BM 77 - Blood pressure monitor BEURER - Free user manual and instructions
Find the device manual for free BM 77 BEURER in PDF.

Frequently Asked Questions - BM 77 BEURER
Download the instructions for your Blood pressure monitor in PDF format for free! Find your manual BM 77 - BEURER and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. BM 77 by BEURER.
USER MANUAL BM 77 BEURER
Quelle: WHO, 1999 (World Health Organization)
Ruheindikator
Read these instructions for use carefully and keep them for later use, be sure to make them accessible to other users and observe the information they contain.
Dear customer,
Thank you for choosing a product from our range. Our name stands for high-quality, thoroughly tested products for applications in the areas of heat, weight, blood pressure, body temperature, pulse, gentle therapy, massage, beauty, air and baby.
With kind regards
Your Beurer team
Contents
- Included in delivery.. 23
- Signs and symbols 24
- Intended use 24
4.Warnings and safety notes 25 - Device description 28
- Initial use 29
- Usage 30
- Cleaning and maintenance 39
- Accessories and replacement parts 39
10.What if there are problems? 39 - Disposal 40
- Specifications 41
13.Warranty/service. 42
1. Included in delivery
Check that the exterior of the cardboard delivery packaging is intact and make sure that all contents are present. Before use, ensure that there is no visible damage to the device or accessories and that all packaging material has been removed. If you have any doubts, do not use the device and contact your retailer or the specified Customer Service address.
1x upper arm blood pressure monitor
1x upper arm cuff (24-40 cm)
1x instructions for use
1x brief instructions
1x storage bag
1x USB cable
1x cuff holder
4x 1.5 V AAA LR03 batteries
2. Signs and symbols
The following symbols are used on the device, in these instructions for use, on the packaging and on the type plate for the device:
| WARNING indicates a hazardous situation which, if not avoided, could result in death or serious injury. | |
| CAUTION indicates a hazardous situation which, if not avoided, may result in minor or moderate injury. | |
| Product information Note on important information | |
| Observe the instructions Read the instructions before starting work and/or operating devices or machines | |
| Isolation of applied parts, type BF Galvanically isolated application part (F stands for “floating”); meets the requirements for leakage currents for type BF | |
| Direct current The device is suitable for use with direct current only | |
| Disposal in accordance with EC Directive WEEE (Waste Electrical and Electronic Equipment) |
| Pb Cd Hg | Do not dispose of batteries containing hazardousous substances with household waste |
| 21 PAP | Dispose of packaging in an environmentally friendly manner |
| Manufacturer | |
| Storage / Transport | Permissible storage and transport temperature and humidity |
| Operating | Permissible operating temperature and humidity |
| IP21 | Protected against foreign objects equal to or greater than 12.5 mm in diameter and against vertically falling drops of water |
| SN | Serial number |
| CE0483 | CE labellingThis product satisfies the requirements of the applicable European and national directives |
3. Intended use
Intended use
The blood pressure monitor is intended for the fully automatic, non-invasive measurement of arterial blood pressure and pulse values on the upper arm.
Target group
It is designed for self-measurement by adults in the home environment and is suitable for users whose upper arm circumference is within the range printed on the cuff. The device is also ideal for taking blood pressure measurements on women who are pregnant. This was successfully tested as part of a clinical study (Tempestas, Institut für Medizinische Forschung, Cloppenburg, Germany).
Indication/clinical benefits
The user can record their blood pressure and pulse values quickly and easily using the device. The recorded values are classified according to internationally applicable guidelines and evaluated graphically. Furthermore, the device can detect any irregular heart beats that occur during measurement and inform the user via a symbol in the display. The device saves the recorded measurements and can also output average values of previous measurements. This blood pressure monitor also has a haemodynamic stability display, which is referred to as a resting indicator throughout these instructions for use. This shows whether you, and consequently your circulatory system, are sufficiently at rest when the blood pressure measurement is being taken, and whether the measured blood pressure thus corresponds to your blood pressure when at rest. Read more about this under "Resting indicator" in the section on using the device. The recorded data can provide healthcare service providers with support during the diagnosis and treatment of blood pressure problems, and therefore plays a part in the long-term monitoring of the user's health.
4.Warnings and safety notes
Contraindications
- Do not use the blood pressure monitor on newborns, children or pets.
- People with restricted physical, sensory or mental skills should be supervised by a person responsible for their safety and receive instructions from this person on how to use the device.
- If you have any of the following conditions, it is essential you consult your doctor before using the device: cardiac arrhythmia, circulatory problems, diabetes, pregnancy, pre-eclampsia, hypotension, chills, shaking.
- People with pacemakers or other electrical implants should consult their doctor before using the device.
- The blood pressure monitor must not be used in connection with a high-frequency surgical unit.
- Do not use the cuff on people who have undergone a mastectomy.
- Do not place the cuff over wounds as this may cause further injury.
- Make sure that the cuff is not placed on an arm in which the arteries or veins are undergoing medical treatment, e.g. intravascular access or intravascular therapy, or an arteriovenous (AV) shunt.

General warnings
- The measured values taken by you are for your information only - they are no substitute for a medical examination. Discuss the measured values with your doctor and never make your own medical decisions based on them (e.g. regarding dosages of medicines).
- The device is only intended for the purpose described in these instructions for use. The manufacturer is not liable for damage resulting from improper or incorrect use.
- Using the blood pressure monitor outside your home environment or whilst on the move (e.g. whilst travelling in a car, ambulance or helicopter, or whilst undertaking physical activity such as playing sport) can influence the measurement accuracy and cause incorrect measurements.
- Cardiovascular diseases may lead to incorrect measurements or have a detrimental effect on measurement accuracy.
- Do not use the device at the same time as other medical electrical devices (ME equipment). This could lead to a malfunction of the device and/or an inaccurate measurement.
- Do not use the device outside of the specified storage and operating conditions. This could lead to incorrect measurements.
- Only use the cuffs included in delivery or cuffs described in these instructions for use for the device. Using another cuff may lead to measurement inaccuracies.
- Please note that when inflating the cuff, the functions of the limb in question may be impaired.
-
Do not perform measurements more frequently than necessary. Due to the restriction of blood flow, some bruising may occur.
-
During the blood pressure measurement, the blood circulation must not be stopped for an unnecessarily long time. If the device malfunctions remove the cuff from the arm.
- Place the cuff on your upper arm only. Do not place the cuff on other parts of the body.
- The air line poses a risk of strangulation for small children. Furthermore, included small parts pose a risk of suffocation for small children if swallowed. They should therefore always be supervised.

General precautions
- The blood pressure monitor is made from precision and electronic components. The accuracy of the measurements and service life of the device depend on its careful handling.
- Protect the device from impacts, humidity, dirt, marked temperature fluctuations and direct sunlight.
- Ensure the device is at room temperature before measuring. If the measuring device has been stored close to the maximum or minimum storage and transport temperatures and is placed in an environment with a temperature of 20^ , it is recommended that you wait approx. 2 hours before using the measuring device.
- Do not drop the device.
- Do not use the device in the vicinity of strong electromagnetic fields and keep it away from radio systems or mobile telephones.
- We recommend that the batteries be removed if the device is not to be used for a prolonged period of time.
- Avoid any mechanical restriction, compression or bending of the cuff line.
Measures for handling batteries

- If your skin or eyes come into contact with battery fluid, rinse the affected areas with water and seek medical assistance.
- Choking hazard! Small children may swallow and choke on batteries. Therefore, store batteries out of the reach of small children.
- Risk of explosion! Do not throw batteries into a fire.
- If a battery has leaked, put on protective gloves and clean the battery compartment with a dry cloth.
- Do not disassemble, open or crush the batteries.
- Observe the plus (+) and minus (-) polarity signs.
- Protect the batteries from excessive heat.
- Do not charge or short-circuit batteries.
- If the device is not to be used for a relatively long period, take the batteries out of the battery compartment.
- Use identical or equivalent battery types only.
Always replace all batteries at the same time. - Do not use rechargeable batteries!

