Systo Monitor Connect 400 - Blood pressure monitor SOEHNLE - Free user manual and instructions
Find the device manual for free Systo Monitor Connect 400 SOEHNLE in PDF.

User questions about Systo Monitor Connect 400 SOEHNLE
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Blood pressure monitor in PDF format for free! Find your manual Systo Monitor Connect 400 - SOEHNLE and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. Systo Monitor Connect 400 by SOEHNLE.
USER MANUAL Systo Monitor Connect 400 SOEHNLE
text_image
QR code image containing encoded data, no visible human-readable text
text_image
GET ITON Google Play Download on the App StoreAndroid ^™ : min. 4.4.4 / iOS ^® : min. 9.0 / Bluetooth ^® : min. 4.0
Guangdong Transtek Medical Electronics Co., Ltd. Zone A, No.105, Dongli Road, Torch Development District, Zhongshan, 528437, Guangdong, China
INTRODUCTION......18
Safety information
Scope of delivery
General description
Indications for Use
Measurement Principle
Device description
Power supply
Inserting and replacing the batteries
The LCD screen
PRIOR TO THE FIRST MEASUREMENT.. 24
Setting date, time, and unit of measure
Selecting the user
Soehnle Connect app
MEASURING......25
Measurement information
Cuff placement
Starting the measurement
DATA MANAGEMENT 26
Recalling stored values
Deleting stored values
Data transmission
INTERPRETING THE RESULTS......27
CUSTOMER INFORMATION ......28
Maintenance and Storage
Contact
Customer Service
Guarantee
TROUBLESHOOTING......29
TECHNICAL DATA....30
LIST OF COMPLIED
EUROPEAN STANDARDS .... 31
EMC GUIDANCE....144
Safety information
 | Symbol for "THE OPERATION GUIDE MUST BE READ" | [KZTA] | Symbol für „MIT HOCHFREQUENZSENDER" |
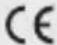 | Symbol for "COMPLIES WITH MDD 93/42/EEC REQUIREMENTS" | [2ZBW] | Symbol for "TYPE BF APPLIED PARTS" |
| 0123 |  | Symbol for "ENVIRONMENT PROTECTION - Electrical waste products should not be disposed of with household waste. Please recycle where facilities exist. Check with your local authority or retailer for recycling advice" | |
| [H336] | Symbol for "MANUFACTURER" | ||
 | Symbol for "SERIAL NUMBER" | ||
| [Z235] | Symbol for "DIRECT CURRENT" | [H3KA] | Symbol for "Authorised Representative in the European Community" |
 | Symbol for "MANUFACTURE DATE" |  | Caution: These notes must be observed to prevent any damage to the device. |
 | The Bluetooth Combination Mark |
0123

IMPORTANT INFORMATION! PLEASE KEEP!
Carefully read these user instructions and in particular the safety information prior to using this device and keep these user instructions for future use. When handing the device over to third parties, please also include these user instructions. Non-compliance with these instructions may cause injuries or damage the device.

Information on use
- The device is intended for the independent measurement and monitoring of arterial blood pressure. Only a medical doctor can provide a diagnosis.
- The user must review the functionality and proper condition of the device prior to using it. Continued use of a damaged device may result in injuries and erroneous measurement results or pose a serious risk. In case of technical problems with the device, please contact Soehnle's CUSTOMER SERVICE. Never open or repair the device.
- The device is intended to be used only by adults. Do not use the device if you are pregnant or suspect a pregnancy.
- This device may be used only for the purpose described in these instructions. The manufacturer does not accept any liability for damages based on improper use.
- The device is designed only for personal use.
- It must not be used on any extremities but the arms.
- If you take medication please ask your doctor about the best time to measure your blood pressure.
- If the device is used for measurements on patients with widespread arrhythmia, e.g. extrasystoles of the atrium or ventricularextrasystoles, the best results are generally achieved with deviations. Please discuss the results with your doctor.
- If the cuff pressure is higher than 40 kPa (300 mmHg), the device releases air automatically. If the cuff does not release air at a pressure of 40 kPa (300 mmHg), please remove the cuff from your arm and push the START/STOP key to prevent further inflation.
- The device is not approved for AP or APG categories.
- Avoid interference signals emitted by strong electromagnetic fields or rapid transient disturbances to prevent measuring errors.
- The device is not suited for continuous blood pressure monitoring in medical emergencies. Blood circulation may be disrupted.
- The cuff material has been tested and is proven to correspond to the requirements of ISO 10993-5:2009 and ISO 10993-10:2010. It does not cause skin irritation or other reactions.
- Please use only the ACCESSORIES and removable parts prescribed
EN
by the manufacturer. Otherwise, danger may arise for the user and the device.
- Please inform Soehnle customer service of unexpected operating status or events.
- The optional adapter is a component of the MEDICAL ELECTRICAL DEVICE.
- The plugs/pins of the adapter plug isolate the device against the mains power supply.
- Please do not place the device in a location in which it is difficult to disconnect from the power supply.
- Keep the device outside of the reach of small children/pets to prevent the inhalation or swallowing of small parts. The cable/cord poses a risk of strangulation.
- When stored at the minimum storage temperature between uses, electrical medical devices must be warmed up for at least 30 minutes before they are operational. When stored at the maximum permissible storage temperature between uses, electrical medical devices must cool off for at least 30 minutes before they are operational.
- Please carefully monitor a possible interruption of blood circulation and a possible injury of the patient.
- Do not place the cuff over a wound as this may cause further injuries.
-
Do not place the cuff on the same arm on which other electrical medical devices are placed.
-
Please ensure that the operation of the device does not interrupt blood circulation for a longer period.
- Avoid squeezing or constriction of the connecting tube.
- The Blood Pressure Monitor has been clinically tested pursuant to the requirements of EN 1060-4:2004.

