device has multiple modes, covering TENS and EMS. If you are using the device for the first time, it is recommended that you start with the default Mode 1, which combines different pulse frequencies. Some modes are particularly effective for certain users, but you may need to select the mode that is best for you. You may need to try a few modes before finding the one that suits you the best. The best choice of modes may vary from one user to another, in spite of having the same type of symptom.
| Pulse frequency (Hz) MODE DESCRIPTION INTENDED USE | |||
| Mode 1 Combination of Mode 2-8 Combination of below | Pain relief and muscle stimulation with combined pulse rates | ||
| Mode 2 69 Hz | Pulse on for 3.4 second and off for 1.6 second | Pain relief with a pulse rate of 69 Hz | |
| Mode 3 12.5-55.5 Hz | Pulse on for 20 second and off for 1 second | Muscle stimulation with variable pulse rate | |
| Mode 4 1.2 Hz | Pulse on every 0.85 second | Pain relief with a pulse rate of 1.2 Hz | |
| Mode 5 100 Hz | Pulse on for 10 second and off for 2.5 second | Pain relief with a pulse rate of 100 Hz | |
| Mode 6 100 Hz | Pulse on for 20 second and off for 1 second | Pain relief with a pulse rate of 100 Hz and longer on time | |
| Mode 7 20 Hz | Pulse on for 5 second and off for 1 second | Muscle stimulation with a fixed pulse rate of 20 Hz | |
| Mode 8 160 Hz | Pulse on for 10 second and off for 2 second | Pain relief with a pulse rate of 160 Hz | |
DEVICE DESCRIPTION
Remove the device and accessories from the packaging. The accessories include a USB cable used for charging, and a wrap with conductive silver pads.
Accessories included in the package.
1x Tens unit controller
1x Wrap
1x USB line
1x Manual
1x Bottle
text_image
BATTERY INDICATOR MODE CHANGE M + T +/-HEAT TIMER CHANGE HEAT LEVEL INTENSITY INCREASE INTENSITY DECREASE
text_image
CHARGING PORT OleMICRO USB TO USB CHARGER
ON / OFF: HOLD 2 SEC.
USE ON/OFF BUTTON TO
LOCK SCREEN
The following steps are used to guide the device operation, and the details about each step are listed in the following table.
01 Charge control unit before first use
02 Lightly dampen conductive silver pads with water
03 Apply brace to body part
NOTE: Skin should be clean. Remove sweat and lotions prior to use.
04 Connect control unit to brace
Note: Connect gold magnets to gold connectors, silver magnets to silver connectors.
The screen should be legible from the user looking down.
05 Turn control unit on (Hold 2 sec.)
06 Select program
07 Change simulation time
08 Adjust stimulation intensity
09 Adjust heat level
10 Turn control unit off when done
NOTE: Intensity + button only works when device is attached to wrap and wrap is in contact with skin.
| 1st Step - Charge control unit before first use |  |  |
| The control unit comes with a built-in rechargeable battery, and can be used as received. If the battery icon on the turned-on control unit keeps flashing, it means the battery is running out. Charge the control unit with the enclosed USB cable. The battery icon flashes during charging, and becomes solid when the control unit is charged fully. | CONTROL UNIT USB CABLE | |
| 2nd Step - Dampen wrap pads for best results |  | |
| Lightly dampen conductive silver pads with clean water | ||
| 3rd Step - Apply to the body part |  |   |
| Apply wrap to the body part using velcro strap/s to secure in place | ||
| 4th Step - Connect control unit to the brace | MAGNETS | |
| Apply control unit to brace using magnets to secure connectors. Connect gold magnets to gold connectors, and silver magnets to silver connectors. The screen should be right reading from the user looking down. | ||
| 5th Step - Turn control unit on | ON/OFF BUTTON | |
| Hold On/Off button for 2 seconds to turn on device. Front display panel will light-up, indicating the device is on. Press the On/Off button once to lock/unlock the screen buttons. When buttons are locked, no adjustments can be made. Hold On/Off button for 2 seconds to turn off the device. | ||
| 6th Step - Select program | ||