Notes on electromagnetic compatibility
- The device is suitable for use in all environments listed in these instructions for use, including domestic environments.
- The use of the device may be limited in the presence of electromagnetic disturbances. This could result in issues such as error messages or the failure of the display/device.
-
Avoid using this device directly next to other devices or stacked on top of other devices, as this could lead to faulty operation. If, however, it is necessary to use the device in the manner stated, this device as well as the other devices must be monitored to ensure they are working properly.
-
The use of accessories other than those specified or provided by the manufacturer of this device can lead to an increase in electromagnetic emissions or a decrease in the device's electromagnetic immunity; this can result in faulty operation.
- Failure to comply with the above can impair the performance of the device.
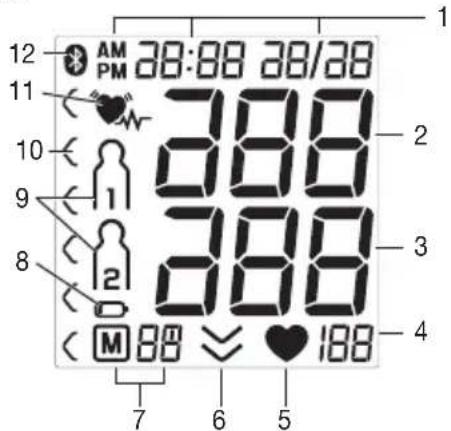
5. Device description
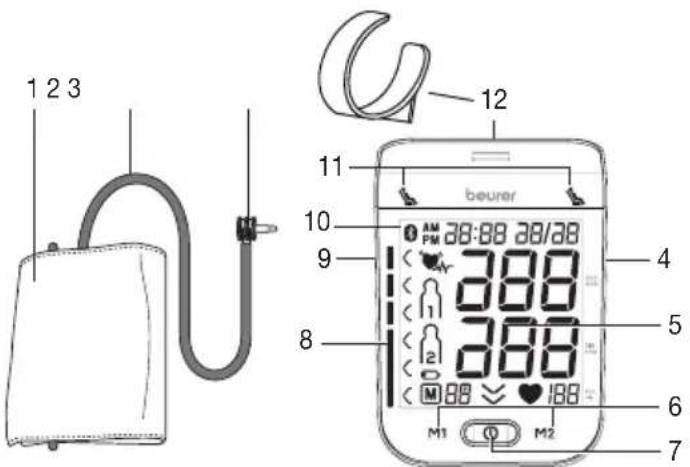
Blood pressure monitor and cuff
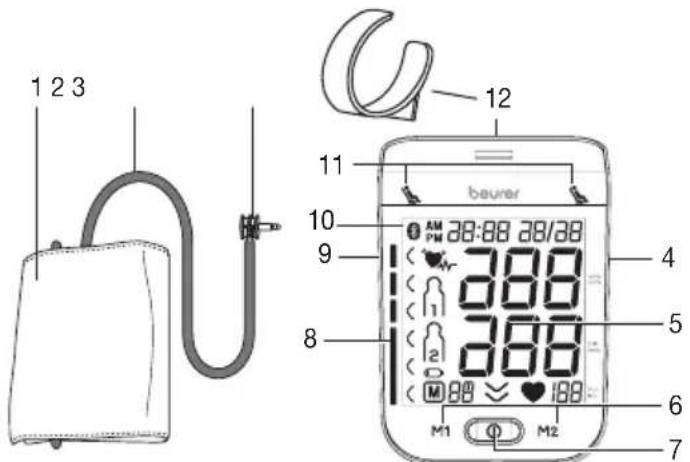
- Cuff
- Cuff line
- Cuff connector
- Connection for mains part and USB interface
- Display
- Memory buttons M1/M2
- Start/stop button ①
- Risk indicator
- Connection for cuff connector (left-hand side)
- Symbol for Bluetooth® transfer
- Resting indicator display
- Cuff holder
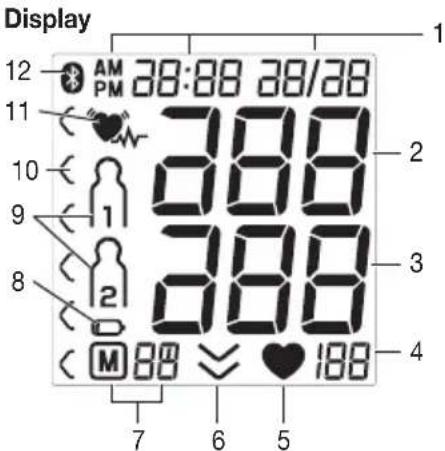
- Date / time
- Systolic pressure
- Diastolic pressure
- Pulse value
- Pulse symbol
- Release air
- Number of memory space/memory display average value (A), morning (B), evening (C)
- Battery replacement symbol
- User memory
- Risk indicator
11.Cardiac arrhythmia symbol - Symbol for Bluetooth® transfer
6. Initial use
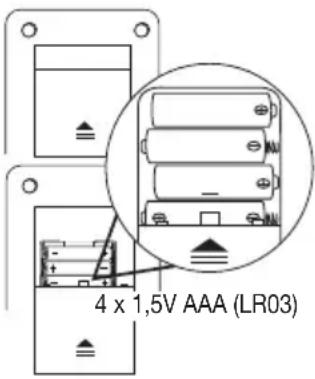
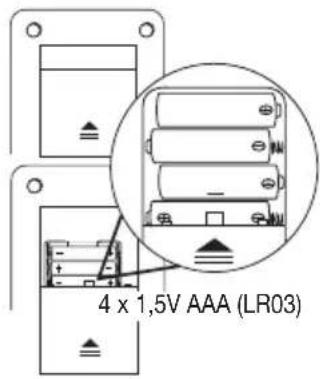
Inserting the batteries
- Remove the battery compartment lid on the rear of the device.
- Insert four 1.5 V AAA (alkaline type LR03) batteries. Make sure that the batteries are inserted the correct way round.
- Close the battery compartment lid again carefully.
- All display elements are briefly displayed, 2 flashes in the display. Set the date and time as described below.
If the battery replacement symbol is permanently displayed, you can no longer perform any measurements and must replace all batteries. Once the batteries have been removed from the device, the date and time must be set again. Any saved measurements are retained.
Operation with the mains part
You can alternatively also operate this device with a mains part (not included in delivery). However, before connecting the device with the mains part, please ensure that you have removed the batteries from the device. During mains operation, there must not be any batteries in the battery compartment, as this could damage the device.
- To avoid any potential damage, the device may only be operated with a mains part that meets the specifications described in the chapter "Technical specifications".
- Furthermore, the mains part must only be connected to the mains voltage that is specified on the type plate.
- First, remove the protective cap of the mains part connector - located on the rear of the device - and insert the mains part into the connection intended for this purpose.
- Then insert the mains plug of the mains part into the mains socket.
- After using the blood pressure monitor, unplug the mains part from the mains socket first and then disconnect it from the blood pressure monitor. As soon as you unplug the mains part, the blood pressure monitor loses the date and time setting but the saved measured values are retained.
Set the hour format, date, time and Bluetooth® settings
It is essential to set the date and time. Otherwise, you will not be able to save your measured values correctly with a date and time and access them again later.
There are two different ways to access the menu from which you can adjust the settings:
- Before initial use and after each time you replace the battery:
When inserting batteries into the device, you will be taken to the relevant menu automatically.
- If the batteries have already been inserted:
With the device switched off, press and hold the START/ STOP button for approx. 5 seconds.
In this menu you can adjust the following settings in succession:

The hour format now flashes on the display.
- Choose the desired hour format with the M1/M2 memory buttons and confirm with the Start/stop button ①

