SIH 21 - Inhaler SANITAS - Free user manual and instructions
Find the device manual for free SIH 21 SANITAS in PDF.

User questions about SIH 21 SANITAS
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Inhaler in PDF format for free! Find your manual SIH 21 - SANITAS and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. SIH 21 by SANITAS.
USER MANUAL SIH 21 SANITAS
europeanity in the 19th century.
european countries, including the United States, have been involved in the development of new technologies and new products.
European countries are also becoming increasingly active in the development of new technologies and new products.
ELEKTKOMagnetische KOMPATIBILITÄT
Riedlinger StraBe 28,
88524 Uttenweiler, Germany
geltend zu machen.
- Getting to know your instrument 15
- Signs and symbols 15
- Safety information and warnings.. 16
4.Description of device and accessories 17 - Initial use 18
- Operation 19
- Changing the filter 20
- Cleaning and disinfection 20
- Disposal 22
- Troubleshooting 22
- Technical specifications 23
Included in delivery
For details of the delivery scope, see description of the device and accessories on page 17.
- Nebuliser
- Atomiser
- Compressed air hose
- Mouthpiece
Adult mask - Child mask
- Spare filter
These instructions for use
1. Getting to know your instrument
Dear valued customer
Please read these instructions for use carefully and keep them for later use, be sure to make them accessible to other users and observe the information they contain.
Area of application
This nebuliser is a device used for the nebulisation of liquids and liquid medication (aerosols) and for the treatment of the upper and lower respiratory tract.
The nebulisation and inhalation of medication prescribed or recommended by a doctor can prevent against diseases of the respiratory tract, treat the side effects of these disorders and accelerate healing.
For more information on possible applications, consult your doctor or pharmacist.
The device is suitable for inhalation at home. Medicines should only be inhaled when instructed by a doctor. Inhalation should be performed in a calm and relaxed atmosphere. Inhale slowly and deeply to enable the medicine to reach the small bronchi deep into the lungs. Exhale normally.
2. Signs and symbols
The following symbols appear in these instructions.

Warning Warning instruction indicating a risk of injury or damage to health.

Important Safety note indicating possible damage to the unit/accessory.

Note Note on important information.
The following symbols are used on the packaging and on the nameplate of the device and accessories.
| Type BF applied part | |
| Read the instructions for use | |
| Device in protection class 2 | |
| Manufacturer | |
| I | On |
| O | Off |
| SN | Serial number |
| 30 ON/30 OFF | 30 minute operation, followed by a 30 minute break before operation continues. |
| Keep dry. |
3. Safety information and warnings

Warning
- Before use, ensure that there is no visible damage to the unit or accessories. When in doubt, do not use the unit and contact your dealer or the customer service address provided.
- In the event of device faults, please see Chapter "10. Troubleshooting".
- Using the unit is not a substitute for consultation with or treatment from a physician. Whenever you have any pain or illness, always contact your physician first.
If you have health concerns of any kind, contact your general practitioner! - When using the atomiser, always follow the basic hygiene procedures.
- Always follow your doctor's instructions regarding the type of medicine to be used and the dose, frequency, and duration of inhalation.
- Only use medicines that have been prescribed or recommended by your doctor or pharmacist.
Please note:
For treatment, only use parts indicated by your doctor according to the particular diagnosis.
- Check whether there are contraindications for use with the usual systems for aerosol therapy on the medication instruction leaflet.
- If the unit does not function correctly or if you begin to feel unwell or feel pain, stop using the unit immediately.
- During use, hold the unit away from the eyes, as some nebulised medicines may cause damage to the eyes.
- Do not operate the unit in the presence of flammable gases.
- This device is not intended to be used by children or persons with restricted physical, sensory (e.g. insensitivity to pain) or mental abilities or persons lacking in the required experience or knowledge for safe operation of the device, unless supervised or instructed by a person responsible for their safety.
- The device must be switched off and the plug pulled out before every cleaning and/or maintenance procedure.
- Keep children away from packaging materials (risk of suffocation).
- To avoid the risk of entanglement and strangulation, store cables and air lines out of the reach of small children.
- Do not use attachments that are not recommended by the manufacturer.
- Connect the unit only to the mains voltage listed on the type plate.
- Do not dip the unit into water and do not use it in wet rooms. Do not allow any liquid to penetrate the unit.
- Protect the unit from strong impacts.
- Never touch the mains cable with wet hands, this can cause an electric shock.
- Do not pull the mains plug from the socket by the cord.
- Do not pinch or bend the mains cable, pull it over sharp objects, or allow it to hang freely, and protect it against sources of heat.
- We recommend that the power cable is completely unrolled to avoid dangerous overheating.
- If the mains cable or housing is damaged, contact our customer service department or your local dealer.
- Opening the unit carries a risk of electric shock. Disconnection from the mains supply is only guaranteed if the mains plug has been removed from the socket.
- Do not use the unit if it has fallen or been dropped on the floor, been exposed to extreme humidity or is otherwise damaged. In case of doubt, contact our customer service department or your local dealer.
- The SIH 21 nebuliser must only be used with suitable atomisers and with the appropriate accessories. The use of other atomisers and accessories can impair therapeutic efficiency and may damage the device.