Metrological maintenance and care information
- Do not perform any metrological maintenance if the device is in use.
-
For reliable operation, we recommend that the device is subject to metrological control once every two years pursuant to Sec. 14 of the Medical Product Operating Directive (MPBetriebV). We recommend testing the device's performance once every two years and after metrological control and repair, at a minimum by testing the tolerance requirements of the pressure display and tightness of the cuff (testing at least at 50 mmHg and 200 mmHg).
-
The metrological control must be performed by a licensed tester or testing institute.
• We recommend cleaning the device and cuff only with a soft cloth.

Storage information
- If the device is not being used, store it and the adapter in a dry location away from heat, dust, fibres, lint, and out of direct sunlight. Do not place heavy objects on the storage box.

Information about handling batteries
- Do not mix new and used batteries.
- Do not use different types of batteries at the same time.
- Do not throw batteries into a fire. Batteries may explode or leak. Remove the batteries if the device is not used for a long time.

Disposal information

Battery disposal EC Directive 2008/12/EC
Batteries are not a part of your regular household waste. You must return batteries or rechargeable batteries to your municipality's public collection or wherever batteries of the respective type are being sold.

Disposal of electric and electronic devices, EC Directive 2012/19/EU
This product is not to be treated as regular household waste but must be returned to a collection point for recycling electric and electronic devices. Further information is available from your municipality, your municipality's waste disposal services, or the retailer where you purchased your product.