The year flashes on the display.
- Choose the desired year with the M1/M2 memory buttons and confirm with the Start/stop button ①
The month flashes on the display.
- Choose the desired month with the M1/M2 memory buttons and confirm with the Start/stop button ①
The day flashes on the display.
- Choose the desired day with the M1/M2 memory buttons and confirm with the Start/stop button ①
If the hour format is set as 12h, the day/month display sequence is reversed.
The hour flashes on the display.
- Choose the desired hour with the M1/M2 memory buttons and confirm with the Start/stop button ①
The minute flashes on the display. - Choose the desired minute with the M1/M2 memory buttons and confirm with the Start/stop button ①
The Bluetooth® symbol flashes on the display.
- Use the M1/M2 memory buttons to choose whether automatic Bluetooth® data transfer is activated (Bluetooth® symbol flashes) or deactivated (Bluetooth® symbol is not shown) and confirm with the START/STOP button ①
Bluetooth® transfers will reduce the battery life.
7. Usage
General rules when measuring blood pressure yourself
-
In order to generate as informative a profile of the progression of your blood pressure as possible and ensure that the measured values can be compared, you should measure your blood pressure regularly and always at the same times of day. It is recommended that you measure your blood pressure twice a day: once in the morning after getting up and once in the evening.
-
You should always carry out the measurement when you are sufficiently physically rested. You should therefore avoid taking measurements during stressful periods.
- Do not take a measurement within 30 minutes of eating, drinking, smoking or exercising.
- Before the initial blood pressure measurement, make sure always to rest for 5 minutes.
- Furthermore, if you want to take several measurements in succession, make sure always to wait for at least 1 minute between the individual measurements.
- Repeat the measurement if you are unsure of the measured value.
Attaching the cuff
- Fundamentally, blood pressure can be measured on both arms. Certain deviations between the measured blood pressure on the right arm and left arm are due to physiological causes and completely normal. You should always perform the measurement on the arm with the highest blood pressure values. Before starting self-measurement, consult your doctor in this regard. From this point on, always take measurements on the same arm.
- The device may only be operated with one of the following cuffs. This should be selected in accordance with your upper arm circumference. The fit should be checked before measurement using the index mark described below.
| Ref. no. | Designation Arm | circumference |
| 163.764* U | universal cuff 24-40 cm |
-
Included in standard delivery
-
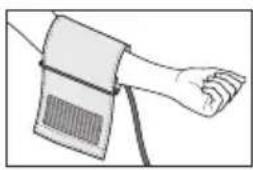
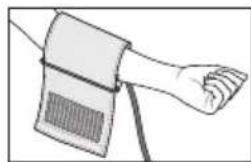
Place the cuff onto the bare upper arm. The circulation of the arm must not be hindered by tight clothing or similar.
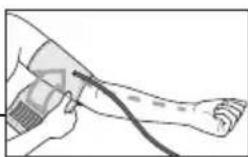
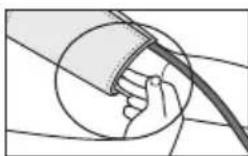
- The cuff must be placed on the upper arm so that the bottom edge is positioned 2 - 3cm above the elbow and over the artery. The line should point to the centre of the palm here.
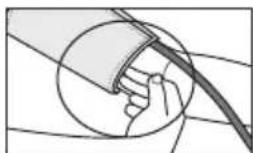
- Guide the end of the cuff that is sticking out through the metal ring, fold it back over the arm and close the cuff using the hook-and-loop fastener. The cuff should be fastened tightly, but not too tightly, so that two fingers can still fit under the closed cuff.
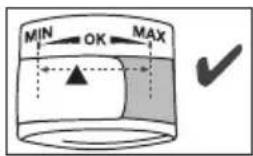
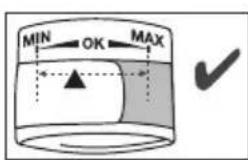
- The cuff is suitable for you if the index mark (▼) is within the OK range after fitting the cuff.
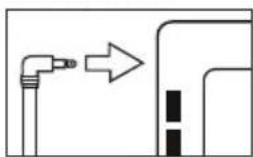
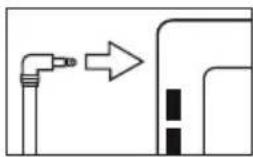
- Now insert the cuff line into the connection for the cuff connector.
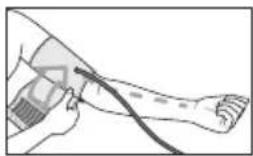
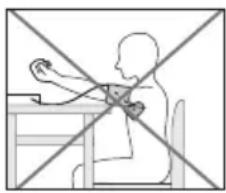
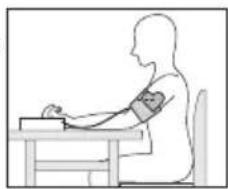
Adopting the correct posture
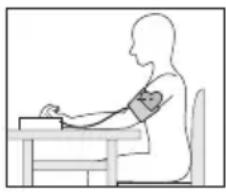
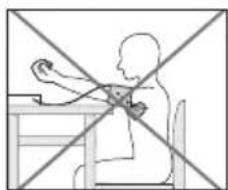
- To carry out a blood pressure measurement, make sure you are sitting upright and comfortably. Lean back and place your arm on a surface. Do not cross your legs. Place your feet next to each other flat on the floor.
- Always make sure that the cuff is at heart level.
- To avoid distorting the measurement, you should remain as still as possible during the measurement and not speak.
Performing the blood pressure measurement
W
- To start the blood pressure monitor, press the Start/stop button ① All display elements are briefly displayed.
The blood pressure monitor will begin the measurement automatically after approx. 3 seconds. The cuff inflates as the first step..
Measuring can be cancelled at any time by pressing the Start/stop button ①.
- Then the air pressure in the cuff is slowly released. Now the measurement starts. As soon as a pulse is found, the pulse symbol is displayed.
- If a tendency towards high blood pressure can already be identified, it is possible that the cuff may inflate again to a higher pressure level whilst the air is being released.
- The remaining air is released quickly once the measurement is complete.
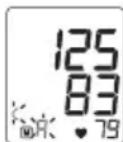
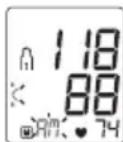
Systolic pressure, diastolic pressure and pulse measurements are displayed. On the display, the resting indicator lights up green or red depending on the assessment of your haemodynamic stability.


- E_ appears if the measurement could not be performed properly. In this case, please read the section "What if there are problems?".
- Now select the desired user memory by pressing the M1 or M2 memory buttons. If you do not select a user memory, the measurement is stored in the most recently used user memory. The relevant symbol or appears on the display.
- If the Bluetooth® function has been activated, the data transfer to the "beurer HealthManager" app starts after confirming the user selection using the START/STOP button.
- The Bluetooth® symbol flashes on the display. The device now attempts to connect to the app for approx. 30 seconds.
- The Bluetooth® symbol stops flashing as soon as the connection is established. All measurement data is transferred to the app. Once the data transfer is successfully complete, the device switches off automatically.
- If a connection to the app cannot be established after 30 seconds, the Bluetooth® symbol goes out and the device switches off automatically after 3 minutes.
- If you forget to turn off the device, it will switch off automatically after approx. 3 minutes. In this case too, the value is stored in the selected or most recently used user memory.
Transfer of measurements via Bluetooth®
In addition to displaying and saving your measurements locally on the device, you have the option of transferring your measurements to your smartphone using Bluetooth® low energy technology.
The software can be downloaded free of charge from the download area under Service at www.beurer.com.
System requirements for the "beurer HealthManager" app
- iOS ≥ 10.0, Android™ ≥ 5.0
-Bluetooth 值 ≥4.0
List of compatible devices:

To transfer the measured values, proceed as follows:

Step 1: BM 77
Activate the Bluetooth® function on your device as described in section "Setting the hour format, date, time and Bluetooth".

Step 2: "beurer HealthManager" app
In the "beurer HealthManager" app, add the BM 77 under Settings / Devices and follow the instructions.
Also note the following information:
- Ensure that the "beurer HealthManager" app on your smartphone is always activated and launched when you start the data transfer on the device.
- You can tell that the data transfer is in progress by the Bluetooth® symbol shown on the display.
- If your smartphone has a protective cover, remove it to ensure that there is no interference during the transfer.
Transfer of measurements via USB
Alternatively, you can also transfer your measured values to the PC.
To do this, you require a commercially available USB cable (included in delivery) and the "beurer HealthManager" PC software. This can be downloaded free of charge from the download area under Service at www.beurer.com.
System requirements for the „ beurer HealthManager“ PC software
from Windows 8.1
from USB 2.0 (Type-A)
To transfer the measured values, proceed as follows:
- Connect your blood pressure monitor to your PC using the USB cable provided. Please note that you cannot start a data transfer to the PC whilst a measurement is being taken.
- PC is shown on the display. Begin the data transfer in the "beurer HealthManager" PC software.
- During the data transfer, an animation is shown on the display. A successful data transfer is displayed as in figure 1. If the data transfer is unsuccessful, an error message appears as in figure 2. In this case, interrupt the PC connection and start the data transfer again.
After 30 seconds of not being in use or if communication with the PC is interrupted, the blood pressure monitor switches itself off automatically.
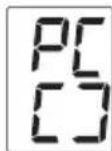
figure 1