Important
- Power cuts, sudden faults or other unfavourable conditions may render the unit unusable. You are therefore recommended to keep a spare device or replacement medicine (as agreed with the doctor).
- If adapters or extensions are required, these must comply with the applicable safety regulations. The electrical capacity and the maximum capacity specified on the adapter must not be exceeded.
- The unit and the mains cable must be stored away from sources of heat.
- Do not use the unit in a room in which a spray has previously been used. If this is the case, ventilate the room before commencing treatment.
- Do not allow any objects to enter or obstruct the ventilation openings.
- Do not use the device if you notice an unusual noise.
- For hygiene reasons, each user must use his own accessories.
-
Always disconnect from the mains after use.
-
Store the device in a location protected against environmental influences. The device must be stored in the specified ambient conditions.
FUSE
- The device contains an overload protection fuse. This must only be changed by authorised specialist personnel.
General notes

Important
- Use the unit only:
-On humans,
- for its intended purpose (aerosol inhalation) and in the manner described in these instructions for use.
- Any improper use can be dangerous!
- In acute emergencies, first aid takes priority.
- Apart from the medication, use only distilled water or a saline solution in the unit. Other liquids may cause defects to the nebuliser or atomiser.
- This unit is not intended for commercial or clinical use, but only for individual, private household use!
Before using the unit for the first time

Important
- Before you use the unit for the first time, remove all packaging materials.
- Protect the unit against dust, dirt and moisture. Never cover the unit during operation.
- Do not use the unit in very dusty environments.
- Switch the unit off immediately if it is defective or malfunctioning.
- The manufacturer shall not be held liable for damage or injuries caused by improper or incorrect use.
Repairs

Note
- Never open or attempt to repair the unit yourself, as otherwise proper function is no longer guaranteed. Failure to observe this regulation shall void the warranty.
- If you need to have the unit repaired, contact customer service or an authorised dealer.
4. Description of device and accessories
Overview of the nebuliser
1 Mains cable
2 Lid
3 On/off switch
4 Compartment for accessories
5 Filter cap with filter
6 Hose connection
7 Holder for atomiser

