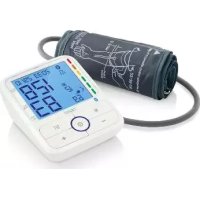
IAN 290990 - Blood pressure monitor SILVERCREST - Free user manual and instructions
Find the device manual for free IAN 290990 SILVERCREST in PDF.
User questions about IAN 290990 SILVERCREST
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Blood pressure monitor in PDF format for free! Find your manual IAN 290990 - SILVERCREST and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. IAN 290990 by SILVERCREST.
USER MANUAL IAN 290990 SILVERCREST
Instructions for Use and Safety Notes
FR BE
tensiOmotrepOur avant-bras
Before reading, unfold both pages containing illustrations and familiarise yourself with all functions of the device.
FR B
GB/IE Instructions for Use and Safety Notes Page 45
Link: www.healthforyou.lidl
Intended Use... Page 48
Improper Use.. 48
Parts Description.. Page 49
Scope of delivery.. Page 50
Technical Data.. 50
Important Safety Notes.. Page 53
Battery safety information.. 55
Initial use.. Page 57
App / web version of „HealthForYou“ ..... Page 58
Configuring the date/time/bluetooth Page 58
Measuring blood pressure... Page 59
Attaching the cuff.. Page 60
Adopting the correct posture.. Page 60
Selecting a memory.. Page 61
Carrying out the blood pressure measurement Page 61
Transferring measurements via Bluetooth® .......Page 63
Evaluating Measurements.. Page 64
Classification of measurement results.. Page 65
Saving, requesting and deleting
measurements Page 66
Cleaning and Care.. Page 68
Storage 68
Troubleshooting.. Page 69
Disposal Page 70
Warranty/Service Page 71
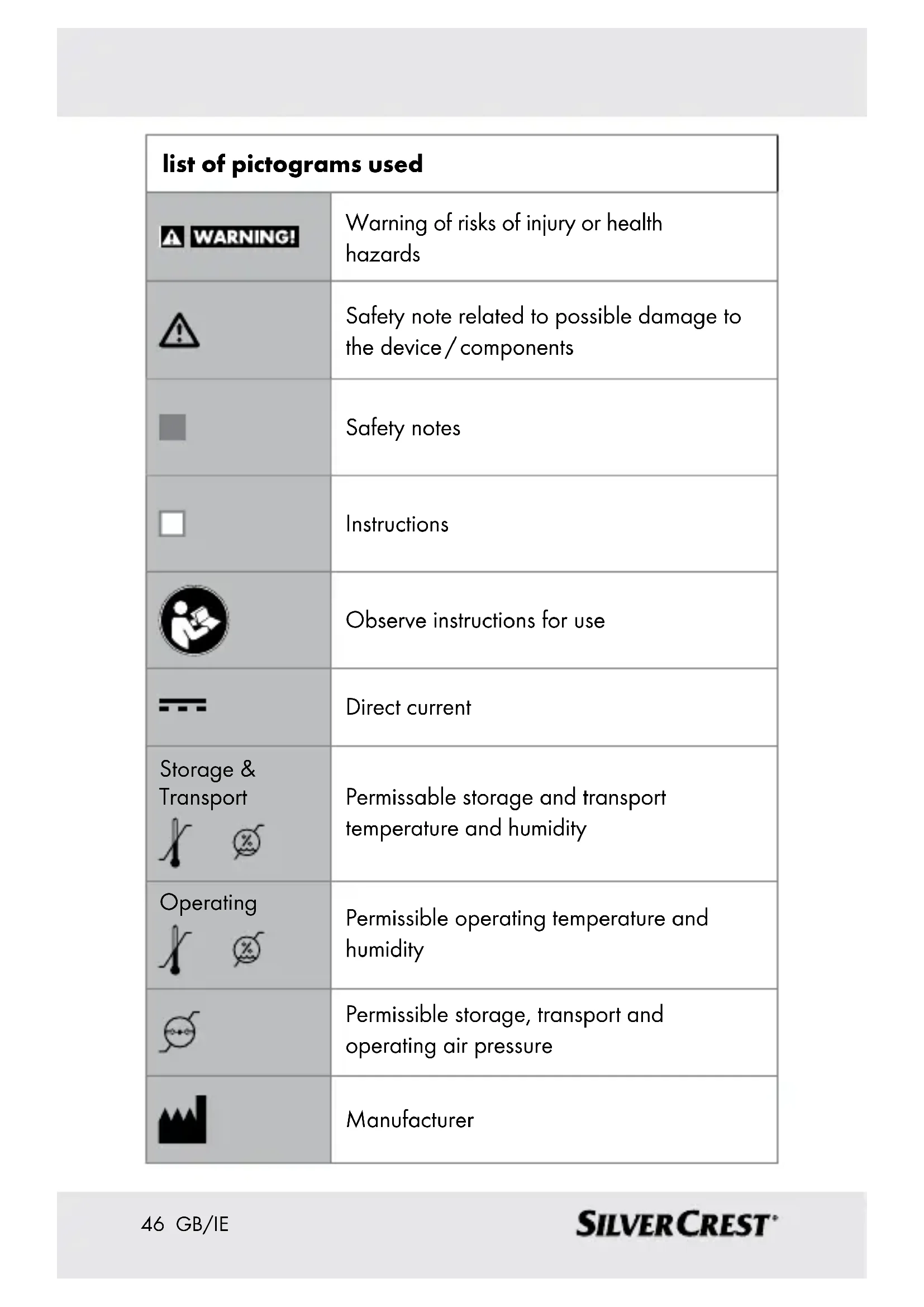
| list of pictograms used | |
| WARNING! | Warning of risks of injury or health hazards |
| Safety note related to possible damage to the device / components | |
| Safety notes | |
| Instructions | |
| Observe instructions for use | |
| Direct current | |
| Storage & Transport | Permissible storage and transport temperature and humidity |
| Operating | Permissible operating temperature and humidity |
| Permissible storage, transport and operating air pressure | |
| Manufacturer | |
| Applied part, type BF | |
| SN | Serial number |
| CE 0483 | The CE mark certifies compliance with the basic requirements of Medical Devices Directive 93/42/EEC. |