| There are eight stimulation modes. Press the “M” button to select a desired pulse mode. The mode selected will be shown on the display. |  | M = MODE CHANGE |
| 7th Step - Change simulation time | ||
| Default timer is set to 30 min. Press the “T” button to select a desired time (30, 40, 50, 60, 10 and 20 min) interval. The time selected will be shown on the display screen. |  | T = TIMER CHANGE |
| 8th Step - Adjust stimulation intensity | ||
| Press and release the “+” button to increase the intensity, and the “-” button to decrease the intensity. The device will beep and the intensity level will flash with each change. Note: With the increase of intensity (20 total levels), you may experience sensations like tingling, vibration, pain, etc. Therefore, gradually increase the intensity, and stop increasing when a comfortable level is reached. | 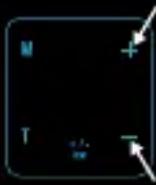 | + = INTENSITY INCREASE- = INTENSITY DECREASE |
| 9th Step - Adjust heat level | ||
| Press the HEAT +/- button to select desired level of heat. |  | HEAT + / - =HEAT OFF, LEVEL 1, LEVEL 2 |
| 10th Step - Turn control unit off when done. | ||
| When the countdown timer is up, the device will turn off automatically. The device can also be turned off by holding the On/Off button, indicated by the display light turning off. | OFF / ON | |
MAGNETS
When using the device for the first time, we recommend starting from the default Mode 1, which combines the different frequencies. If you use one of the specific TENS or EMS modes, please refer to the following for details.
USE AS TENS
The control unit includes the following TENS frequencies, 69Hz (Mode 2), 1.2 Hz (Mode 4), 100Hz (Modes 5 and 6), and 160Hz (Mode 8). For treatment, place the wrap and conductive silver pads at the site of pain, and connect the device to the wrap.
For arthritis pain, inflammation of the joints, place the conductive silver pads on or near the area of the arthritis pain. The TENS mode of the device generates electrical pulses that are sent through the conductive silver pads for pain relief.
USE AS EMS
The control unit also includes the EMS frequencies, 12.5-55.6Hz (Mode 3) and 20Hz (Mode 7). For treatment, place the wrap and conductive silver pads where you desired muscle firming and strengthening, and connect the device to the wrap. Then select modes 3 or 7.
Recommended Practice for Both TENS and EMS:
Start from the lowest intensity and gradually adjust the intensity to a comfortable level at a scale from from 1 to 20.
Good skin care is important for a comfortable use of device. Be sure the treatment site is clean of dirt and body lotion.
CONDUCTIVE WRAP
The conductive wrap accessory is connected to the control unit through its magnentic connectors. The wrap accessory holds the conductive silver pad material that will be in contact with the skin. The electrical stimulation will be delivered to the body through these pads.
PERFORMANCE SPECIFICATIONS
Power Source 3.7V Battery
Number of Output Modes 8 auto pulse modes
Timer Range (minutes) 10-60
Dimensions (mm) [L x W x D] 89 x 77 x 18 mm
Waveform Biphasic
Shape Rectangular
Maximum Output Voltage 64V@500Ω
Maximum Output Current 128mA@500Ω
Pulse Duration 100μSec
Maximum Frequency 160Hz
CLEANING AND MAINTENANCE
Hand wash in warm water using mild soap, rinse thoroughly. Air dry.
Recharging the device at least once every three months if not used for a long time.
TROUBLE SHOOTING
If your device is not operating properly, please check below for common problems and suggested solutions. If the recommended action does not solve the problem, please contact the seller.
STIMULATION IS WEAK OR NON-EXISTENT
- Be sure skin is clean and pads are firmly attached to skin.
- Be sure to lightly dampen the conductive silver pads for best results.
- Check if the battery is low and needs to be recharged.
DEVISE DOES NOT TURN ON
- Check if battery is low and needs to be charged.
SKIN TURNS RED
- Stop treatment.
• If problem persists, contact your physician.
WARRANTY
Within two years from the date of purchase, if the product fails due to a defect in material or workmanship, Compex will repair or replace the product, or necessary components, free of charge.