Declaration of conformity
0123 This device is in compliance with the basic requirements and the other pertinent regulations of the Directive 93/42/EEC.
Scope of delivery
1x blood pressure monitor Systo Monitor Connect 400
1x cuff (22–42 cm/8.66–16.54 inch)
4x alkaline battery AAA 6 V DC
1x user instruction
A power adapter is NOT included in the scope of delivery. It is available separately, if needed. Information is available under "Power supply".
General description
Thank you for choosing the blood pressure monitor Soehnle Systo Monitor Connect 400 for use on the upper arm.
Measurements taken with Soehnle Systo Monitor Connect 400 are equal to those taken by a trained professional.
This user instruction contains important information concerning safety, care of the device, and explain the proper use step-by-step.
EN
Indications for Use
The Soehnle Systo Monitor Connect 400 is digital monitors intended for use in measuring blood pressure and heartbeat rate with arm circumference ranging from 22 – 42 cm / 8.66 – 16.54 inch.
It is intended for adult indoor use only.
Measurement Principle
This product uses the Oscillometric Measuring method to detect blood pressure. Before every measurement, the unit establishes a “zero pressure” equivalent to the air pressure. Then it starts inflating the arm cuff, meanwhile, the unit detects pressure oscillations generated by beat-to-beat pulsatile, which is used to determine the systolic and diastolic pressure, and also pulse rate.
Device description
Please note the fold-out page.
A Cuff
B Air hose
© LCD display
D Pump
Power plug connection
F Battery compartment
G Memory key (Q)
Settings key (☐)
1 Start/Stop key ( ) START
Power supply
-
Battery operation:
4 alkaline batteries AAA 6 V DC -
Operation with optional power adapter:
$$ 6 \mathrm{V} = 1 \mathrm{A} $$
Please use only the adapter approved by the manufacturer. Available separately (Item No. 89165/5).
Inserting and replacing batteries
- Open the battery compartment.
- Insert the batteries. Note the correct polarity.
- Close the battery compartment cover.
The normal service life of new and unused batteries is 300 measurements at a measurement period of 60 seconds each.
The LCD screen
| Symbol Description Explanation | ||
| SYS | Systolic blood pressure Upper blood pressure value | |
| DIA | Diastolic blood pressure Lower blood pressure value | |
| PUL /min | Pulse display Pulse in beats per minute | |
| Symbol pressure release | Air is released from the cuff | |
| Memory Displays that the device is in memory mode | ||
| kPa | kPa | Unit of measurement for blood pressure (1 kPa = 7.5 mmHg, see page 24) |
| mmHg | mmHg | Unit of measurement for blood pressure (1 mmHg = 0.133 kPa, see page 24) |
| Battery charge display Displays charge status of the battery | ||
| Irregular pulse Irregular pulse detected during measurement | ||
| Blood pressure level | Indicates blood pressure level (based on WHO scale, see page 27) | |
| BB/BB | Current time Year/month/day, hour/minute | |
| Heartbeat | The blood pressure monitor detects the heartbeat during the measurement | |
| User 1 Active user profile (user 1) | ||
| User 2 Active user profile (user 2) | ||
| Movement indicator Indication of result distorting movement | ||
| AVG | Average value Average value of the blood pressure | |
| Bluetooth symbol Shows that Bluetooth is activated | ||
| Data transmission | Shows that no data was transmitted to the smartphone | |
EN
EN
Setting date, time, and unit of measure
Date and time must be set prior to using the blood pressure monitor so that every measurement value that is stored can be time stamped.
- If the device is switched off, push the key (☐) to display the time. Then push and hold the key (☐) to set the year.
- Push the key (Q) to change the [YEAR]. Each time you push the key, the year increases by one.
- Once the year has been set, push key (☐) to save the information and continue.
- Repeat steps 2 and 3 to set [MONTH], [DAY], [HOUR], [MINUTE] and [UNIT OF MEASURE]
You can switch between the unit of measure "kPA" (kilo pascal) or "mmHg" (millimetre mercury column), where mmHg is the most common unit for measuring blood pressure.
- After setting the unit of measure, the LCD display shows "done" and then displays all settings entered by you before switching off.
Selecting the user
A total of two users can be programmed. 60 datasets can be stored for each user.
- If the device is switched off, push and hold the key (☐) to select the user. The user symbol (☐) fl ashes.
- Push key (Q) again to select between user 1, 2 or "Guest" (guest mode).
The measurements are not stored or transmitted in guest mode.
- Confirm the selection of the respective user by pushing (The respective user and "done" are shown before the display switches off.
Soehnle Connect app
To use all functions of your Systo Monitor Connect 400, we recommend that you connect the device to the Soehnle Connect app prior to first use.
- Download and install the free Soehnle Connect app on your smartphone or tablet. Scan the QR code to access the app directly. Or find it in Google Play or the App Store..