| Improper disposal of batteries can harm the environment! | |
| IP21 | Device protected against foreign matter ≥12.5 mm and against water falling vertically |
| Dispose of the packaging and the blood pressure monitor in an environmentally friendly manner! | |
Upper arm blood pressure monitor
Introduction

Please make yourself familiar with the upper arm blood pressure monitor before preparing it for use or using it for the first time. Please carefully
read the following instructions for use and the important safety notes. Failure to observe the notes may result in personal injury or property damage. The product must only be
used as described in these instructions for use and for the specified fields of application. Keep these instructions for use in a safe place. If you pass the upper arm blood pressure monitor on to anyone else, please ensure that you also pass on all the documentation.
intendeduse
This upper arm blood pressure monitor is intended to measure and monitor the arterial blood pressure values of adults in a non-invasive way.
Using this device, you can quickly and easily measure your blood pressure, save the measurements and display the progression and average of these measurements. You are warned in cases of pre-existing cardiac arrhythmia. The values determined are classified and evaluated graphically. Any use other than previously mentioned or any product modification is prohibited and may result in injuries and / or product damage. The manufacturer assumes no liability for damage arising out of improper use.
The product is only intended for use in private environments (at home).
improperuse
WARNING! Do not use this product on newborns and preeclampsia patients. We recommend consulting your doctor before using the product if you are pregnant.
The measurements determined by you are only intended for your information - they do not replace a medical examination. Discuss the measurements with your doctor. Do not self-diagnose or self-treat based on the measurement results without consulting your attending physician. Do not start new medication without consulting a doctor and do not modify the type and / or dosage of current medications.
The product may not be used in conjunction with a high-frequency surgical unit.