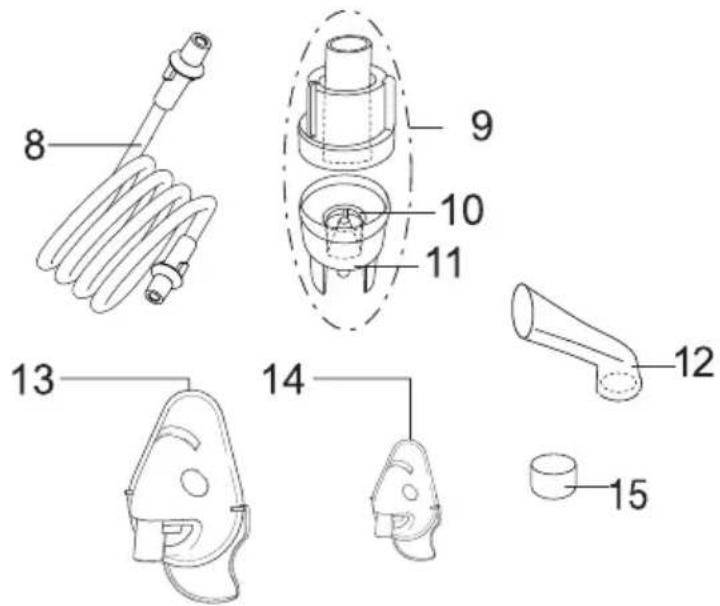
Overview of atomiser and accessories
8 Compressed air hose
9 Atomiser
10 Nozzle attachment
11 Medicine container
12 Mouthpiece
13 Adult mask
14 Child mask
15 Replacement filter
5. Initial use
Setup
Remove the unit from the packaging. Place it on a flat surface.
Ensure that the ventilation slits are free.
Fold open the lid [2] to access the accessories.
Before using the unit for the first time

Note
- The atomiser and accessories must be cleaned and disinfected before they are used for the first time. For information about this, see "Cleaning and disinfection" on page 20.
- Attach the compressed-air hose [8] at the bottom of the medicine container [11].
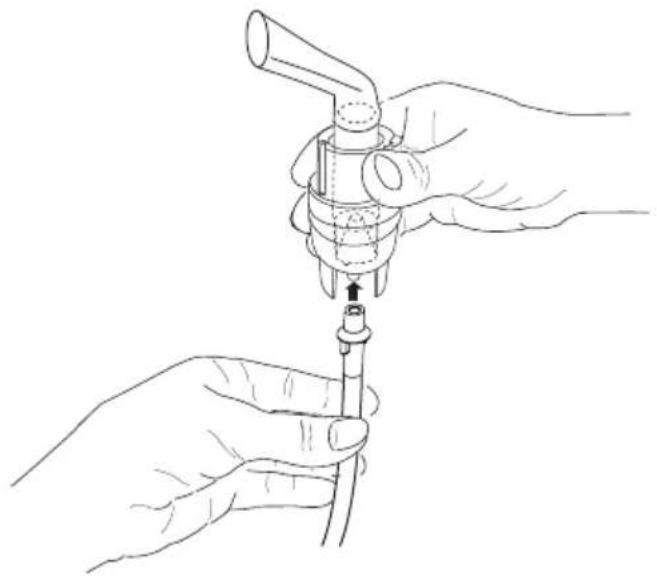
- Connect the other end of the hose [8] to the hose connector [6] on the nebuliser by lightly turning it.
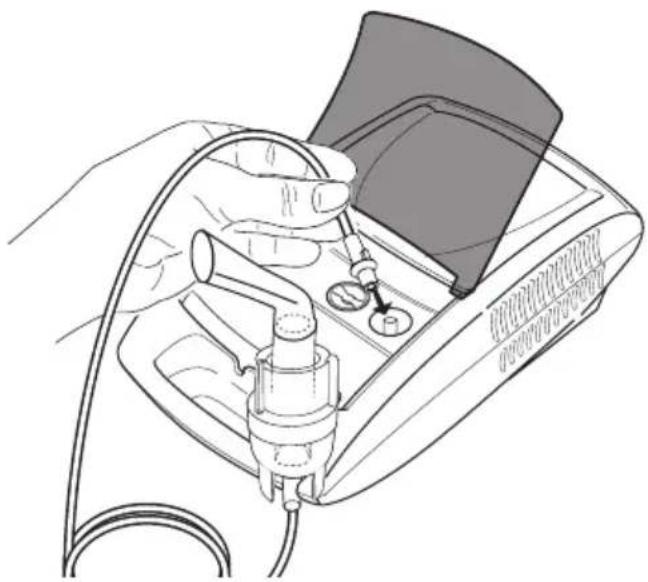
Mains connection
Connect the unit only to the mains voltage listed on the type plate.
- Insert the mains cable plug [1] into a suitable socket.
- Push the plug completely into the mains socket.
Note
- Ensure that there is a socket near to where the unit will stand.
- Lay the mains cable in such a way that no one can trip over it.
- To disconnect the nebuliser from the mains after use, first switch off the device and then remove the plug from the socket.
6. Operation