figure 2
Evaluating the results
General information about blood pressure
- Blood pressure is the force with which the bloodstream presses against the arterial walls. Arterial blood pressure constantly changes in the course of a cardiac cycle.
-
Blood pressure is always stated in the form of two values:
-
The highest pressure in the cycle is called systolic blood pressure. This arises when the heart muscle contracts and blood is pumped into the blood vessels.
- The lowest is diastolic blood pressure, which is when the heart muscle has completely stretched back out and the heart fills with blood.
- Fluctuations in blood pressure are normal. Even during repeat measurements, considerable differences between the measured values may occur. One-off or irregular measurements therefore do not provide reliable information about the actual blood pressure. Reliable assessment is only possible when you perform the measurement regularly under comparable conditions.
Cardiac arrhythmia
This device can identify potential disruptions of the heart rhythm when measuring and, if necessary, indicates this after the measurement with the symbol. This can be an indicator for arrhythmia. Arrhythmia is an illness in which the heart rhythm is abnormal because of flaws in the bioelectrical system that regulates the heartbeat. The symptoms (skipped or premature heart beats, pulse being slow or too fast) can be caused by factors such as heart disease, age, physical make-up, excess stimulants, stress or lack of sleep. Arrhythmia can only be determined through an examination by your doctor. If the symbol
appears frequently, please consult your doctor. Only they are able to determine any arrhythmia during an examination.
Risk indicator
The measurements can be classified and evaluated in accordance with the following table.
However, these standard values serve only as a general guideline, as the individual blood pressure varies in different people and different age groups etc.
It is important to consult your doctor regularly for advice. Your doctor will tell you your individual values for normal blood pressure as well as the value above which your blood pressure is classified as dangerous.
The bar chart on the display and the scale on the device show which category the recorded blood pressure values fall into.
If the values of systole and diastole fall into two different categories (e.g. systole in the high normal category and diastole in the normal category), the graphical classification on the device always shows the higher category; for the example given this would be high normal.
| Blood pressure value category | Systole (in mmHg) | Diastole (in mmHg) | Action | |
| Level 3: severe hypertension | red ≥18 | 0≥110 | Seek medical attention | |
| Level 2: moderate hypertension | orange | 160-179 100 | -109 | Seek medical attention |
| Level 1: mild hypertension | yellow | 40–159.90–99 | 99 | Regular monitoring by doctor |
| High normal green | 130–139.85–89 | 89 | Regular monitoring by doctor | |
| Normal green | 120–129.80–84 Self-monitoring | |||
| Optimal green | <120<80 | Self-monitoring | ||
Source: WHO, 1999 (World Health Organization)
Resting indicator
One of the most common errors made when taking a blood pressure measurement is not ensuring that the user's circulatory system is sufficiently at rest when taking the measurement. In this case, the measured systolic and diastolic blood pressure values do not correspond to the blood pressure at rest. However, it is this blood pressure at rest that should be used to assess the measured values.
This blood pressure monitor uses integrated haemodynamic stability diagnostics (HSD) to measure the user's haemodynamic stability when taking the blood pressure measurement. This enables it to indicate whether the blood pressure was taken when the user's circulatory system was sufficiently at rest.
Resting indicator lights up GREEN

The measured blood pressure value was obtained when the user's circulatory system was sufficiently at rest and reliably corresponds to the blood pressure at rest.
| Resting indicator lights up RED | Indicates that the value was obtained when the user's circulatory system was not sufficiently at rest. The blood pressure values measured in this case generally do not correspond to the blood pressure at rest. The measurement should therefore be repeated after a period of physical and mental rest lasting at least 5 mins. |
| Resting indicator does not light up | During the measurement it was not possible to determine whether the user's circulatory system was sufficiently at rest. In this case too, the measurement should be repeated after a period of rest lasting at least 5 mins. |
If the user's circulatory system was not sufficiently at rest, this can be caused by various factors such as physical or mental strain or distraction, speaking, or experiencing cardiac arrhythmia during the measurement.
In an overwhelming number of cases, HSD will give a very good guide as to whether the user's circulatory system is rested when taking a blood pressure measurement.
However, certain patients suffering from cardiac arrhythmia or chronic mental conditions may remain haemodynamically unstable even in the long-term, something which persists even after repeated periods of rest. The accuracy of the results for the blood pressure at rest is reduced in these users.
Like any medical measurement method, the precision of HSD is limited and can lead to incorrect results in some cases. Nevertheless, the blood pressure measurements taken when the user's circulatory system is sufficiently at rest represent particularly reliable results.
Displaying and deleting measurements
The results of every successful measurement are stored together with the date and time. If there are more than 60 measurements, the oldest measurements are lost.
- To access memory recall mode, the blood pressure monitor must first be started. To do this press the Start/stop button ①
-
Within 3 seconds of the full-screen display appearing, select the desired user memory (_1 / _2) with the M1 or M2 memory button.
-
To view the measurements for user memory,press the M1 memory button.
- To view the measurements for user memory, press the M2 memory button.
If Bluetooth® is activated (the 3 symbol appears on the display), measurements are automatically transferred. Press the M1/M2 button to cancel data transfer. The device will show average values. The 3 symbol is no longer shown.
If you have selected user memory1, the M1 memory button must be pressed.
If you have selected user memory2, the M2 memory button must be pressed.
R flashes on the display.
The average value of all saved measured values in this user memory is displayed.
- Press the relevant memory button (M1 or M2).
flashes on the display.
The average value of the morning measurements for the last 7 days is displayed (morning: 5.00 a.m. - 9.00 a.m.).
- Press the relevant memory button (M1 or M2).
Pn flashes on the display.
The average value of the evening measurements for the last 7 days is displayed (evening: 6.00 p.m. - 08.00 p.m.).

- When the relevant memory button (M1 or M2) is pressed again, the last individual measurement is displayed (in this example, measurement 03).

- When the relevant memory button (M1 or M2) is pressed again, you can view your individual measurements.
- To switch the device off again, press the Start/stop button 1 r wait 30 sec.
You can exit the menu at any time by pressing the Start/stop button
- To delete all measurements saved for a particular user, first select the user as described by pressing memory button M1/M2.
- R flashes on the display and the average value of all saved measured values in this user memory is displayed.
- Depending on which user memory was selected, press and hold memory button M1 or M2 for approx. 5 seconds.
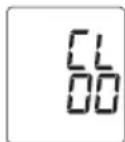
- CL00 appears on the display. All the values in the current user memory are deleted.
To clear individual measurements from the relevant user memory, you must first select a user memory.
- Start the retrieval of the individual measured values.
- Press and hold the memory button M1/M2 for 5 seconds (depending on the user memory you are in).
- The selected value is deleted. The device briefly displays CL 00.
- If you would like to delete other values, repeat the process described above.
You can switch off the device at any time by pressing the START/STOP button.
8. Cleaning and maintenance
- Clean the device and cuff carefully using a slightly damp cloth only.
- Do not use any cleaning agents or solvents.
- Under no circumstances hold the device and cuff under water, as this can cause liquid to enter and damage the device and cuff.
- If you store the device and cuff, do not place heavy objects on the device and cuff. Remove the batteries. The cuff line should not be bent sharply.
9. Accessories and replacement parts
Accessories and replacement parts are available from the corresponding service address (according to the service address list). Please state the corresponding order number.
| Designation Item number and/or | or- der number |
| Universal cuff (24-40 cm) 163.764 | |
| Mains part (EU) | 071.95 |
| USB cable | 162.928 |
10. What if there are problems?
| Error message | Possible cause Solution | |
| EE | You moved or spoke during the measurement. | Please wait one minute and repeat the measurement. Ensure that you do not speak or move during the measurement. |
| Er | The measured blood pressure values are outside the measurement range. | |
| E1 | The cuff was not attached correctly, the cuff line has not been inserted properly, or inflation takes longer than 15 seconds. | Please wait one minute and repeat the measurement, making sure to observe the information in chapter “Attaching the cuff”. Please also check whether the cuff line has been inserted properly and ensure that neither your arm nor other heavy objects are pressing on the line, and that the line is not bent. |
| E2 | The inflation pressure is higher than 300 mmHg. | Please take another measurement to check whether the cuff can be correctly inflated. Make sure that neither your arm nor other heavy objects are pressing on the line, and that the line is not bent. |
| E3 | An error occurred when saving the measurement. | Please wait one minute and repeat the measurement. Ensure that you do not speak or move during the measurement. |
| E4 | Unable to transfer the data via Blue-tooth®. | Please observe the information in chapter “Trans-fer of measurements via Bluetooth®”. |
| PcEr | Unable to transfer the data to the PC. | Please observe the information in chapter “Trans-fer of measurements via USB”. |
| O | The batteries are almost empty. | Insert new batteries into the device. |
11.Disposal
Repairing and disposing of the device
- Do not repair or adjust the device yourself. Proper operation can no longer be guaranteed in this case.
- Do not open the device. Failure to comply will invalidate the warranty.
- Repairs must only be carried out by Customer Services or authorised retailers. Before making a claim, please check the batteries first and replace them if necessary.
- For environmental reasons, do not dispose of the device in household waste at the end of its service life. Dispos of the device at a suitable local collection or recycling point in your country. Dispos of the device in accordance with EC Directive - WEEE (Waste Electrical and Electronic Equipment). If you have any questions, please contact the local authorities responsible for waste disposal.