-PartsDescription
1 Cuff
2 Cuff tubing
3 Cuff plug
4 Display
5 Memory button M
6 Settings button SET
7 Function button - / + 8 START/STOP button 1
9 Scale for classifying measurement results
10 Connection for cuff plug
11 Battery compartment
Display indicators
12 Time and date 13 Systolic pressure 14 Diastolic pressure
15 Release air
16 Pulse value determined
17 Classification of measurement results
Symbol for cardiac arrhythmia Symbol pulse
19 Memory display: Average value (A), morning (AM), evening (PM), number of the memory space
20 Symbol for replace battery
21 User memory
22 Symbol for Bluetooth® transfer
- Scope of delivery
1 Upper arm blood pressure monitor
1 Upper arm cuff (Material no. 163.243) with cuff tubing
4 Batteries 1.5 V Type AA
1 Instructions for use
1 Quick start guide
-TechnicalData
Model no.: SBM 68
Measuring method Oscillometric, non-invasive blood pressure measurement on the upper arm
Measurement range: Cuff pressure 0-300 mmHg, systolic 50-250 mmHg, diastolic 30-200 mmHg, pulse 40-180 beats per minute
Display accuracy: systolic ± 3 mmHg, diastolic
± 3 mmHg , pulse ± 5 % of the
displayed value
Measurement
uncertainty: max. permissible standard deviation
according to clinical testing: systolic
8 mmHg / diastolic 8 mmHg
Memory: 4 × 30 memory spaces
Dimensions: L 131 mm x W 107 mm x H 56 mm
Cuff: suitable for an arm circumference
between 22 and 36~cm
Weight: approx. 282 g (without batteries)
and cuff)
Permissible
operating conditions: +5^ to +40^ , 15 to 93% rela
tive humidity (non-condensing)
Permissible storage
conditions: -25 °C to +70 °C, < 93% relative
humidity, 700-1060 hPa ambient
pressure
Power supply: 4 × 1.5 ~V AA batteries
Product expected
lifespan: 5 years
Battery life:
Approx. 200 measurements, depending on blood pressure level and inflation pressure/Bluetooth® transfer
Components:
Instructions for use, 4 × 1.5 ~V AA, batteries, storage pouch
Classification: Internal supply, IP21, no AP or APG, continuous operation, applied part Type BF
Data transfer via Bluetooth®
wireless technology: The product uses Bluetooth® low energy technology, frequency band 2.402 - 2.480 GHz, maximum transmission power radiated in the frequency band < 4 dBm, compatible with Bluetooth® 4.0 smart phones / tablets

For a list of compatible smart phones, information about the HealthForYou app and software, as well as further details about the devices, please visit: www.healthforyou.lidl
System requirements
of the ,HealthForYou"
web version -MS Internet Explorer MS Edge Mozilla Firefox Google Chrome Apple Safari (in the respective updated version)
System requirements of the „HealthForYou“
app iOS from Version 9.0, Android TM from Version 5.0, Bluetooth® 4.0.
Technical specifications subject to change without notice due to updates.

Important Safety Notes
WARNING! This device is not intended to be used by persons with limited physical, sensory or mental abilities or lacking experience and / or knowledge unless supervised or instructed on its use by an person responsible for their safety. Children should be supervised to ensure they do not play with the device.
Cardiovascular system diseases may result in incorrect measurements and/or impairments to the device's measurement accuracy. This is also the case when it comes to low blood pressure, diabetes, circulatory problems and dysrhythmia, as well as chills and shaking.
Only use the product on individuals who have the upper arm range stated for the product.
Ensure that the limb is not functionally impaired during inflation.
- Blood circulation should not be restricted by the blood pressure measurement for an unnecessary amount of time. Remove the cuff from the arm if the device malfunctions.
- Avoid mechanically restricting, compressing or kinking the cuff tubing.
- Avoid sustained pressure to the cuff, as well as frequent measurements. The resulting curtailing of the blood flow can lead to injuries.
Ensure that the cuff is not placed on an arm whose arteries or veins are undergoing medical treatment, i.e. intravascular access or intravascular therapy or an arteriovenous (A-V) shunt. - Do not place the cuff on individuals who have had a mastectomy.
- Do not place the cuff over wounds as this could lead to further injuries.
Only place the cuff on the upper arm. Do not place the cuff on other body parts.
Attention: The device may only be used in conjunction with the original cuff provided.
Do not use rechargeable batteries.
If no button is pressed, the automatic shutoff will switch off the blood pressure monitor within 1 minute to save the batteries.
Attention! Potential damage to the device! The blood pressure monitor is made up of precision components and electronic components. The accuracy of the measurements and the service life of the device depend on the device being used with a due amount of care:
- Protect the device from impacts, moisture, dirt, severe temperature fluctuations and direct sunlight.
Do not drop the device.
- Do not use the device in the vicinity of strong electromagnetic fields, keep it away from radio equipment and mobile telephones.
Only use the cuff provided or an original replacement cuff. Otherwise, measurements could be miscalculated.
Remove the batteries from the device if it is not used over a longer period of time.