THIS WARRANTY EXCLUDES:
- Battery
• Damage caused by abuse and/or mishandling
• Repairs not carried out by the manufacturer
• Devices not used in accordance with the care instructions - Abnormal storage
- Failure to provide proof of purchase
CONTACT INFORMATION
Distributed by
DJO France S.A.S
Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com
Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
FR
TABLE DES MATIÈRES
Introduction......26
Contre-indications....27
Avertissements....27
Précautions....28
LA STIMULATION EST FAIBLE OU INEXISTANTE
Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com

Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
ES
ÍNDICE DE CONTENIDO
| Test Frequency (MHz) | Band^a | Service^a | Modulation^a | Maximum Power (W) | Distance (M) | Immunity Test Level (V/m) |
| 385 380-390 TETRA 400. Pulse Modulation | b18Hz | 1.8 0.3 27 | ||||
| 450 430-470 GMRS 460. FRS 460. FM | c+5kHz sine 2 0.3 28 | |||||
| 710 | 704-787 LTE Band 13. 17. | Pulse Modulation^b 217 Hz | 0.2 0.3 9745 | |||
| 780 | ||||||
| 810 | 800-960 | GSM 800/900.TETRA 800. iDEN 820.CDMA 850. LTE.Nand 5. | Pulse Modulation^b 18 Hz | 2 0.3 28 | ||
| 870 | ||||||
| 930 | ||||||
| 1720 | 1700-1990 | GSM 1800. CDMA.1900.GSM 1900. DECT.LTE Nand 1, 3.4, 25; UTMS. | Pulse Modulation^b 217 Hz | 2 0.3 28 | ||
| 1845 | ||||||
| 1970 | ||||||
| 2450 2400-2570 | Bluetooth WLAN.802.11 b/g/n.RFID 2450.LTE Band 7. | Pulse Modulation^b 217 Hz | 2 0.3 28 | |||
| 5240 | 5100-5800 WLAN 802. | 11 a/n. | Pulse Modulation^b 217 Hz | 0.2 0.3 95500 | ||
| 5785 | ||||||
| NOTE If necessary to achieve the IMMUNITY TEST LEVEL, the distance between the transmitting antenna and the ME EQUIPMENT or ME SYSTEM may be reduced to 1m. The 1m test distance is permitted by IEC 61000-4-3. | ||||||
| a) For some services, only the uplink frequencies are includedb) the carrier shall be modulated using a 50% duty cycle square wave signal.c) As an alternative to FM modulation at 18 Hz may be used because while it does not represent actual modulation, it would be worst case. | ||||||
CÓMO FUNCIONA EL DISPOSITIVO
Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com

Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
IT
SOMMARIO
Introduzione....70
| Test Frequency (MHz) | Band^a | Service^a | Modulation^a | Maximum Power (W) | Distance (M) | Immunity Test Level (V/m) |
| 385 380-390 TETRA 400. Pulse Modulation | b18Hz | 1.8 0.3 27 | ||||
| 450 430-470 GMRS 460. FRS 460. FM | c+5kHz sine 2 0.3 28 | |||||
| 710 | 704-787 LTE Band 13. 17. | Pulse Modulation^b 217 Hz | 0.2 0.3 9745 | |||
| 780 | ||||||
| 810 | 800-960 | GSM 800/900.TETRA 800. iDEN 820.CDMA 850. LTE.Nand 5. | Pulse Modulation^b 18 Hz | 2 0.3 28 | ||
| 870 | ||||||
| 930 | ||||||
| 1720 | 1700-1990 | GSM 1800. CDMA.1900.GSM 1900. DECT.LTE Nand 1, 3.4, 25; UTMS. | Pulse Modulation^b 217 Hz | 2 0.3 28 | ||
| 1845 | ||||||
| 1970 | ||||||
| 2450 2400-2570 | Bluetooth WLAN.802.11 b/g/n.RFID 2450.LTE Band 7. | Pulse Modulation^b 217 Hz | 2 0.3 28 | |||
| 5240 | 5100-5800 WLAN 802. | 11 a/n. | Pulse Modulation^b 217 Hz | 0.2 0.3 95500 | ||
| 5785 | ||||||
| NOTE If necessary to achieve the IMMUNITY TEST LEVEL, the distance between the transmitting antenna and the ME EQUIPMENT or ME SYSTEM may be reduced to 1m. The 1m test distance is permitted by IEC 61000-4-3. | ||||||