text_image
QR code image containing encoded data, no visible human-readable text

Android ^ : min. 4.4.4 / iOS ^ : min. 9.0 / Bluetooth ^* : min. 4.0
- Activate Bluetooth on your smartphone or tablet and open the Soehnle Connect app. When opening the app for the first time, you will automatically be guided through the process to connect a Soehnle end device.
Once you have already used the Soehnle Connect app, to connect in the app go to the "main menu" and press "my devices" and then "add devices".
Compatibility
The Soehnle Connect app is compatible with Google Fit® and Apple Health®.
Measuring information
The measurements may be incorrect under the following circumstances:
• Within 1 hour after a meal or a drink
• Measurements immediately after consuming tea, coffee, or after smoking
• Within 20 minutes after a bath
• While speaking or moving fingers
• In a very cold environment
- With a strong urge to urinate
- Relax for 5 min. before measuring.
- Wait at least 3 min. between two measurements. This allows the blood flow in your arm to return to normal.
- For a reasonable comparison of various measurement values, always attempt to measure in the same situations. For example, take your measurement daily at around the same time and with the upper arm in the position as shown to you by a doctor.
Cuff placement
-
Place the cuff on the upper arm and then turn the hose towards the inside of the arm in line with the pinkie finger. You can also align the arterial mark so that it is placed above the largest artery (on the inside of the arm). You can find the largest artery by gently pushing 2 fingers approximately 2 cm above the crook of your arm, on the inside of your left arm. Find where the pulse can be felt the strongest. This is where the largest artery is located. (Fig. 1)
-
The cuff should be snug but not too tight. It must be possible to insert one finger between the cuff and your arm. (Fig. 2)
- Sit in a comfortable position and place the arm on which the measurement will be taken on a level surface.
Sit down comfortably prior to starting the measurement: do not cross your legs, place the soles of your feet on the floor, your back and arm should be supported.
4. For users with hypertension:
The centre of the cuff should be at the level of the right atrium (Fig. 3).
Starting the measurement
- If the blood pressure monitor is switched off, switch it on by pushing the key ( ) The device performs the measurement.
- The device will first zero adjust itself
- The cuff will then be inflated and the device will measure the blood pressure and pulse.
Push the key ( ∅START ) to stop the measurement at any time.
- Once the measurement is completed, the air is released from the cuff and the measured values will be displayed. They will be automatically allocated to the selected user and stored. Year, date, and time are shown alternately.
- Push the key ( ∅START ) to switch the device off. The device switches off automatically after 1 minute.
EN
Recalling stored values
- Push (Q) to switch the blood pressure monitor on. The average value of the last three datasets will be displayed.
- Push the key (✗) or (○) to get to the desired datasets. While scrolling, the number of the displayed dataset and the total number of stored measurements will be shown in the upper right corner.
If you leave a dataset displayed, date and time of the measurement will also be shown. They will be put through automatically.
- If you would like to recall the datasets of another user, first push the key ( ) to switch the device off if the blood pressure monitor is still in the “recall memory” mode.
Push and hold the key (☐) to enter the mode "select user", push the key (☐) again to select between user 1 and user 2. Push the key (☐) to confirm the user, then recall the measured datasets of the selected user with (☐).
The most current dataset (1) is shown first. Each new measurement is stored at dataset (1). All other datasets are moved back by one (2 becomes 3, etc.) and the last dataset (60) is removed from the list.
Deleting stored values
If a measurement is not performed correctly, you can delete all measurement results for the selected user as follows.
- Push and hold key (Q) for approximately 3 seconds while the blood pressure monitor is in the "recall memory" mode. "dEL ALL" (delete ALL) will flash and the selected user will be displayed.
Push ( ) to exit the "delete" mode without deleting datasets.
- Push (☐) to confirm deletion, and "user+dEL done" will be displayed. The blood pressure monitor then switches off automatically.
Data transmission
After each measurement, your personal blood pressure information is transmitted via Bluetooth to your mobile device on which the Soehnle Connect app is installed.
- After the measurement, the Bluetooth symbol (*) flashes on the top right of the screen. If a connection with your mobile device is established, the Bluetooth symbol (*) freezes.
- The new measurement data will be transmitted after a successful connection with your mobile device has been established.
- After successful transmission, the Systo Monitor Connect 400 displays "done" on the screen.
In the event of an error during data transmission, all stored values (for both users) can be re-send. To do this, push and hold the start/stop button for 3 seconds until the Bluetooth symbol flashes on the screen. Then repeat steps 2 and 3.
What is systolic and diastolic blood pressure?
When the heart ventricles contract to pump blood out of the heart, the blood pressure reaches its maximum value in the cycle, the so-called systolic value. The blood pressure reaches its lowest value in the cycle when the heart ventricles relax. This is the diastolic blood pressure.
How are blood pressure values usually categorized?
The classification of blood pressure published in 1999 by the World Health Organization (WHO) and the International Society of Hypertension (ISH) is as follows:
| Classifica-tion | SYS(mm HG) | DIA(mm HG) | |
| Grade 3 HT ≥ 180 ≥ 110 | |||
| Grade 2 HT 160 – 179 100 – 109 | |||
| Grade 1 HT 140 – 159 90 – 99 | |||
| High normal 130 – 139 85 – 89 | |||
| Normal 120 – 129 80 – 84 | |||
| Optimal < 120 < 80 | |||
| HT = Hypertension (high blood pressure) | |||
IMPORTANT
Only a medical doctor can determine your normal blood pressure range. Please see a doctor if your measurement results are outside of this range.
Recognizing irregular pulse
The device recognizes irregular pulse (arrhythmia) if the heartbeat fluctuates while measuring the systolic and diastolic blood pressure. The blood pressure monitor records heartbeat intervals and calculates the average value during each measurement. If an average value is greater than or equal 25%, the measurement result also displays a warning signal for irregular pulse.