- Routinely inspect the device to ensure the device is not visibly damaged before use and the battery level is sufficient. When in doubt, do not use and contact service or an authorised retailer. Do not use additional parts not recommended or sold as components by the manufacturer.
- Never open or repair the device or proper function cannot be guaranteed. Non-compliance will void the warranty.
Please contact service or an authorised retailer for repairs. However, before making a complaint, firstly check the batteries and replace them if necessary.

Battery safety information
DANGER TO LIFE! Keep batteries out of the reach of children. Seek immediate medical attention if swallowed!

XPLOSION HAZARD! Never recharge
disposable batteries. Never short-circuit and / or open batteries. The battery may overheat, explode.
Never throw batteries into fire or water.
Do not exert mechanical stress on batteries.
Risk of battery leakage
Avoid extreme conditions and temperatures which may affect batteries, e.g. radiators / direct sunlight.
Avoid contact with the skin, eyes and mucous membranes! In the event of contact with battery acid, flush the affected area with clean water and seek immediate medical attention!

Wear Protective Gloves! Leaked or damaged batteries can cause burns on contact with the skin. Therefore wear suitable safety gloves in this case.
In the event of a battery leak, immediately remove the batteries from the product to prevent damage.
Only use the same type of batteries. Do not mix old and new batteries!
Remove the batteries if the product will not be used for an extended period.
Risk of product damage
Only use the specified battery type!
Insert the batteries according to the polarity marking (+) and (-) on the battery and the product.
Clean the contacts on the battery and inside the battery compartment with a dry cloth before inserting the battery!
- Immediately remove drained batteries from the product.
Do not use rechargeable batteries.
- Initial use
Verify the parts listed in the scope of delivery are complete.
Remove all packaging material.
- Inserting/Replacing Batteries
Slide the battery cover off the battery compartment (Fig. C).
Insert the four type 1.5V AA (Alkaline Type LR6) batteries (with the correct polarity) into the device (Fig. C). Please note the marking inside the battery compartment.
Carefully close the battery cover on the battery compartment [11].
Do not use rechargeable batteries.
Note: If the battery indic. is displayed continuously, measurement is not possible and you must replace all of the batteries with new batteries. As soon as the batteries have been removed from the device, the date and time will be reset.
□ All display elements will be displayed briefly and ^ 24h^ will flash on the display. Configure the date, time and Bluetooth® (see „Configuring date/ time/Bluetooth®").
App/web version of „HealthForYou“
Download the free app „HealthForYou“ from the Apple App Store and Google Play.
Follow the installation instructions on the screen.
□ Alternatively, you can also visit the website www.healthforyou.lidl. Here you will be able to enter measurements and find guidance for assessment.

- Configuring the date / time / bluetooth®
Configure the date, hour format and time. It is only in this way that you will be able to correctly save these measurements with the date and time and request them later on.
Configure the date, Bluetooth® and time as follows:
Insert the batteries, or press the SET button for over 5 seconds for batteries that have already been inserted.
□ Configure the 12-hour or 24-hour mode using the 7 button. Press the SET button 6 to confirm.
The year will flash.
Configure the year using the + button. Press the SET button 6 to confirm.
The month will flash.
□ Configure the month using the ^+ button. Press the SET button 6 to confirm.
Configure the day, hour and minute consecutively as described above. Press the SET 6 button to confirm each entry.
TheBluetooth symbol will be shown on the display. "On" or "Off" will flash at the same time.
Using the function buttons / + " choose whether you want to activate (,0,n^ flashes) or deactivate (,0,f off flashes") automatic Bluetooth® data transfer. Press the SET button 6 to confirm.
IfBluetooth ® is activated (_ ^ ) data transfer will begin automatically after the measurement has been taken.
Note: The battery life is reduced by transfer via Bluetooth®.
- Measuring blood pressure
Note: To guarantee that you will be able to compare values adequately, always measure your blood pressure at the same time of day. Rest for approx. 5 minutes before every measurement!
If you wish to undertake multiple measurements on the one person, wait for 5 minutes between taking each measurement.
You should not eat, drink, smoke or be active in the 30 minutes before each measurement.
Repeat the measurement process if the measured values seem incorrect.
Bring the device to room temperature before the measurement.
- Attaching the cuff
ATTENTION! The device may only be used in conjunction with the original cuff provided. The cuff is suitable for an arm circumference between 22 and 36~cm .