| a) For some services, only the uplink frequencies are includedb) the carrier shall be modulated using a 50% duty cycle square wave signal.c) As an alternative to FM modulation at 18 Hz may be used because while it does not represent actual modulation, it would be worst case. | ||||||
Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com
Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
DE
INHALTSVERZEICHNIS
Einführung....92
Indikationen 92
| Test Frequency (MHz) | Band^a | Service^a | Modulation^a | Maximum Power (W) | Distance (M) | Immunity Test Level (V/m) |
| 385 380-390 TETRA 400. Pulse Modulation | b18Hz | 1.8 0.3 27 | ||||
| 450 430-470 GMRS 460. FRS 460. FM | c+5kHz sine 2 0.3 28 | |||||
| 710 | 704-787 LTE Band 13. 17. | Pulse Modulation^b 217 Hz | 0.2 0.3 9745 | |||
| 780 | ||||||
| 810 | 800-960 | GSM 800/900.TETRA 800. iDEN 820.CDMA 850. LTE.Nand 5. | Pulse Modulation^b 18 Hz | 2 0.3 28 | ||
| 870 | ||||||
| 930 | ||||||
| 1720 | 1700-1990 | GSM 1800. CDMA.1900.GSM 1900. DECT.LTE Nand 1, 3.4, 25; UTMS. | Pulse Modulation^b 217 Hz | 2 0.3 28 | ||
| 1845 | ||||||
| 1970 | ||||||
| 2450 2400-2570 | Bluetooth WLAN.802.11 b/g/n.RFID 2450.LTE Band 7. | Pulse Modulation^b 217 Hz | 2 0.3 28 | |||
| 5240 | 5100-5800 WLAN 802. | 11 a/n. | Pulse Modulation^b 217 Hz | 0.2 0.3 95500 | ||
| 5785 | ||||||
| NOTE If necessary to achieve the IMMUNITY TEST LEVEL, the distance between the transmitting antenna and the ME EQUIPMENT or ME SYSTEM may be reduced to 1m. The 1m test distance is permitted by IEC 61000-4-3. | ||||||
| a) For some services, only the uplink frequencies are includedb) the carrier shall be modulated using a 50% duty cycle square wave signal.c) As an alternative to FM modulation at 18 Hz may be used because while it does not represent actual modulation, it would be worst case. | ||||||
FUNKTIONSWEISE DES GERÄTS
natural_image
Blue ribbon cable illustration on black background (no text or symbols)natural_image
Three line drawings of a hand holding a spray gun, with no text or symbols present.
natural_image
Simple diagram with four circles and a white arrow pointing to the top circle (no text or symbols)MAGNETS

text_image
ON/OFF BUTTON A#Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com
Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
NL
INHOUD
Introductie 114
Contra-indicaties....115
VEILIGHEIDSWAARSCHUWING
CONTRA-INDICATIES
| Test Frequency (MHz) | Band^a | Service^a | Modulation^a | Maximum Power (W) | Distance (M) | Immunity Test Level (V/m) |
| 385 380-390 TETRA 400. Pulse Modulation | b18Hz | 1.8 0.3 27 | ||||
| 450 430-470 GMRS 460. FRS 460. FM | c+5kHz sine 2 0.3 28 | |||||
| 710 | 704-787 LTE Band 13. 17. | Pulse Modulation^b 217 Hz | 0.2 0.3 9745 | |||
| 780 | ||||||
| 810 | 800-960 | GSM 800/900.TETRA 800. iDEN 820.CDMA 850. LTE.Nand 5. | Pulse Modulation^b 18 Hz | 2 0.3 28 | ||
| 870 | ||||||
| 930 | ||||||
| 1720 | 1700-1990 | GSM 1800. CDMA.1900.GSM 1900. DECT.LTE Nand 1, 3.4, 25; UTMS. | Pulse Modulation^b 217 Hz | 2 0.3 28 | ||
| 1845 | ||||||
| 1970 | ||||||
| 2450 2400-2570 | Bluetooth WLAN.802.11 b/g/n.RFID 2450.LTE Band 7. | Pulse Modulation^b 217 Hz | 2 0.3 28 | |||
| 5240 | 5100-5800 WLAN 802. | 11 a/n. | Pulse Modulation^b 217 Hz | 0.2 0.3 95500 | ||
| 5785 | ||||||