The warning "irregular pulse" shows that an irregular pulse has been determinated during the measurement, which might indicate arrhythmia. Generally, this is NOT a cause for concern. However, if the symbol is displayed frequently, we recommend that you speak with your doctor. The device does not replace a cardiology examination but provides an early detection of irregular pulse.
EN
Maintenance and Storage
Your device will provide the best measurement accuracy if you comply with the following instructions:
- Store in a dry location and avoid direct sunlight
- Avoid contact with water, clean with a soft cloth if necessary.
- Avoid strong vibrations and impact.
- Do not store in dusty environments or varying temperatures.
- Do not clean the reusable cuff with water and do not submerge it in water.
Contact
Soehnle Customer Service
Hotline +49 (0) 26 04 - 977 0 Mon - Fri, 7:00 a.m. - 6:00 p.m.
Further information about our products can be found at www.soehnle.com.
Distributor:
Soehnle is a brand of Leifheit AG, Leifheitstraße 1 56377 Nassau/Germany

Manufactured by:
Guangdong Transtek Medical Electronics Co., Ltd. Zone A, No.105, Dongli Road, Torch Development District, Zhongshan, 528437, Guangdong, China

European representative:
MDSS - Medical Device Safety Service GmbH, Schiffgraben 41, 30175 Hanover, Germany
Guarantee
Leifheit AG offers a 2 year guarantee for this quality product as of the purchase date (or in case of orders, as of receipt of the goods). Guarantee claims must be asserted within the guarantee period and immediately after occurrence of the defect. The guarantee includes product quality.
The following are excluded from the guarantee:
- Wear caused by use or other natural wear,
- Damage caused by improper use or handling (e.g. impact, shock, drop),
- Damage caused by non-compliance with these user instructions. In case of a guarantee claim, Leifheit, at its own discretion, either repairs the defective components or replaces the product. If a repair cannot be performed and an identical product is no longer available in the product range for the purpose of replacement, you will receive a replacement product of the closest equivalent value. Reimbursement of the purchase price is not possible in case of a guarantee claim. This guarantee furthermore does not grant any claims for damages. To assert the guarantee, please contact the seller where you have acquired the product and present the defective product and the purchase receipt (copy). This guarantee is valid worldwide. Your statutory rights, in particular warranty rights, continue to apply and are not limited by this guarantee.
| PROBLEM POSSIBLE | CAUSE RESOLUTION | |
| Display does not light up | Batteries are empty Replace batteries | |
| Batteries are not inserted correctly. | Insert batteries correctly. | |
| The DC adapter is not inserted correctly. | Insert the DC adapter correctly. | |
| lo+qis displayed | Low battery charge The charge status of the battery is too low; batteries must be replaced. | |
| Display is difficult to see or slow | Battery charge is too low. | Replace batteries |
| E 1 is displayed The cuff is too tight or too loose. | Reposition cuff and repeat measurement | |
| E 3 is displayed The pressure of the cuff is excess. | Relax for a moment and repeat the measurement. | |
| E 10 or E 11 is displayed | The blood pressure monitor displays movement during the measurement. | Movements may affect the measurement. Relax for a moment and repeat the measurement. |
| E 20 is displayed No pulse was detected during measurement. | Remove the clothing from your arm and repeat the measurement. | |
| E 21 is displayed A precise measurement could not be performed. | Relax for a moment and repeat the measurement. | |
| EExx is displayed. A “calibration error” occurred. (XX can indicate a digital signal, e.g. 01, 02 etc.) | Repeat the measurement. If the problem occurs again please contact your seller or our customer service for further assistance. | |
| “Out” is displayed Outside of the measurement range | Relax for a moment. Reposition the cuff and repeat the measurement. If the problem occurs again please contact your doctor. | |
EN
EN
| Item number 68097 (Modell: TMB-1585-BT) | |
| Power supply Battery operation: 4 alkaline batteries AAA 6V DCPower adapter operation 6 V---1 A(Please use only the recommended power adapter.) | |
| Display mode LCD display (100 x 68 mm / 3.94 × 2.68 inch) | |
| Measurement mode Oscillographic testing mode | |