Disposing of the batteries
- The empty, completely flat batteries must be disposed of through specially designated collection boxes, recycling points or electronics retailers. You are legally required to dispose of the batteries.
- The codes below are printed on batteries containing harmful substances: Pb = Battery contains lead, Cd = Battery contains cadmium, Hg = Battery contains mercury.
12. Specifications
Device
| Model no. BM 77 | |
| Type BM 75 | |
| Measurement method | Oscillometric, non-invasive blood pressure measurement on the upper arm |
| Measurement range | Cuff pressure 0 – 299 mmHg, systolic 30–260 mmHg, diastolic 30–260 mmHg, Pulse 40–199 beats/minute |
| Display accuracy | Systolic ± 3 mmHg, diastolic ± 3 mmHg, pulse ± 5 % of the value shown |
| Measurement inaccuracy | Max. permissible standard deviation according to clinical testing: systolic 8 mmHg /diastolic 8 mmHg |
| Memory 2 x 60 memory spaces | |
| Dimensions L 175 mm x W 117 mm x H 50 mm | |
| Weight Approx. 518 g (without batteries, with cuff) | |
| Cuff size 24 to 40 cm | |
| Permissible operating conditions | +10 °C to + 40 °C, 15 % - 85 % relative air humidity (non-condensing) |
| Permissible storage and transport conditions | - 10 °C to + 60 °C, 10 % - 90 % relative air humidity, 700 -1013 hPa ambient pressure |
| Power supply | 4x1,5V — — AAA batteries |
| Battery life | For approx. 120 measurements, depending on the blood pressure level and/or pump pressure |
| Classification | Internal supply, IP21, no AP or APG, continuous operation, application part type BF |
| Data transfer via Bluetooth® wireless technology | The blood pressure monitor uses Blue-tooth® low energy technology, 2402MHz - 2480MHz frequency band, Transmission power max. 4.54 dBm, Compatible with Bluetooth® 4.0 smart-phones/tablets |
The serial number is located on the device or in the battery compartment.
Technical information is subject to change without notification to allow for updates.
- This unit is in line with European Standard EN 60601-1-2 (in accordance with CISPR 11, IEC61000-3-3, IEC61000-4-2, IEC61000-4-3, IEC61000-4-4, IEC61000-4-5, IEC61000-4-6, IEC61000-4-8, IEC61000-4-11) and is subject to particular precautions with regard to electromagnetic compatibility (EEC). Please note that portable and mobile HF communication systems may interfere with this unit.
- This device is in line with the EU Medical Devices Directive 93/42/EEC, the "Medizinproduktegesetz" (German Medical Devices Act) and the standards EN 1060-1 (non-invasive sphygmomanometers, Part 1: General requirements), EN 1060-3 (non-invasive sphygmomanometers, Part 3: Supplementary requirements for electro-mechanical blood pressure measuring systems) and IEC 80601-2-30 (Medical electrical equipment - Part 2 - 30: Particular requirements for the safety and essential performance of automated non-invasive blood pressure monitors).
- The accuracy of this blood pressure monitor has been carefully checked and developed with regard to a long useful life. If using the device for commercial medical purposes, it must be regularly tested for accuracy by appropriate means. Precise instructions for checking accuracy may be requested from the service address.
- We hereby confirm that this product complies with the European RED Directive 2014/53/EU.
The CE Declaration of Conformity for this product can be found under:
Mains part
Model no. LXCP12-006060BEH
| Input 100-240V,50-60Hz,0.5Amax | |
| Output 6 V DC, 600 mA, only in connection with beurer blood pressure monitor. | |
| Supplier Shenzhen longxc power supply co., Ltd | |
| Protection This device is double insulated and protected against short circuit and overload by a primary thermal fuse. Make sure to take the batteries out of the compartment before using the mains part. | |
| Polarity of the DC voltage connection | |
| Double insulated / equipment class 2 | |
| Enclosures and ProtectiveCovers | Equipment enclosed to protect against contact with live parts, and with parts which can become live (finger, pin, hook test). The operator shall not contact the patient and the output plug of AC mains part simultaneously. |
13. Warranty/service
Beurer GmbH, Söflinger Straße 218, 89077 Ulm, Germany (hereinafter referred to as "Beurer") provides a warranty for this product, subject to the requirements below and to the extent described as follows.
The warranty conditions below shall not affect the seller's statutory warranty obligations which ensue from the sales agreement with the buyer. The warranty shall apply without prejudice to any mandatory statutory provisions on liability.
Beurer guarantees the perfect functionality and completeness of this product.
The worldwide warranty period is 5 years, commencing from the purchase of the new, unused product from the seller.
The warranty only applies to products purchased by the buyer as a consumer and used exclusively for personal purposes in the context of domestic use. German law shall apply.
During the warranty period, should this product prove to be incomplete or defective in functionality in accordance with the following provisions, Beurer shall carry out a repair or a replacement delivery free of charge, in accordance with these warranty conditions.
If the buyer wishes to make a warranty claim, they should approach their local retailer in the first instance: see the attached "International Service" list of service addresses.
The buyer will then receive further information about the processing of the warranty claim, e.g. where they can send the product and what documentation is required.
A warranty claim shall only be considered if the buyer can provide Beurer, or an authorised Beurer partner, with a copy of the invoice/purchase receipt, and the original product.
The following are explicitly excluded from this warranty:
- deterioration due to normal use or consumption of the product;
- accessories supplied with this product which are worn out or used up through proper use (e.g. batteries, rechargeable batteries, cuffs, seals, electrodes, light sources, attachments and nebuliser accessories);
- products that are used, cleaned, stored or maintained improperly and/or contrary to the provisions of the instructions for use, as well as products that have been opened, repaired or modified by the buyer or by a service centre not authorised by Beurer;
- damage that arises during transport between manufacturer and customer, or between service centre and customer;
- products purchased as seconds or as used goods;
- consequential damage arising from a fault in this product (however, in this case, claims may exist arising from product liability or other compulsory statutory liability provisions).
Repairs or an exchange in full do not extend the warranty period under any circumstances.
FRANÇAIS

Chere cliente, cher client,
Mesures relatives aux piles

Source: WHO, 1999 (World Health Organization)
Voyant de repos
Fuente: WHO, 1999 (World Health Organization)
Indicador de calma
Entrada 100-240V,50-60Hz,0.5Amax
Fase 2: App "beurer HealthManager"
Fonte: WHO, 1999 (World Health Organization)
Kaynak: WHO, 1999 (World Health Organization)
Sukunet gÖstergesi
Giris 100-240V,50-60Hz,0.5Amax
Oe Mepe IpeoctopoxHocTn
- Pπb6op ΠIa I3MpeHnK pOBaHOrO daBJIeHn COCToT n3 BbICOKOTOchIx 3JIeKTpoHHbIX KOMIOHeHTOB. ToHOCt b I3-MpeHn I cPcK cnyXbI np6bopa 3aBucrT OT 6epeXHoro 6paueHn C Hm.
3aunuane npnbop ot ynapOB, BlaXHocTN, 3arp3HeHnCnIbHbIX KOle6aHm TeMnpaTypbl INPpMbIX COJIHeHbIX Jy-yei.
-Дя npoBeHnI3MepeHn TeMnepaTpy np6bopa DoJXHa COOTBcTcBOBaT KOMHaTHOJ. Ecl np6op xpaHnCra npi TemnepaType, 6n3KoN K MaKcMaJIbHOu Nm MInHMaJIbHOI TemnepaType XpaHEnH N TpaHCnOpTIpOBKn, n 6bl IomeUeH B cpey C TemnepaTpyo 20°C, peKOMeHnyETcnoJIOxJaTa b OKoNo 2 Yacob nepeero nCNOJIb3OBAHNem. - He pohnyte npnbop.
He nCnoB3yIte np6Op B6Jn3n CnIbHbIX 3JIeKTPomarHHTbIX noJe, depKInTe erO Ha 3HaunTeJbHom pacCToHHNOT paAnopapatypblnn MoBnbHbIX TeJefoHOB. - Ecni npi6op dInTeJbHoe BpeMr He nCIOJIb3yeTcra, N3BJeKnTe 6aTapeKeKn.
- Ⅲ36eraIte MexaHueCKoro cyXeHn, CdaBImBaHn nn Crn-6aHn MaHKeTbl.
Yka3aHnno 06paueHHo c 6aTapeiKaAMn