| NOTE If necessary to achieve the IMMUNITY TEST LEVEL, the distance between the transmitting antenna and the ME EQUIPMENT or ME SYSTEM may be reduced to 1m. The 1m test distance is permitted by IEC 61000-4-3. | ||||||
| a) For some services, only the uplink frequencies are includedb) the carrier shall be modulated using a 50% duty cycle square wave signal.c) As an alternative to FM modulation at 18 Hz may be used because while it does not represent actual modulation, it would be worst case. | ||||||
HOE HET APPARAAT WERKT
natural_image
Simple blue ribbon illustration on black background (no text or symbols)BEDIENINGSEENHEID USB-KABEL

natural_image
Three line drawings of hands performing a manual task: spraying, holding a tool, and holding a rolled-up object (no text or symbols)
natural_image
Black square with four small circular icons and a white arrow pointing to the top-right circle (no text or symbols)MAGNETS

text_image
ON/OFF BUTTON AidHET APPARAAT GAAT NIET AAN
Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com
Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
SV
INNEHÅLLSFÖRTECKNING
Inledning....136
| Test Frequency (MHz) | Band^a | Service^a | Modulation^a | Maximum Power (W) | Distance (M) | Immunity Test Level (V/m) |
| 385 380-390 TETRA 400. Pulse Modulation | b18Hz | 1.8 0.3 27 | ||||
| 450 430-470 GMRS 460. FRS 460. FM | c+5kHz sine 2 0.3 28 | |||||
| 710 | 704-787 LTE Band 13. 17. | Pulse Modulation^b 217 Hz | 0.2 0.3 9745 | |||
| 780 | ||||||
| 810 | 800-960 | GSM 800/900.TETRA 800. iDEN 820.CDMA 850. LTE.Nand 5. | Pulse Modulation^b 18 Hz | 2 0.3 28 | ||
| 870 | ||||||
| 930 | ||||||
| 1720 | 1700-1990 | GSM 1800. CDMA.1900.GSM 1900. DECT.LTE Nand 1, 3.4, 25; UTMS. | Pulse Modulation^b 217 Hz | 2 0.3 28 | ||
| 1845 | ||||||
| 1970 | ||||||
| 2450 2400-2570 | Bluetooth WLAN.802.11 b/g/n.RFID 2450.LTE Band 7. | Pulse Modulation^b 217 Hz | 2 0.3 28 | |||
| 5240 | 5100-5800 WLAN 802. | 11 a/n. | Pulse Modulation^b 217 Hz | 0.2 0.3 95500 | ||
| 5785 | ||||||
| NOTE If necessary to achieve the IMMUNITY TEST LEVEL, the distance between the transmitting antenna and the ME EQUIPMENT or ME SYSTEM may be reduced to 1m. The 1m test distance is permitted by IEC 61000-4-3. | ||||||
| a) For some services, only the uplink frequencies are includedb) the carrier shall be modulated using a 50% duty cycle square wave signal.c) As an alternative to FM modulation at 18 Hz may be used because while it does not represent actual modulation, it would be worst case. | ||||||
HUR ENHETEN FUNGERAR
Timerintevall (minuter) 10–60
Dimensioner (mm) [L x B x D] 89 x 77 x 18 mm
Vågform Bifasisk
Form Rektangulär
Maximal utspänning 64V@500Ω
Maximal uteffekt 128mA@500Ω
Pulsens varaktighet 100μSek
Maximal frekvens 160 Hz
RENGÖRING OCH UNDERHÅLL
Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com

Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
FI
SISÄLLYSLUETTELO
Johdanto 158
Käyttöaiheet....158
SYMBOLIT JA MERKITYKSET
| Test Frequency (MHz) | Band^a | Service^a | Modulation^a | Maximum Power (W) | Distance (M) | Immunity Test Level (V/m) |
| 385 380-390 TETRA 400. Pulse Modulation | b18Hz | 1.8 0.3 27 | ||||
| 450 430-470 GMRS 460. FRS 460. FM | c+5kHz sine 2 0.3 28 | |||||
| 710 | 704-787 LTE Band 13. 17. | Pulse Modulation^b 217 Hz | 0.2 0.3 9745 | |||
| 780 | ||||||