Place the cuff on a bare upper left arm (Fig. D). The blood flow of the arm should not be constricted by clothing which is too tight, etc.
The cuff should be placed on the upper arm in such a way that the lower edge lies 2 - 3cm over the crook of the arm and the artery. The cuff tubing points to the centre of the palm.
Place the free end of the cuff closely, but not too tightly, around the arm and close the Velcro strip. The cuff should not be too tight (Fig. E). Check that two fingers can still fit beneath the cuff (Fig. F).
Now insert the cuff tubing with the cuff plug 3 into the connector (Fig. G).
- Adopting the correct posture
Note: Stay still during the measurement process and do not talk to prevent the measurement result from being falsified.
You can carry out the measurement while sitting or lying down. Always ensure that the cuff is at the same level as the heart (Fig. H-J).
Sit comfortably for the blood pressure measurement. Decline your back and arms. Do not cross your legs. Put your feet flat on the ground.
In order not to falsify the measurement result, it is important that you stay still and do not talk.
- Selecting a memory
Press the SET button. Select the desired user memory 品 .. 21 by pressing the function buttons -/+ 7. You have 4 memories with 30 memory spaces for saving the measurement results of 4 different people separately.
Confirm your choice using the START / STOP button or wait 3 seconds. Your choice will then be saved automatically.
- Carrying out the blood pressure measurement
Place the cuff on your arm as previously described and assume the position in which you wish to carry out the measurement.
Switch on the blood pressure monitor using the START / STOP button ① 8.
After the full-screen display, the last measurement result saved will be displayed. If no measurement has been saved, the device will display the value of 0 .
The cuff will inflate automatically. The cuff air pressure is released slowly. If a tendency towards high blood pressure has been detected, it will be repressurised and the cuff pressure will be increased once more. As soon as a pulse has been detected, the pulse symbol will be displayed.
The measurement results for systolic pressure, diastolic pressure and pulse will be displayed.
You can stop the measurement at any time by pressing the START/STOP button ① 8.
If the measurement has not been carried out correctly, "Er" will be displayed. Also take a look at "Troubleshooting" and repeat the measurement process.
The measurement result will be saved automatically.
If you have activated Bluetooth® data transfer, the data will be transferred. The blood pressure monitor displays the symbol for Bluetooth® transfer during transfer. Switch the blood pressure monitor off by pressing the START / STOP button once more.
To power down press the START / STOP button 8.
note: The device switches off automatically after one minute if no button has been pressed. Wait for at least 5 minutes before taking another measurement!
-Transferringmeasurements via Bluetooth®
You have the option of transferring the measured values and values saved on your device to your smartphone using Bluetooth® low energy technology.
Activate Bluetooth in your smartphone settings and open the "HealthForYou" app.
Note: The „HealthForYou“ app must be activated for transfer to occur.
Add "SBM 68" in the app.
Warning! The displayed values are intended merely as a visualisation and may not be used as the basis for therapeutic measures. The software is not part of a diagnostic medical system.
We wish to expressly indicate that the software at hand is not a medical product in accordance with the EU directive 93/42/EEC.
Note: During data transfer, button functions cannot be executed.
To transfer measurements to your smartphone via Bluetooth® proceed as follows:
Manual transfer
Go to save mode (see „Saving, requesting and deleting measurements"). As soon as the average value of all measurements is displayed, a Bluetooth® connection will automatically be established. The data will be transferred and the Bluetooth® symbol will be shown on the display.
Switch the blood pressure monitor off by pressing the START/STOP button ① 8.
Automatic transfer
If you forget to switch off the blood pressure monitor, it will automatically switch off after one minute. In this case, the value will be saved in the selected user memory and the data will be transferred if Bluetooth® data transfer has been activated. The Bluetooth® symbol will be shown on the display during transfer.
EvaluatingMeasurements
Cardiac arrhythmia
This device can identify any potential cardiac arrhythmia disorders and, if necessary, indicates this after the measurement process with the symbol. This can be an indicator for arrhythmia.
Arrhythmia is an illness in which the heart rhythm is abnormal due to errors in the bioelectric system, which controls the heartbeat. The symptoms (skipped or premature heartbeats, slow or rapid pulse) can stem from heart conditions, age, physical predispositions, an excess of luxury foods, stress or a lack of sleep. Arrhythmia can only be diagnosed by a doctor after a physical examination.