-
Пи пonaдани кидкости n3 akkymyлгота на Коку ИИ N B rna3a Heo6xOДIMO npombyt b COOTBeTCTByUcIy yactOK 60JIbшm KOJIneCTBOM BObl N O6paNTbCЯ KВpaY.
-
Onachoctb nporlaTbBaHnme MeKnx DeTanei! MaJeHbKne Detn MOry TnporIOTntb 6aTapeuKN I NpOaNtBcRIMN. Po3tomy 6aTapeuKN Heo6xOdmo XpaHTb B HeDoCTyHOM DnIeTeN MeCte!
- OnachocTb B3pbIbA! He 6pocaTe 6aTaapeKn B OROHb.
- Ecnn 6atapeika notekna, ounctnte otcek nny 6atapeek cyxon canfeTKo, npedbapntbHO hadeB 3aunT-Hbie nepuATKn.
- He pa36bpaaiTe, He oTKpbBauiTe u He pa36bbauiTe 6aatapeiKu.

- 06paaIte BHMaHne Ha 3HaKN NOJApHOCTN: PIIOC (+) IN MInHyC (-).
- 3aunuau Te 6atapeyn ot neperpeba.
- He 3apjaKaIte 6aTapeiKn i He 3ambikaIe nx HakoPOTko.
- Ecni np6op dnteBhoe Bpem He nCnObl3yeTc, n3-BneKtTe 6aTaapeiKn.
- IcnoJb3yIe 6aTapeKn OndHoro Tnna nn paBHOceHNbIX TnIOB.
- 3amehnyTe BCE 6aTapeKn cpa3y.
He nCnoIb3yIte nepe3apJxJaEmblc aKKymJITopbl!