| Measurement range Target cuff pressure: 0 mmHg - 300 mmHg (0 kPa - 40 kPa) measurement pressureSYS: 60 mmHg - 230 mmHg (8.0 kPa - 30.7 kPa)DIA: 40 mmHg - 130 mmHg (5.3 kPa - 17.3 kPa)Pulse: (40 - 199) beats per minute | |
| Accuracy Pressure: 5 °C - 40 °C within ± 3 mmHg (0.4 kPa) pulse at: ± 5 % | |
| Normal operating conditions | Temperature: 5 °C to 40 °CRelative humidity: ≤85 % RHAtmospheric pressure: 86 kPa to 106 kPa |
| Storage and transport conditions | Temperature:-20 °C to 60 °CRelative Humidity: 10 % RH-93 % RHAtmospheric Pressure: 50 kPa-106 kPa |
| Measurement perimeter on the upper arm | 22 - 42 cm / 8.66 - 16.54 inch |
| Weight approx. 277 g / 9.7 oz (excluding batteries and cuff) | |
| External dimensions approx. 154 ×106 × 57 mm / 6.06 × 4.17 × 2.24 inch | |
| Accessories 4 alkaline batteries AAA | |
| Operating mode Continuous operation | |
| Protection category Applied part type BF | |
| Water ingress protection | IP21 means that the blood pressure monitor is protected against solid foreign bodies with a size of 12.5 mm / 0.49 inch and larger as well as vertically dripping water. |
| Device Classification Battery Powered Mode: Internally Powered ME EquipmentAC Adaptor Powerde Mode: Class II ME Equipment | |
| Software version V01 | |
| Bluetooth Bluetooth Module No.: AW 2540 MV 1High frequency range: 2402 MHz to 2480 MHzOutput power level: -1 dBmSupply Voltage: 2 V - 3.6 VTransmitting Distance: 10 meters | |
WARNING: Modifications of the device are not permitted.
| Risk management EN ISO 14971:2012 Medical devices - Application of risk management to medical devices | |
| Labeling EN 980:2008 Symbols for use in the labelling of medical devices | |
| User manual EN 1041:2008 Information supplied by the manufacturer of medical devices | |
| General Requirements for Safety | EN 60601-1:2006 Medical electrical equipment - Part 1: General requirements for basic safety and essential performanceEN 60601-1-11:2015 Medical electrical equipment - Part 1-11: General requirements for basic safety and essential performance - Collateral standard: Requirements for medical electrical equipment and medical electrical systems used in the home healthcare environment |
| Electromagnetic compatibility | EN 60601-1-2:2007 Medical electrical equipment - Part 1-2: General requirements for basic safety and essential performance - Collateral standard: Electromagnetic compatibility - Requirements and tests |
| Performance requirements | EN ISO 81060-1:2012 Non-invasive sphygmomanometers - Part 1: Requirements and test methods for non-automated measurement typeEN 1060-3:1997+A2:2009 Non-invasive sphygmomanometers - Part 3: Supplementary requirements for electro-mechanical blood pressure measuring systems |
| Clinical investigation | EN 1060-4:2004 Non-invasive sphygmomanometers - Part 4: Test procedures to determine the overall system accuracy of automated non-invasive sphygmomanometers |
| Usability EN 60601-1-6:2010 Medical electrical equipment - Part 1-6: General requirements for basic safety and essential performance - Collateral standard: UsabilityEN 62366:2008 Medical devices - Application of usability engineering to medical devices | |
| Software life-cycle processes | EN 62304:2006/AC: 2008 Medical device software - Software life-cycle processes |
EN
INTRODUCTION
INTRODUCTION......32
GESTION DES DONNÉES......40
INFORMATIONS POUR LE CLIENT .....43
Maintenance et stockage Contact Service clients Soehnle Garantie
DÉPANNAGE.... 44
DONNÉES TECHNIQUES ...... 45
LISTE DES NORMES EUROPÉENNES APPLICABLES ...... 46
DIRECTIVE CEM....144
Application Soehnle Connect
text_image
QR code image containing encoded data, no visible human-readable text
text_image
GET ITON Google Play Download on the App StoreAndroid ^® : min. 4.4.4 / iOS ^® : min. 9.0 / Bluetooth ^® : min. 4.0
Service clients Soehnle
Guangdong Transtek Medical Electronics Co., Ltd. Zone A, No.105, Dongli Road, Torch Development District, Zhongshan, 528437, Guangdong, Chine
text_image
QR code image containing encoded data, no visible human-readable text
text_image
GET ITON Google Play Download on the App StoreAndroid ^ : min. 4.4.4 / iOS ^ : min. 9.0 / Bluetooth ^* : min. 4.0
Zone A, No.105, Dongli Road, Torch Development District, Zhongshan, 528437, Guangdong, Cina
ECREP Rappresentanza europea:
MDSS - Medical Device Safety Service GmbH, Schiffgraben 41, 30175 Hannover, Germania
Garanzia
KLANTINFORMATIE....75
Onderhoud en opslag
Contact
Soehnle klantenservice
Garantie
STORINGEN VERHELPEN......76
TECHNISCHE SPECIFICATIES......77
LIJST VAN NAGELEEFDE
EUROPESE NORMEN 78
EMC RICHTLIJN 144
text_image
QR code image containing encoded data, no visible human-readable text