Repeat the measurement process if the symbol is shown on the display after the measurement. Please guarantee that you rest 5 minutes before the measurement and do not talk or move during it.
WARNING! If the symbol displayed repeatedly, contact your doctor. Self-diagnosis and self-treatment on the basis of measurement results can be dangerous. Follow your doctor's instructions.
- Classification of measurement results
In accordance with the guidelines / definitions provided by the World Health Organisation (WHO) and the latest findings, the measurement results can be classified and evaluated according to the following table:
| Range of blood pressure values | Systole (in mmHg) | Diastole (in mmHg) | Remedies |
| Stage 3 severe hypertension | ≥ 180 ≥ 110 | seek medical | attention |
| Stage 2: moderate hypertension | 160–179 | 100–109 | medical attention |
| Stage 1: mild hypertension | 140–159 | 90–99 | monitoring by a doctor |
| High, normal | 30–139 | 89 | monitoring by a doctor |
| Normal 120– | 129 | Self-monitor | |
| Ideal < 120 < | 80 Self-monitor |
Source: WHO, 1999
Note: These standard values merely serve as a general guideline as blood pressure deviates depending on the individual, different age groups, etc.
It is important that you consult your doctor at regular intervals. Your doctor will give you your particular values for normal blood pressure as well as the value for when your blood pressure is classified as dangerous.
The bar chart on the display and the scale on the device state which range the blood pressure measured is in. If the systole and diastole values are in two different ranges (i.e. systole is "high, normal" and diastole is "normal") the graphic categorisation on the device will always show the higher range i.e. in the above example this would be "high, normal".
- Saving, requesting and deleting measurements
The results of each successful measurement are saved together with the date and time. If there are over 30
measurement values, the oldest measurement value will be overwritten.
Press the SET button. Press the function buttons +/-7 to select the desired user memory.. Confirm your choice using the START/STOP button 8.
Press the memory button to display the average value "A" of all saved measurements pertaining to the user memory. Press the memory button M5 again to display the average value of the last 7 days for the morning measurement. (Morning: 5.00 am - 9.00 am, display "AM"). Press the memory button M5 again to display the average value of the last 7 days for the evening measurement. (Evening: 6 pm - 8 pm, display "PM"). By pressing the memory button M again, the last individual measurement will be displayed with the date and time.
Press the START / STOP button to switch off the device.
Note: The device switches off automatically after one minute when no button has been pressed.
Deleting data
Press the memory button to delete the entire memory of the respective user.
Wait until the Bluetooth symbol no longer flashes. Keep the function buttons - / + 7 pressed simultaneously for 5 seconds. After successful deletion the "CL 00" will appear in the display.
The data has been deleted.
- Cleaning and Care
ATTENTION! Possible property damage! Never hold the product under water as liquid could enter the product and damage it.
Clean the device and carefully using a slightly damp cloth.
Do not use cleaners or solvents.
Storage
Attention! Possible property damage! Do not place any heavy objects on the device and cuff 1.
If the product will not be used for more than one month, remove both batteries from the device to prevent possible battery leakage.
Do not kink the cuff tubing 2.
Store the product in a dry location (relative humidity ≤ 93% ). High humidity can shorten the service life or damage the product. Store the product in a location with ambient temperatures between -25 °C and +70 °C.
Troubleshooting
| Troubleshooting | Problem Solution | |
| Er1 The pulse cannot be measured correctly. | Repeat the measurement process. Be sure not to move or talk. | |
| Er2 You have been talking or moving during the measurement process. | ||
| Er3 The cuff was attached too tightly or too loosely. | ||
| Er4 An error occurred during the measurement process. | ||
| Er5 The inflation pressure is over 300 mmHg. | ||
| Er6 | The batteries are almost dead. | Reinsert the batteries or replace them with new batteries. |
| Er7 | Data cannot be sent via Bluetooth®. | Activate Bluetooth® on your smartphone and start the "HealthForYou" app. |
Disposal

The packaging is made entirely of recyclable materials, which you may dispose of at local recycling facilities.

Observe the marking of the packaging materials for waste separation, which are marked with abbreviations (a) and numbers (b) with following meaning: 1-7: plastics / 20-22: paper and fibreboard / 80-98: composite materials.