Yka3aHnI NO 3JIeKTPOMaTHNTHOIN COBmectUMOCTN
- Pnp6op npedha3Haen dIpa60Tb By yCNoBnX, nepeHcJIeHHbIX B HactoJeuHCTpyKuIN NO pPmHeHIO, B TOM YncIe B DOMaHnx ycNoBnX.
- Pn Hauuun 3neKtpomarHHTbIX NOMEX BO3MOXHOCTN NCNoIb3OBaHnepn6opa MoryT 6bITb OppaHueHb. B 3ToM Cnyae BO3MOXHbI, K npmepy, COo6UeHnO 6 OwN6kax nIN BbIXoN 3 cTpor ducnner nn camoro np6opa.
He nCnoIb3yIte daHHbI np6Op pIOM C dpYrIMn ycTPOINCTBAMn He yCTaHabInBaIte erO Ha dpYrme np6Opbl. 3To MoKcET npNBecTe K c6oRm B pa6Ote. Ecn np6Op BCE Je npNXODITcN cNoJIb3OBaTb B ONCAHHbIX BblIe ycIOBnX, CJeDyET H6JIHOdaTb 3a Hm N dpYrIMn ycTPOINCTBAMn, YTO6bl y6eITbC, YTO OHn pa6OtaOT HndNeKaUIM o6pa3OM.
-
PpimmeHne npHaadJeXHoCTe, otJnuaOuixxcr ot yka3aHbIX npOn3BOiTeIeM IIN npUnaRaembIX K daHHOMy np6Oppy, MoKet npBecTn K poCTy 3JIeKtpomarHHTbIX NOMEX IIN ocJa6IeHIO nomExocToHBOcTn np6Opa N TEM cMbIM Bb3BaTb C6oN B erO pa6Ote.
-
HecobioeHne daHHoro yka3aHnM MoKeT OtpuataIbHo cKa3aTbCra Ha pa6oUnx xapaKTepuCtNkax np6opa.
5. Onicahne yctpoiCTBa
Pp6op dIy I3MepeHnKPOBHOrO daBHeHn C MaHXeToI
- MaHxeta
2.卍naHr MaHKeTbI
3.LITEkep MaHKeTbI - Pa3bemДЯ ПОДКЛHOUeHЯ 6JIOKa PITaHЯ INHTepfec USB
5.Диспел - Khonk coxpanen M1/M2
- Khonka BKJI./BbIKI. ①
8.Индикатор рисka - Pa3bEm dIy ITeKepa MaHKeTbI (c JEBOc TOpOhbl)
- Cnmbon nepedaa nnHbix No Bluetooth 8
11.Диспели Иndикатopa coctoHnI nokor
12.ДерхателдmaMaHxeTbI
Aunnei:
1.Дata/BpeMЯ
2.CnctoJIneckoe daBHeHne
3.ДиacToJIuYeCKoeДaBJIeHne
4.ИЗмеренhoeЗаченипульca
5.CmboJ npIbca
6. Bbinyck Bo3dyxa
7.Homep qeyKn namrtn/INHnKauncpeHero 3haehn u3 coxpaehhhbx B narn(),yTPOm ()Beepom(P
8.CmBBoJ INHnKauCm CmeHb6TaapeKn
9.ПользователбckаяпамтБ
10.ИндikaTop риcka
11.CIMBOJ HApUeHn cepDeuHoro pNTMa
12. CnmboJ npepaun daHbIX no Bluetooth®
6.Подrostobkaкрабote
YctaHOBka 6atapeek
CHIMITE KpbIuKy OTeJeHnA Dn8 6aTapeek Ha 3aHne CToP oHe np6opa.
BCTaBbTe YeTbIpe 6aTapeKn 1,5 B AAA (aKaJIINHObBiE, TnLR03).O63aTeJbHo npocJeDHTe 3a TEM, YTObI 6aTaapeKu 6blN yCTaHOBJIeHbIC npaBnJIb-HOH NOJaPHOCTbIO B COOTBeTCTBnC MapKnPOBKO.
CHOBA TuaTeJIbHo 3aKpoIte KpbIuKy OTdEHeHnI dIg 6aTapeek.
Bce 3nemEnbI dncnpe OTO6pa3rTcHa KOpOTKoe BpemHa dncnlee 6ydt MIRATb HndkauncaNcTaHOBtTe DaTy IN Bpem, BblONHb ONCAHHbe Hxke DeiCTBn.
PnDInTeBHom OTo6paXeHm CmBOJa 3aMeHb6aTapeek
I npoBeHne H3MepeHn HeBO3MOxHO, CJeDyET 3aMHeHTb BCE 6aTapeKn. IocNe H3BLeueHn 6aTapeek N3I np6Opa DaTy n Bpemr Heo6XoIMO yCTaHaBJIbNAbTb 3aHOBO. CoXpaHeHHbE B NaMaTn pe3YJbTaTbI 3MepeHn He NCye3aOT.
IcnoB3OBAHHe C 6IOKOM NITaHn
Pn6op MoXHO TaKHe NCIOJb3ObaTb C 6I0KOM NtAHN (He BXoHIT B KOMnEKT NoCTaBKn). Ipeep noKJIoueHEm 6Ioka NtAHN yBeHITcB TOM, UTO n3 np6opa n3BLeueHb 6a-tapeKn. Pn NtAHMOT cETn B np6ope He DoJxHO 6bITb 6aTaapeek, iHaue Bo3MOXHO NOBpeJeHne np6opa.
- 4To6bI npedOTBpaNTb BO3MOxHbIE NOBpeXdHnA, nCNoJIb-3yInTe np6Op NCKIOHTeNbHO C 6NOKOM NTaHnA, COOTBeTCTByUOUM CNEuΦKAcuM, KOtOpbIe ONuCaHb I B rJaBe «TexHnueckne DaHHbe».
- BIOK NITaHnMoXHO NOIckHOaTH ToIbKO K CeTeBOMy Ha-npJxKeHnO, yKa3aHHOMy Ha dIpMehHO Ta6NIuKe.
- CHINMITE 3aunTHbI KONNaOc pa3bema nIg 6noka nITaHnHa 3aDHe CTOpOHe npBopa nIOKJIOnTe 6nOK nITaHnK InpEDyCMOTpeHHOMy dIg Hero pa3bemy.
- 3aTeM BCTaBbTe CeTeBOI uTEKepe 6LOKa nITaHnB pO3eTKy.
- Pocne nCnoJb3ObaHnnp6opa dIy I3MepeHnKpOBaHO rDaBHeHn CHaJaI OTKJIouHTe 6NOK NITAHn I3 PO3ETKN, a 3aTeM OTcoeHNHTe erO OT np6opa dIy I3MepeHn KpOBaHOro DaBHeHn. PpN oEcbToUBAHN 6Noka nITAHn HAcTPOKn DaTbI IN BpeMeHn Ha np6ope dIy I3MepeHn KpOBaHOro DaBHeHn C6paCbIBaOTcR, OHaKO CoXpaHeHHbIE B NaMaTn pe3yJbTaTbI I3MepeHn He npOnaJaOT.
HactpoJaΦopMaTa OTo6paXeHnBpeMeHn, DaTbI, BpeMeHn n Bluetooth®
B 063aTeNbHOM NopRAnke Heo6xOJMo yCTaHObNTb DaTy n Bpemr. 3TO N03BOJIT npaBnIbHo COxpaHrTb B NaMrtu pe3yJIbTaTbI 3MepeHnC INx DaToN IN BpemeHem, a 3aTeM BblBOJITb INx Ha 3KpaH.
MeHIOIINBbIIOJIHeHnHaCTpoEKMoxHO Bbl3BaTbDByMn cnocO6amN.
- Ipeep nepBbIM NcnoJIb3OBAHnEM n noCne KaKdoJ 3aMeHbI 6aTaapeek
Iocne yctaHOKn 6aTaapeek B npnbop aBtOMaTnueckN OTKpbIBaETcCOOTBeTCTByIOUeMeHIO. - Ecn6aTapeKnyxeyCTaHOBnEhbl
Ha BbIKJIIOeHHOM npI6ope HaXMMTe KhoNka START/ STOPP ① n ydepKnaBaiTe ee B TeueHne 5 cekyHd.
B 3TOM MeHIO Bbl MoKeTe NocJeIOBaTeIbHO BblONHHTb CJIeDyUOHe HAcTpOKn:
Ha nciJIee 3amnraIOT yacbl.
C NOMOUIK HONOK COXpAHEN M1/M2 BbI6epNTe HxH0E KOJIueCTBO YacOB IN NOITBepnTe BbI6Op HaxKaTneM KHONK START/STOP①
Ha nncnnee 3amiraiot MmHyTbI.
C NOMOUIK HONOK COXpAHEN M1/M2 BbI6epNTe HxHcE KOJIueCTBO MNHT IN NOITBepNTe BbI6Op HaxKaTnEM KHONK START/STOP①
Ha dincnee 3amnraet cunboj Bluetooth®.
C NOMOJIbIO KHOJOK COXpAHEnIaM1/M2 BKNIOUHTe (3AMIRAET CMBON Bluetooth®) INN OTKIOUHTe (CMBOBn Bluetooth® He 6yIeT OTObpaKaTbcra Ha DmCPIee) aBTOMATNUeCKyO aKTINBaUNIO NepeHOCa DaHHbIX Yepe3 Bluetooth® I NOITBePdNTe BblOp HaxKaTneM KHOJKN BKJI./BblKI. ①
Bo Bpem nepeaun daHHbix c nomoubbluoteo pacxoyetc3apn aKKymyIaTopa.
7.Ппимеоннe
Osshne npabnna npi camocToTebHom n3MepeHnKpOBaHoro daBHeHH
Дя co3dahnHaH6oJee noka3aTeIbHOrO npoФnIaN3MeHeHnApTePnAIBHOrDabJeHnC CONOCTaBIMMbIMBeINuHaMnpeyIpyHNO 3mepeTApTePnAIBHoe daJIeHne B OndHo nTO JKe Bpem cyTOK.PekOMeHdyETc npOBODITb 3Mepe
#
100
HINI DBAKDbI B DeHb: yTPOM NOCJI NODbEMa C NOCTeIN IBe- Yepom.
- N3mepehne Bcerda doJHKHO npoBOuTbcB COCTOHN Hn 3nueckoro nokor. He npoBOUte n3mepehne B COCTOHN CTpecca.
- Ipeep n3mepeHnem CneIyET BO3depKnBaTbC8 OT npnema NIIuN JxNkOcTN, KpyHnN Hpyn3Nuecknx Harpy3OK B TeueHne He MeHee 30 MNHyT.
- Npeed nepBbIM n3MpeHem KPOBraHOro daBJIeHnBaCerda OtDbixaIte B TeHeHne 5 MNHyT!
- Pn npOBeHm HecKoJIbKxN NocJeIOBaTeJbHbIX CeaHCOB 3MepeHn INTepBaI MeKdy HmN DoJKeH CoCTaBJIr T He MeHee 1 MInHyTbl.
-Пин haJIuHm COMHeHm OTHoNteIbHo nOlyeHHbIX pe3yIb-TaTOB NOBToPte N3MepeHne.
HaKnaJaBbAHHe MaHXKeTbl
- I3MepaTb daBHeHne MOxHO Ha o6eHX pyKax. OnpeJeHbIe OTKIOHeHn MEXdy I3MepeHHbIM apTePnaJbHbIM daBLeHnEM Ha npaBoi n JbeO pyKe obycIOBHebl φN3IOLOrUeCKmN Oc06EHHOCTaMn N a6COJIOTHO HOpMaJbHbl. Heo6XoIMMo BcERda pOBODtB I3MepeHn Ha pyKe c 6oJee BbICOKmN POKa3aTeJIaMn daBHeHn. IpeD tem kak npicTuNaTb K I3-MepeHIO CBOeRO daBHeHn, IpOKOHcyIbTnpuYteCb C Bpa-OM. I3MepaTe daBHeHne BcERda Ha odHOn pyKe.
- Pnp6op MoXHO nCNoJb3OBA Tb TOJbKO C OdHoi N3 CneDyUoIx MaHKeT, KOTOpYIO Heo6XoDnMO BbIbNpaTb B COOTBeTCTBn C OxBaTOM INeya. IpeE N3MepeHem CneDyET npoBepNTb PpAunlbHoCTb NOLOXeHnMaHKeTb C NOMOuB0 OTMeTK, yKa3aHHoH HNXe.
Ccbil. N2 HanmeHObaHne Oxbat pykn
163.764* YHnBepcaJIbHaMaHKeTa 24-40 cm
* BXoNT B CTAHdapTHbI KOMJIeK T NOCTaBKn.
HaonKeMaHKeTy Ha rOlyo pyKy BbIe JIOKTe. KpOBOCha6XeHne pyKn He DOnxHO 6bITb HApUHeo n3-3a CInuKOM y3KO ODeXDbI N.
HaKaJaBaIe MaHKeTy Ha IneHo TaK, YTo6bl ee HIXKn H Kpa paCno- JaIcra Bblwe JOKTeBOrO crna apTepn Ha 2-3 cm. LJaHr donJxH e 6bItb HapabEn B CTOPOHy JaDoHN No ceHTpy.
- PpOpyCTnTe BbICTyHaOnu KoneMaHKeTbI Yepe3 MeTaNInueckyUCKo6y,nepeBepHnTe erO oINH pa3N 3aKpOte MaHKeTy 3aCTeXKoJ-NiNpyKoM.MaHKeTa DOJIxHa npIneratbNIOtHO,HO He CNlbHO DaBnTB: NOH Neu D0JXHbI NOMEuaTbCra DBa NaIb-za.
MaHxeta Meet noDxOpa pa3-Mep,ecnnoCne ee HaoKeHna OTMeTKa yKa3aTeJI HaxoNTcB npedeJax dmaNa3OHa OK.
BCTaBbTe WJNaHr MaHKeTbIB pa3beM Dnra WTeKepaMaHKeTbI.
PpaBnIbHoe nOIOXeHne TeNa
-IMnI3MepeHnKPOBHaHO DaBHeHn KOMΦoPTHO yCraDbTecb C BblpmaHHo HcHnOH.ObopntEcBa cHnHy nNoJoxNte pyky Ha onopy.He KnaDHT HORY Ha HORY, NOCTaBbTe CTyHn Ha noI dpyr prdom C dpYrom.
- 06raTeNbHO cneInte 3a TEM, yTo6bMaHKeTa HaxOuInacb Ha yPobHe cepua.
YTO6bI N36ExKaTb NCKaKeHnpe3yNbTaTOB N3MepeHn, BO BpEmN3MepeHn BeDTe Ce6a CnOKoHn He pa3roBaPnBaIe.
U3mepeHne apTeepaIbHOro daBHeHn
-ДяЗаусякприбopaдяизмерени apTeрmaлььно давлени нахмITE KHOПКУ START/STOP ①Bce зелем entbl dincnlen OTO6paKaIOTcRaHa KOpOTkn npomexkyTOK BpeMHn.
-Пюцeccизмереня abTOMaTnueckn
наниhaeTс рпбл.Чepe3 3ceKHyдbl.
Снayana HabakhyBaetcmaHKeTa.