Caution
- For hygiene reasons, the atomiser [9] and the accessories must be cleaned after each treatment and disinfected after the last treatment of the day.
- The accessories may be used only by one person; use by several people is not recommended.
- If several different medicines are to be inhaled in succession during therapeutic use, the atomiser [9] must be rinsed under warm tap water after each use. For information about this, see "Cleaning and disinfection" on page 20.
- Follow the instructions for changing the filter contained in these instructions.
- Before each use, check that the hose connections on the nebuliser [6] and the atomiser [9] are tight.
- Before use, check that the device is functioning correctly. To do this, switch on the nebuliser (including the connected atomiser, but without any medication) for a short time. If the atomiser [9] expels air, the device is functioning correctly.
1. Inserting the nozzle attachment
- To open the atomiser [9], twist the upper section anti-clockwise against the medicine container [11]. Place the nozzle attachment [10] in the medicine container [11].
- Ensure that the cone for administering medication fits well on the cone for the air duct inside the nebuliser.
2. Filling the atomiser
- Pour an isotonic saline solution or the medicine directly into the medicine container [11]. Do not overfill! The maximum recommended fill level is 8ml
- Use medication only on the advice of a physician, and check the appropriate inhalation duration and quantity for your needs.
- If the required volume of medication is less than 2ml , top up this quantity to at least 4ml using an isotonic saline solution. Viscous medicines may also require dilution. Always follow the instructions of your doctor.
3. Closing the atomiser
- To close the atomiser [9], twist the upper section clockwise against the medicine container [11]. Ensure the parts are connected correctly.
4. Connecting the accessories to the atomiser
- Connect the required accessory part (mouthpiece [12], adult mask [13] or child mask [14]) to the atomiser [9].

Note
The most effective form of nebulisation is by using the mouthpiece. Nebulisation using a mask is only recommended if it is not possible to use a mouthpiece (e.g. for children who are not yet able to inhale medication using a mouthpiece).
- Before commencing treatment, pull the atomiser upwards out of its holder [7].
- Start the nebuliser using the on/off switch [3].
- A flow of spray from the atomiser indicates that the device is functioning properly.
5. Treatment
- When inhaling, sit upright and relaxed at a table and not in an armchair to avoid compressing the airways and therefore impairing the effectiveness of the treatment.
- Inhale the nebulised medicine deeply.

Important
The device is not suitable for continuous operation; after 30 minutes of operation it must be switched off for 30 minutes.

Note
During treatment, hold the atomiser straight (vertical), otherwise the atomisation does not work and correct function cannot be guaranteed.

Important
Essential oils, cough medicines, solutions designed for gargling, and drops for application to the skin or for use in steam baths are wholly unsuitable for inhalation using a nebuliser. These substances are often highly viscous and can impair the correct function of the device and hence affect the effectiveness of its application in the long term.
In the case of oversensitivity of the bronchial system, medicines containing essential oils can sometimes cause acute bronchiaspasm (a sudden, cramp-like tightening of the bronchi accompanied by breathlessness). Always ask your doctor or pharmacist for advice!
6. End of inhalation
If the spray is only output sporadically or if the noise changes during inhalation, you can end the treatment.
- Switch off the nebuliser after treatment using the on/off switch [3] and disconnect it from the mains.
After use, replace the atomiser [9] in its holder [7].
7. Changing the filter
In normal operating conditions, the air filter must be replaced after approx. 500 operating hours or one year. Please check the air filter regularly (after 10-12 nebulisation procedures).
If the filter is very contaminated or blocked, replace the used filter. If the filter has become wet, it must also be replaced with a new one.

Caution
- Do not try to clean and re-use the used filter!
- Only use original filters supplied by the manufacturer. Otherwise, your nebuliser may become damaged or effective therapy can no longer be guaranteed.
- Do not repair or maintain the air filter while it is in use.
- Never operate the device without a filter.
7. Clean the device
For information about this, see "Cleaning and disinfection" on page 20.
To replace the filter, proceed as follows:

Caution
- First switch off the unit and unplug it.
-
Allow the unit to cool down.
-
Remove filter cap [5] upwards.