Android*: min. 4.4.4 / iOS*: min. 9.0 / Bluetooth*: min. 4.0
Guangdong Transtek Medical Electronics Co., Ltd. Zone A, No.105, Dongli Road, Torch Development District, Zhongshan, 528437, Guangdong, China

text_image
QR code image containing encoded data, no visible human-readable text
text_image
GET ITON Google Play Download on the App StoreAndroid™: mín. 4.4.4 / iOS®: mín. 9.0 / Bluetooth®: mín. 4.0
Guangdong Transtek Medical Electronics Co., Ltd. Zone A, No.105, Dongli Road, Torch Development District, Zhongshan, 528437, Guangdong, China
INTRODUKTION......96
Säkerhetsinstruktioner
Leveransomfång
Allmän beskrivning
Användningsområde
Mätprincip
text_image
QR code image containing encoded data, no visible human-readable text

Android*: min. 4.4.4 / iOS*: min. 9.0 / Bluetooth*: min. 4.0
Guangdong Transtek Medical Electronics Co., Ltd. Zone A, No.105, Dongli Road, Torch Development District, Zhongshan, 528437, Guangdong, Kina
ECREP EU-representant :
MDSS - Medical Device Safety Service GmbH, Schiffgraben 41, D-30175 Hannover, Tyskland
Garanti
text_image
QR code image containing encoded data, no visible human-readable text
text_image
GET ITON Google Play Download on the App StoreAndroid ^® : min. 4.4.4 / iOS ^® : min. 9.0 / Bluetooth ^® : min. 4.0
Guangdong Transtek Medical Electronics Co., Ltd. Zone A, No.105, Dongli Road, Torch Development District, Zhongshan, 528437, Guangdong, Čína