| 810 | 800-960 | GSM 800/900.TETRA 800. iDEN 820.CDMA 850. LTE.Nand 5. | Pulse Modulation^b 18 Hz | 2 0.3 28 | ||
| 870 | ||||||
| 930 | ||||||
| 1720 | 1700-1990 | GSM 1800. CDMA.1900.GSM 1900. DECT.LTE Nand 1, 3.4, 25; UTMS. | Pulse Modulation^b 217 Hz | 2 0.3 28 | ||
| 1845 | ||||||
| 1970 | ||||||
| 2450 2400-2570 | Bluetooth WLAN.802.11 b/g/n.RFID 2450.LTE Band 7. | Pulse Modulation^b 217 Hz | 2 0.3 28 | |||
| 5240 | 5100-5800 WLAN 802. | 11 a/n. | Pulse Modulation^b 217 Hz | 0.2 0.3 95500 | ||
| 5785 | ||||||
| NOTE If necessary to achieve the IMMUNITY TEST LEVEL, the distance between the transmitting antenna and the ME EQUIPMENT or ME SYSTEM may be reduced to 1m. The 1m test distance is permitted by IEC 61000-4-3. | ||||||
| a) For some services, only the uplink frequencies are includedb) the carrier shall be modulated using a 50% duty cycle square wave signal.c) As an alternative to FM modulation at 18 Hz may be used because while it does not represent actual modulation, it would be worst case. | ||||||
KUINKA LAITE TOIMII
Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com

Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
DA
INDHOLDSFORTEGNELSE
Indledning....180
Indikationer for brug....180
Sikkerhedsadvarsel....187
Kontraindikationer....187
Advarsler....187
Forholdsregler....182
Bivirkninger....184
Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com
Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
NO
INNHOLD
Innledning....202
MILJ∅FORHOLD FOR TRANSPORT OG LAGRING
Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com

Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
PT
ÍNDICE
Introdução....224
| Test Frequency (MHz) | Band^a | Service^a | Modulation^a | Maximum Power (W) | Distance (M) | Immunity Test Level (V/m) |
| 385 380-390 TETRA 400. Pulse Modulation | b18Hz | 1.8 0.3 27 | ||||
| 450 430-470 GMRS 460. FRS 460. FM | c+5kHz sine 2 0.3 28 | |||||
| 710 | 704-787 LTE Band 13. 17. | Pulse Modulation^b 217 Hz | 0.2 0.3 9745 | |||
| 780 | ||||||
| 810 | 800-960 | GSM 800/900.TETRA 800. iDEN 820.CDMA 850. LTE.Nand 5. | Pulse Modulation^b 18 Hz | 2 0.3 28 | ||
| 870 | ||||||
| 930 | ||||||
| 1720 | 1700-1990 | GSM 1800. CDMA.1900.GSM 1900. DECT.LTE Nand 1, 3.4, 25; UTMS. | Pulse Modulation^b 217 Hz | 2 0.3 28 | ||
| 1845 | ||||||
| 1970 | ||||||
| 2450 2400-2570 | Bluetooth WLAN.802.11 b/g/n.RFID 2450.LTE Band 7. | Pulse Modulation^b 217 Hz | 2 0.3 28 | |||
| 5240 | 5100-5800 WLAN 802. | 11 a/n. | Pulse Modulation^b 217 Hz | 0.2 0.3 95500 | ||
| 5785 | ||||||
| NOTE If necessary to achieve the IMMUNITY TEST LEVEL, the distance between the transmitting antenna and the ME EQUIPMENT or ME SYSTEM may be reduced to 1m. The 1m test distance is permitted by IEC 61000-4-3. | ||||||
| a) For some services, only the uplink frequencies are includedb) the carrier shall be modulated using a 50% duty cycle square wave signal.c) As an alternative to FM modulation at 18 Hz may be used because while it does not represent actual modulation, it would be worst case. | ||||||
COMO FUNCIONA O DISPOSITIVO
Address: 4-5F, Building 12, Hengmingzhu Industrial Park, Xinqiao Tongfuyu Industrial
Area, Shajing, Baoan District, Shenzhen, China
E-mail: info@JKHhealth.com

Shanghai International Holding Corp. GmbH (Europe)
Eiffestrasse 80, 20537 Hamburg, Germany
CE 1639
INTL-CX182IF01 REV A