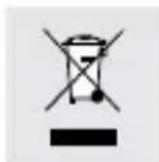
In accordance with European Directive (covering waste electrical and electronic equipment) and its transposition into national legislation, worn-out electrical devices must be collected separately and taken for environmentally compatible recycling.

Contact your local refuse disposal authority for more details of how to dispose of your worn-out product.
Per Directive 2006 / 66 / EC and its amendments, defective or used batteries must be recycled. Return the batteries and/or the product to a collection site.
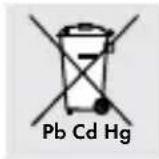
Improper disposal of batteries can harm the environment!
Never dispose of batteries in your household bin. They may contain toxic heavy metals and are subject to hazardous waste regulations. The chemical symbols of the heavy metals are: Cd = cadmium, Hg = mercury, Pb = lead. Therefore dispose of used batteries through your local collection facilities.
Warranty/service
We provide a 3-year warranty covering faults in materials or manufacturing of the blood pressure monitor from the purchase date.
The warranty does not cover:
- Damage due to improper use.
Wearparts. - Damage caused by the customer.
If the device has been opened by an unauthorised repair shop.
This warranty does not affect the customer's legal rights. The customer must provide proof of purchase in order for any warranty claims within the warranty period to be honoured. The warranty claim must be filed with Hans Dinslage GmbH, Uittenweiler, Germany, within 3 years from the date of purchase.
In the event of a complaint, please contact our Service Department listed below:
Service Hotline (free of charge):
GB
0800 93 10 319 (Mon-Fri: 8 am-6 pm)
service-uk@sanitas-online.de
IE
1800 93 92 35 (Mon-Fri: 8 am-6 pm)
service- ie@sanitas-online.de
If we ask you to return the defective blood pressure monitor, please send it to the following address:
send to:
NU Service GmbH
Lessingstraße 10b
DE-89231 Neu-UIm
GERMANY
IAN 290990
This device complies with European Standard EN60601-1-2 and is subject to special precautions with respect to electromagnetic compatibility. Please note, portable and mobile HF communication devices may interfere with this device. For details, please use the customer service address below or refer to the end of these instructions for use.
This device complies with EU Medical Devices Directive 93/42/EEC, the Medical Devices Act and EN1060-3 standards (non-invasive blood pressure monitors Part 3: Supplementary requirements for electro-mechanical blood pressure measuring systems) and standard IEC80601-2-30 (Medical electrical equipment Part 2-30: Particular requirements for basic safety and essential performance of automated non-invasive blood pressure monitors).
The accuracy of this blood pressure monitor has been thoroughly tested and was developed with regard to a long utilizable service life. If the device is used in medicine, technical inspections using the suitable equipment are to be carried out. Precise information on how to test the accuracy of the device can be requested from our service address.
We hereby guarantee that this product complies with the directive 2014/53/EU.
Please contact the above service address for details, such as the CE Declaration of Conformity.
Bluetooth® and the related logo are registered trademarks of Bluetooth SIG, Inc. Use of these brands by Hans Dinslage GmbH is under license. All other brands and trade names are the property of the respective owners.
758.828-0617
Guidelines and manufacturer's declaration - electromagnetic interference
The SBM 68 is intended for operation in an electromagnetic environment such as is described below. The client or the user of the SBM 68 should ensure that is used in this type of environment.
| Electromagnetic interference measurements | Compliance | Electromagnetic environment - Guideline |
| RF emissions according to CISPR 11 | Group 1 The SB | BM 68 uses RF energy exclusively for its internal function. Therefore, RF emissions are very low and it is improbable that neighbouring electronic devices will be affected by interference. |
| RF emissions according to CI-SPR 11 | Class B The SB | M 68 is intended for use in all indoor areas, including living areas, and is suitable for those which are directly connected to a public supply network which also supplies buildings used for residential purposes. |
Guidelines and manufacturer's declaration - electromagnetic interference
The SBM 68 is intended for operation in an electromagnetic environment such as is described below. The client or the user of the SBM 68 should ensure that is used in this type of environment.