Evropský zástupce:
MDSS - Medical Device Safety Service GmbH, Schiffgraben 41, 30175 Hannover, Německo
Záruka
text_image
QR code image containing encoded data, no visible human-readable text
text_image
GET ITON Google Play Download on the App StoreAndroid ^™ : min. 4.4.4 / iOS ^® : min. 9.0 / Bluetooth ^® : min. 4.0
Zone A, No.105, Dongli Road, Torch Development District, Zhongshan, 528437, Guangdong, Chiny
ECREP Representant europejski:
MDSS - Medical Device Safety Service GmbH, Schiffgraben 41, 30175 Hanower, Niemcy
Gwarancja
1) This product needs special precautions regarding EMC and needs to be installed and put into service according to the EMC information provided, and this unit can be affected by portable and mobile RF communications equipment.
2) Do not use a mobile phone or other devices that emit electromagnetic fields, near the unit. This may result in incorrect operation of the unit.
3) Caution: This unit has been thoroughly tested and inspected to assure proper performance and operation!
4) Caution: This machine should not be used adjacent to or stacked with other equipment and that if adjacent or stacked use is necessary, this machine should be observed to verify normal operation in the configuration in which it will be used.
Table 1
| Guidance and manufacturer's declaration – electromagnetic emissions | ||
| The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that it is used in such an environment. | ||
| Emissions test Compliance Electromagnetic environment - guidance | ||
| RF emissionsCISPR 11 | Group 1 The device uses | RF energy only forits internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. |
| RF emissionsCISPR 11 | Class B The device is suitable for use in all establishments, other than domestic and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes. | |
| Harmonic emissionsIEC 61000-3-2 | not applicable | |
| Voltage fluctuations / flicker emissionsIEC 61000-3-3 | not applicable | |
Table 2
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The device is intended for use in the electromagnetic environment specified below.The customer or the user of the device should assure that it is used in such an environment. | |||
| Immunity test | IEC 60601 test level | Compliance level | Electromagnetic environment - guidance |
| Electrostatic discharge (ESD)IEC 61000-4-2 | ±6 kV cont act±8 kV air | ±6 kV cont act±8 kV air | Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30%. |
| Electrical fast transient/burstIEC 61000-4-4 | power supply lines:±2 kVinput/output lines:±1 kV | not applicable Mains | power quality should be that of a typical commercial or hospital environment. |
| SurgeIEC61000-4-5 | line(s) to line(s):±1 kVline(s) to earth:±2 kV100 kHz repetition frequency | not applicable Mains | power quality should be that of a typical commercial or hospital environment. |
| Voltage dips, short interruptions and voltage variations on power supply input linesIEC 61000-4-11 | <5% UT (>95% dip in UT) for 0.5 cycle40% UT (60% dip in UT) for 5 cycles70% UT (30% dip in UT) for 25 cycles<5% UT (>95% dip in UT) for 5 s | not applicable Mains | power quality should be that of a typical commercial or hospital environment. If the user of the device requires continued operation during power mains interruptions, it is recommended that the device be powered from an uninterruptible power supply or a battery. |
| Power frequency(50Hz/60Hz)magnetic fieldIEC 61000-4-8 | 3 A/m 3 A/m Power frequency magnetic | frequency magnetic | fields should be at levels characteristic of a typical location in a typical commercial or hospital environment. |
| NOTE: UT is the a.c. mains voltage prior to application of the test level. | |||
Table 3
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The device is intended for use in the electromagnetic environment specified below.The customer or the user of the device should assure that it is used in such an environment. | |||
| Immunity test | IEC 60601 Test level | Compliance level | Electromagnetic environment - guidance |
| Conducted RF IEC 61000-4-6Radiated RF IEC 61000-4-3 | 3 Vrms150 kHz to80 MHz3 V/m80 MHz to2.5 GHz | not applicable3 V/m | Portable and mobile RF communications equipment should be used no closer to any part of the device, including cables, than the recommended separation distance calculated from the equation appropriate for the frequency of the transmitter.Recommended separation distances: d = 1.2 d = 1.2 80 MHz to 800 MHz d = 2.3 800 MHz to 2.7 GHzwhere, P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer, d is the recommended separation distance in meters (m). Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey, ^a should be less than the compliance level in each frequency range ^b . Interference may occur in the vicinity of equipment marked with the following symbol: [IMAGE] |
| NOTE 1: At 80 MHz and 800 MHz, the higher frequency range applies.NOTE 2: These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||
| a) Field strengths from fixed transmitters, such as base stations for radio (cellular / cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the device is used exceeds the applicable RF compliance level above, the device should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as re-orienting or relocating the device.b) Over the frequency range 150 kHz to 80 MHz, field strengths should be less than 3V/m. | |||
Table 4
| Recommended separation distances between portable and mobile RF communications equipment and the device. | |||
| The device is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the device can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and the device as recommended below, according to the maximum output power of the communications equipment. | |||
| Rated maximum output power of transmitter (W) | Separation distance according to frequency of transmitter (m) | ||
| 150 kHz to 80 MHz d = 1.2 | 80 MHz to 800 MHz d = 1.2 | 800 MHz to 2.5 GHz d = 2.3 | |
| 0.12 0.12 0.23 | |||
| 0.1 0.38 0.38 0.73 | |||
| 1 1.2 1.2 2.3 | |||
| 10 3.8 3.8 7.3 | |||
| 100 12 12 23 | |||
| For transmitters rated at a maximum output power not listed above, the recommended separation distance d in metres (m) can be estimated using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer.NOTE 1: At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies.NOTE 2: These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||
Guangdong Transtek Medical Electronics Co., Ltd.
Address: Zone A, No.105, Dongli Road, Torch Development District, Zhongshan Guangdong 528437 China
Tel: 86 760 88282930
Fax: 86 760 88282982
Http://www.transtek.cn
E-mail: info@transtek.cn
EU Declaration of Contormity
We Guangdong Transtek Medical Electronics Co., Ltd.
Zone A, No.105, Dongli Road, Torch Development District, 528437
Zhongshan, Guangdong, PEOPLE'S REPUBLIC OF CHINA
declare under our sole responsibility that the product
Product Name: Blood Pressure Monitor
Model name: TMB-1585-BT
Software: V01
Accessories: Adapter(Model:)KH0601000EW; battery 4*AAA
comply with the following directives and regulations:
2014/53/EU (The Radio Equipment Directive)
For the evaluation of the compliance with these Directives and Regulations, the following standards/requirements were applied:
| Safety Article3.1(a) | |
| EMC Article3.1(b) | Draft EN 301 489-1 V2.2.0, Draft EN 301 489-17 V3.2.0 |
| Radio Article3.2 | EN 300 328 V2.1.1, Draft EN 303 345 V.1.1.7 |
| Health Article3.1(a) | EN 62479: 2010 |
The conformity assessment procedure as referenced in Article 17 and detailed in Anne Radio equipment Directive has been followed with the involvement of a notified body: Notified Body: TUV SUD BABT
Responsible for making this declaration is the:
Manufacturer □ Authorised representative established within the EU
Person responsible for making this declaration
Print name/Title: Kevin Tan/R&D management
Guangdong Transktek
Medical Electronics Co., Ltd 2017.9.20
Kevin Tan
SOEHNLE

CONNECT
measure, collect, analyse, advise

text_image
QR code image containing encoded data, no visible human-readable textAndroid™: 4.4.4
iOS:9.0
Bluetooth*: 4.0

Download on the
App Store

GET IT ON
Google Play
Distributed by
LEIFHEIT AG
Leifheitstraße 1
56377 Nassau/Germany
www.soehnle.com

0123