Note
If the filter remains in the unit when the cap is removed, remove the filter from the unit using tweezers or similar.
- Replace the filter cap [5] containing the new filter.
- Check that the filter cap is firmly in place.
8. Cleaning and disinfection
Atomiser and accessories

Warning
Observe the following hygiene instructions in order to avoid any health risks.
- The atomiser [9] and accessories are intended for multiple use. Please note that cleaning requirements differ according to the different application areas.
Notes:
- The atomiser and accessories must not be mechanically cleaned with brushes or similar as this can cause irreparable damage and the success of the treatment can no longer be guaranteed.
- For additional requirements regarding the necessary hygienic preparations (hand washing, handling of medicines or inhalation solutions) in high-risk groups (e.g. cystic fibrosis patients), contact your doctor.
- Ensure that all parts are properly dried following all cleaning and disinfection. Residual moisture or water droplets can result in an increased risk of bacterial growth.
Preparation
-
Directly following each treatment, all parts of the atomiser [9] and any accessories used must be cleaned of medicine residues and contamination.
-
To do this, dismantle the atomiser [9] into its component parts.
- Remove the mouthpiece [12] from the atomiser.
- Dismantle the atomiser by twisting the upper section anti-clockwise against the medicine container [11].
- Remove the nozzle attachment [10] from the medicine container [11].
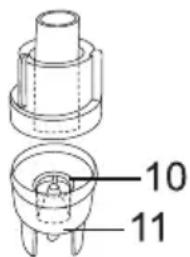
- The nebuliser is later reassembled in the reverse order.
Cleaning

Important
Before you clean the unit, always switch it off, unplug it and let it cool off.
The atomiser and the used accessories such as the mouthpiece, mask, nozzle attachment, etc. must be washed with hot but not boiling water after each use. Carefully dry all components using a soft cloth. Once all components are fully dry, reassemble the atomiser and place the components in a dry, sealed container or carry out disinfection.
When cleaning, ensure that all residues are removed. Never use any substances that may be poisonous when in contact with the skin or mucous membranes, or when swallowed or inhaled.
Use a soft, dry cloth and non-abrasive cleaning products to clean the device.
Do not use abrasive detergents and never immerse the unit in water.

Important
- Ensure that no water penetrates inside the unit!
- Do not put the device or accessories in a dishwasher.
- Do not touch the unit with wet hands while it is plugged in; do not allow any water to be sprayed onto the unit. The unit must be operated only when it is completely dry.
- Do not spray any liquids into the ventilation slots. Any liquid that penetrates into the unit can damage the electrical parts or other components of the nebuliser and impair the function of the device.
Condensation, care of the hose
Depending on the environmental conditions, condensation may collect in the hose. To prevent against bacterial growth and to ensure effective therapeutic use, this moisture must always be removed. To remove moisture from the hose, proceed as follows:
- Remove the hose [8] from the atomiser [9].
- Leave the hose plugged into the nebuliser [6].
- Run the nebuliser until all the moisture has been displaced by the flow of air.
- In the event of significant contamination, replace the hose.
Disinfection
Follow the steps listed below carefully in order to disinfect your atomiser and the accessories. It is advisable to disinfect the individual components after the last time they are used each day at the latest.
(For disinfection, you will simply require a small amount of clear vinegar and distilled water).
- First clean the atomiser and the accessories as described under "Cleaning".
- Place the disassembled nebuliser and mouthpiece in boiling water for five minutes.
- For the remaining accessories, use a vinegar solution consisting of 14 vinegar and 34 distilled water. Make
sure that the volume is sufficient to fully submerge the parts in the solution.
- Leave the components in the vinegar solution for 30 minutes.
- Rinse the parts with water and dry carefully with a soft cloth.

Important
Do not boil or autoclave the masks and air hose.
- Once all components are fully dry, reassemble the atomiser and place the components in a dry, sealed container.