| Immunity tests | IEC 60601-test level | Compliance level | Electromagnetic environment - guideline |
| Electrostatic discharge (ESD) in accordance with IEC 61000-4-2 | ±6 kV Contact discharge ±8 kV air discharge | ±6 kV Contact discharge ±8 kV air discharge | Floors should be made from wood or concrete or be decorated with ceramic tiles. If the floor is covered with synthetic material, the relative humidity should be at least 30%. |
| Magnetic field for supply frequency (50/60 Hz) In accordance with IEC 61000-4-8 | 3 A/m 3 A/m The quality of the supply frequency should correspond to that of a typical commercial or hospital environment. | ||
Guidelines and manufacturer declaration - electromagnetic immunity
The blood pressure monitor SBM 68 is intended for operation in the electromagnetic environment described below. The customer or the user of the blood pressure monitor SBM 68 should ensure that it is used in this type of environment.
| Immunity tests | IEC 60601-Test level | Compliance level | Electromagnetic environment - guideline |
| Portable and mobile communication devices should not be used in closer proximity to the blood pressure monitor SBM 68 (including the cables) than the recommended safety distance, which is calculated according to the equation applicable for the transmission frequency. | |||
| Conducted RF disturbance variables in accordance with IEC 61000-4-6 | 3 VRMS150 KHz to80 MHz | 3 VRMS | Recommended safety distance:d=[3.5/Vi]√P |
| Emitted RF disturbance variables in accordance with IEC 61000-4-3 | 3 V/m80 MHz to2.5 GHz | 3 V/m | d=[3.5]/√Pfor 80 MHz to800d=[7/(E1]√P800 MHz to2.5 GHz |
| where P is the transmitter's rated output in watts (W) in accordance with the information provided by the transmitter manufacturer and d is the recommended safety distance in metres (m). The field strength of stationary radio transmitters, according to an on-site investigationa, should be lower than the compliance level for all frequencies.b. Interference is possible in the same environment as devices which bear the following image. | |||
| NOTE 1 At 80 MHz and 800 MHz the higher frequency range applies. NOTE 2 These guidelines may not be applicable in all cases. Electromagnetic propagation is influenced by absorptions and reflections by buildings, objects and people. | |||
| a The field strength of stationary radio transmitters, such as base stations for wireless telephones and land mobile radios, amateur radio stations, AM and FM radios and TV stations, cannot be predicted theoretically with accuracy. To determine the electromagnetic environment with regard to the stationary transmitter, an electromagnetic site survey should be considered. If the measured field strength exEECs the above compliance level in the location in which the blood pressure monitor SBM 68 is being used, the blood pressure monitor SBM 68 should be observed to verify normal operation. If you observe unusual performance characteristics, additional measures may be required e.g. changing orientation or choosing another location for the blood pressure monitor SBM 68. b Over the frequency range of 150 kHz to 80 MHz, the field strength should be lower than 3 V / m. | |||
Recommended safety distances between portable and mobile RF telecommunication devices and the blood pressure monitor SBM 68
The blood pressure monitor SBM 68 is designed for use in an electromagnetic environment in which RF disturbances are controlled. The customer or the user of the blood pressure monitor SBM 68 can thus help to prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF telecommunication devices (transmitters) and the blood pressure monitor SBM 68 - depending on the communication device's power output as stated below.
| Rated power of the transmitter (W) | Safety distance, depending on the transmission frequency (m) | |
| 80 MHz to 800 MHz d = [3.5/E1]√P | 800 MHz to 2.5 GHz d = [7/E1]√P | |
| 0.01 0.1167 0.2334 | ||
| 0.1 0.3689 0.7378 | ||
| 1 1.1667 2.3334 | ||
| 10 3.6893 7.3786 | ||
| 100 11.6667 23.33 | 34 | |
For transmitters whose maximum rated output is not stated in the above table, the recommended safety distance d may be determined in metres (m)
using the equation applicable to the frequency of the transmitter, whereby P is the maximum rated output of the
transmitter in watts (W) according to the transmitter manufacturer.
NOTE 1 At 80MHz and 800MHz the higher frequency range applies.
NOTE 2 These guidelines may not be applicable in all cases. Electromagnetic propagation is influenced by absorptions and reflections by buildings, objects and people.
"HealthForYou" - MS Internet Explorer
-MS Edge
- Mozilla Firefox
- Google Chrome
-Apple Safari
Service hotline (gratis)
NL
0800 54 30 543 (ma. - fr.: 8-18 klok)
service-nl@sanitas-online.de
BE
0800 70 611 (ma. - fr.: 8-18 klok)
service-be@sanitas-online.de