I3mepeHne MOXHO npeBaTb BJIIO6oe BpemHaKaTnEM KhoNkN START/STOP
- Iocne 3Toro DaBHeHne BO3dyxA B MaHKeTe NoCTeHHO CHNJaETc. 3aTeM NaHHaETc N3MepeHne. Ppi paCNo3HaBaHm NyIbCa NOBJIeTc CnMBoI NyIbCa.
EcHnObHApXKeHaTeHdEHHcH N K NOBbIeHHOMy KPOBbAHOMy DaBHeHIO,TO npN CnYcKe BO3dyxa MaHKeTaMOKeT CHOba HaKaUHbTaBcR Do yPOBbIeHHOroDaBJIeHn. - Ito OKOHuaHn H3MepeHnOCTaBwHcR B03dyX 6bI-CTpo ydaIaETc.
- OTo6pa3aTcpe3yIbTaTbI 3MepeHnCnCTOJIuYeCKoR DaBJIeHn, dnaCTOJIuYeCKoR DaBJIeHn nIyIbCa. HnDnKATop COCTOHN IPOKOr HaN DnCnPHeem 3arOpHTcER3eNeHbIM NII KpaChbIM B 3aBNCUMOCTNOT OUeHNr ReMOdINHAMuYeCKoI CTA6nIb-HOCtN.
E NOBJeTc, ecn H3MepeHne He ydaIOcB BbIOJHHTb npaBnIbHO. B 3tOM Clyuec CM. pa3dE «TO deJaTb npu Bo3- HNKHOBeHn Ipo6IeM?»
Tepe npn nOMOu KNHOK coxpaHeM N M2 BbI6epnte noB3OBaTeNbCKyIO naMyTb. EcNn BbI He Bbl6pAnNoB3OBaTeNbCKyIO naMyTb, To pe3yIbTaT u3-Mepenra 6yET COXPAHE B noB3OBaTeNbCKoN paMaTn nocJeHrnoB3OBaTeJIHa DnCnnee noRBAeTCa COOTBeCTByUOuIN CmBOJ


M3mepenhe
- EcnnnakTbBupoBaHa cyhKcuaa Bluetooth® nocne noDTBepKdEHHa BbIbopa nolb3ObaTeNa C nOMoubIO KhoNkBKL./BblKJ. NaHETcpepa a daHHbIX B npInoxeHne «beurer HealthManager».
- Ha dinncnnee 3amnraet cunbON Bluethoht®. Pnp6bl. B TeueHne 30 cekyHd np6op yctaHabnBaet C8ra3b C npIIOXKeHem.
Kak ToIbKO coeHHeHne 6yTeY yCTaHOHe, CmBOJ Bluetooth® nepeTaHET MuraTb. Bce pe3yNbTaTbI n3MepeHn8 6yDyT aBtOMaTuYeCKn nepeHaHbI B npNJIOKeHne. IocNe ycNeuHoi nepeaun daHHbIX npN6Op aBTOMaTuYeCKn BBIKJIOHTcR.
EcnnB TeueHnE 30 cekyHn He 6ydt YcTaHOBNeHo coeHHHe nC npInIOxKeHnEM, CmBON Bluteoth noTacHet, np6Op IaI N3MepeHn KPOBraHO daJIeHn ABTomATuCeCKN OTKJIIOHTcY epe3 3 MmHyTbI.
Ecnn Bbl 3abydeTe BbIKIOHTb npn6op, OH BbIKIO- qHTcA BtOMaTNUeCKN Upe3 3 MNHytbl. JAnke B 3Tom Cnyae pe3yIbTaT N3MepENH 6yDeT COxpaHeH B Bbl- 6paHHo IIN PocJeHEn NcNoB3OBAHHo NpIb3OBA- TeNbckO NaMRTN.
IpeepaHa n3MepeHHbIX 3NaueHn uee3 Bluetooth®
IOMMIO IOKaJIbHOINHdNkaCmN IN COXpaHeHnI3MepeHHbIX 3NaueHnHa np6ope Bbl moKeTe nepeDaBaTb pe3yNbTaTb I3MepeHnHa cMapToOH npn NOMOu TExHoJorNn Bluteoth low energy.
CnCTeMHbIe TpeBoaHnIy IpnIooKeHnIy « beurer HealthManager»
- iOS ≥ 10.0, Android™ ≥ 5.0
-Bluetooth®≥4.0
Cnncok noDpeKnaeMbix ycTpoiCTB:

IJIa IpeHocA U3MepeHHbIX 3HaueHn BbINOJIHnTe CJIeYIOUne. DeIcTBnI:

War 1:BM77
BknouHrte Bluetooth Ha cboem npnbope, KaK onucaHo B pa3dene «HaCTpOka foPmTa daTbI, BpeMeHn Bluteoth

Uar 2:Пилхенe « beurer HealthManager»
Iobabte BM 77 B npinloxene «beurer HealthManager» B pa3dene «HactpoiKn/Mon yctpoiCTBa» n cneynTe yka3aHnM.

Lar 3:BM77
PpOBeIte I3MepeHne DaBHeHnA.


Uar 4 BM 77
IcToHnK: WHO, 1999 (World Health Organization)
INDukaTop coctoHnno nokor
OHa Hn60Jee pacnpoctpaHeHbIX OuIn6OK
Pn IN3MepeHHN KpOBaHO DaBJeHHN -OTcyTCTBne
y NOb3oBaTeJI DOcTaTOHNO CnOKoHOrO KpOBoo6paUeHHN
Ha MOMeHT IN3MepeHHN. B 3tOM cIyue IN3MepeHHoe
3HaueHne CnCTOJIuYeCKoR N dnaCTOJIuYeCKoR DaBJeHnHe OTo6paKaet KpoBraHoe daBJeHne NOKo, Ho erO BCE JeCJeDyET NCIOJb3ObaTb Iry OceHKn N3MepeHHbIX 3HaueHn.
B daHnom npnbope dIJI n3MpeHnKpOBaHOro daBHeHn IcNoJIb3yETcR BCTpoEHnA DnaIraHocTnKa rEmOduHaMnuecko CTa6uNbHOCTn (HSD), KOTopar n3MepaET rEmOduHaMnueckyo CTa6uNbHOCTb N0JIb3OBAteJIr BO Bpemn 3MpeHnKpOBaHOro DaBHeHn I NO3BOJAE TOnyUHTb INHΦOpMaIIO O TOM, n3MpeHNO JIN KpOBaHOe daBHeHne pPr DoCTaTOUHO CnOKOHOM KpOBoo6paaeHH.
Cd-6atapecoepkntKaMmN,
Hg-6aTapecoepKNT pTyTb.
12. TexHnueckne daHHbIe
Pn6op
MoeJIb N2 BM 77
Bxo100-240B,50-60Γu,0.5A
MaKcImaIbHo
BbIXoD 6 B noct. Toka, 600 mA, Tolbko B KOM6HaCn C npi6opam IJnI3MepeHnApTePnaJIbHOr DaBHeHn Beurer
Zródio: WHO, 1999 (World Health Organization)
Wskacznik spoczynku
The Bluetooth® word mark and logos are registered trademarks owned by Bluetooth SIG, Inc. and any use of such marks by Beurer GmbH is under license. Other trademarks and trade names are those of their respective owners.
Apple and the Apple logo are trademarks of Apple Inc., registered in the U.S. and other countries. App Store is a service mark of Apple Inc., registered in the U.S. and other countries.
Google Play and the Google Play logo are trademarks of Google LLC.
Android is a trademark of Google LLC.

Beurer GmbH · Söflinger Str. 218 · 89077 Ulm, Germany · www.beurer.com
www.beurer-blutdruck.de · www.beurer-gesundheitsratgeber.com · www.beurer-healthguide.com

0483
 ManualGo.com
ManualGo.com 