Note
Ensure that all parts are thoroughly dried during, and in particular after cleaning, otherwise there is an increased risk of bacterial growth.
Sterilisation
Use the cold sterilisation solutions according to the manufacturer's instructions.
Drying
- Place the individual components on a dry, clean and absorbent surface and allow to dry completely (for at least 4 hours).
Material resistance
- As with all plastic components, frequent use and cleaning of the atomiser and the accessories can lead to a certain amount of wear. Over time, this can alter the aerosol properties and may eventually affect the efficiency of therapy. We therefore recommend that you replace the atomiser once a year.
- When choosing a cleaning or disinfecting agent, note the following: Only use mild cleaning or disinfectant agents in the quantities recommended by the manufacturer.
Storage
- Do not store the nebuliser in a damp atmosphere (e.g. in the bathroom) or transport it together with damp objects.
- Store and transport the nebuliser away from direct sunlight.
- The accessories can be safely stored in the accessories compartment [4]. Store the device in a dry place, if possible in its original packaging.
9. Disposal
In the interest of environmental protection, do not dispose of the device in household waste.
Please dispose of the device in accordance with EC Directive - WEEE (Waste Electrical and Electronic Equipment).

If you have any queries, please contact the appropriate local authorities.
10. Troubleshooting
| Problem/Question Possible Cause/Remedy | |
| The atomiser produces little or no aerosol. | 1. Too much or too little medicine in the atomiser. Minimum: 2 ml, Maximum: 8 ml. |
| 2. Check that the nozzles are not blocked If necessary, clean the nozzle (e.g. by rinsing). Use the atomiser again. CAUTION: Only poke implements through the fine holes from the underside of the nozzle. | |
| 3. The atomiser is not held in an upright position. | |
| 4. The medicine solution is unsuitable for nebulising (e.g. too thick). The medicine solution should be specified by the doctor. | |
| Output is too low. Hose kinked, filter blocked, too much inhalation solution. | |
| Which medicines are suitable for inhalation? | Consult your doctor. Essentially all medicines that are suitable and approved for device-based inhalation can be inhaled. |
| Some inhalation solution remains in the atomiser. | This is normal and occurs for technical reasons. Stop inhalation if you hear a notable difference in the noise of the atomiser. |
| What special steps should be taken for babies and children? | 1. In babies, the mask should cover the mouth and nose to guarantee effective inhalation.2. In children, the mask should also cover the mouth and nose. Nebulisation next to a sleeping person is not suitable because insufficient medicine reaches the lungs.Note: Children should only use the device with help and under supervision of an adult. Never leave a child alone with the nebuliser. |
| Inhalation with the mask takes longer. | This is for technical reasons. Less medicine is inhaled per breath through the mask holes than using the mouthpiece. The aerosol is mixed with ambient air through the holes in the mask. |
| Why do I need to replace the atomiser at regular intervals? | There are two reasons for this:1. To guarantee a therapeutically effective particle spectrum, the nozzle holes should not exceed a certain diameter. Due to the thermal and mechanical stresses, the plastic is subjected to a certain amount of wear. The nozzle attachment [10] is particularly susceptible to this wear. This can also cause changes to the droplet composition of the aerosol, which has a direct effect on the effectiveness of the treatment.2. You are also recommended to replace the atomiser on a regular basis for hygiene reasons. |
| Does each person need their own atomiser? | This is absolutely necessary for hygiene reasons. |
11. Technical specifications
| Model SIH 21 | |
| Type SIH 21/2 | |
| Dimensions (WxHxD) | 300 x 180 x 100 mm |
| Weight 1.65 kg | |
| Working pressure | Approx. 0.8 to 1.45 bar |
| Atomiser filling volume | Max. 8 ml Min. 2 ml |
| Medication flow rate | Approx. 0.4 ml/min. |
| Sound pressure | max. 52 dBA (acc. to DIN EN 13544-1 section 26) |
| Mains connection | 230 V~; 50 Hz; 230 VA UK: 240 V~; 50 Hz; 240 VA |
| Operating conditions | Temperature: +10°C~+40°C Relative humidity: 10%–95% |
| Storage and transport conditions | Temperature: 0°C~+60°C Relative humidity: 10%–95% Atmospheric pressure: 500–1060 hPa |
| Aerosol Properties | Flow: 5.31 l/min Aerosol delivery: 0.171 ml Aerosol delivery rate: 0.082 ml/min Particle size (MMAD): 3.16 μm |
Subject to technical modifications.
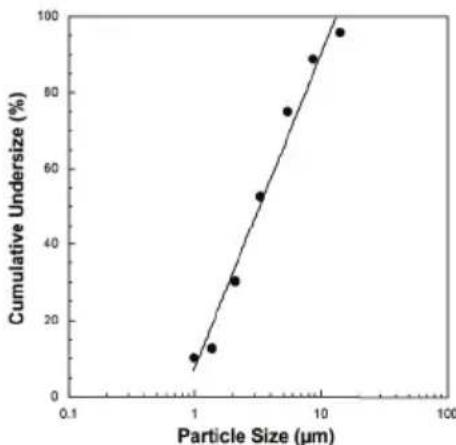
Measurements were performed using a sodium fluoride solution with a "Next Generation Impactor" (NGI). All measurements were obtained with a sodium chloride solution using a laser diffraction method. This diagram may not be applicable for suspensions or highly viscous medicines. More information can be obtained from the relevant medicine manufacturer.
Spare parts
| SIH 21 year pack Mouthpiece, adult/children's mask, atomiser, compressed air hose, filters | REF 601.20 |

Note
If the unit is used outside of the specifications, proper function is no longer guaranteed!
We reserve the right to make technical changes to improve and further develop the product.
This device and its accessories comply with European standards EN60601-1 and EN60601-1-2, as well as EN13544-1, and is subject to special safety measures in terms of electromagnetic tolerance. Note that portable and mobile RF communication equipment can affect this unit. For detailed information, contact customer service at the address provided. The unit conforms to the requirements of the European Directive for Medical Products 93/42/EC, the MPG (German Medical units Act).
ELECTROMAGNETIC COMPATIBILITY
- The device complies with current specifications with regard to electromagnetic compatibility and is suitable for use in all premises, including those designated for private residential purposes. The radio frequency emissions of the device are extremely low and in all probability do not cause any interference with other devices in the proximity.
- It is recommended that you do not place the device on top of or close to other devices. Should you notice any interference with other electrical devices, move the unit or connect it to a different socket.
- Radio equipment may affect the operation of this device.
FRANÇAIS
Sommaire
Chere cliente, cher client,
IJIeHnIcNoB3OBaTbTOJbKO Te DeTaII, PnIMeHHeKOTOpbIX 6blIO peKOMeHDoBaHO BpaOMB COOTBeTCTBnC COCToHEm 3IDOpOBy.
BHHMaTeIbHO npouHaiTe Ha npinaraIOUeMcK JekapCTBeHHOMy npenapaTy ynaKOBOuHOM JnCTKe INHΦOpMaIIO O BO3MOXHbIX npOTnBOJOKa3AHnIX K npIMeHeHIO C O6bIHbIMN CnCTeMaMn a3pO3OJIbHOI TepaIIIN.
- Ecni np6op pa6oTaET HeKOppeKTHO nIy BAc NOBnncb HeDOMOraHne IJN 60JI, cpa3y Je npEeKpATnte NcNoJIb3OBaHne np6Opa.
- Bo Bpemnncnoj3oBaHnne depKnte npnbop Ha doCTaTOCHOM pacCTOAHN nT rna3, paCnblneHHbI INekapCTBeHHbI npenapaT MoKeT npNBecTu Knx NOBpeXDeHNrM.
He nCnoB3yIe npi6op B nOmeueHnX C roPouH Mra3am.
JaHHbI np6Op He npedHa3HaueH Ia nCNoIb3OBaHnJeTbMn nn JIOdbMn C OraPaHnueHHbIMn 0u3NHeCKmN, CeHCOPHBIMn (HaNPmep, COTcyTCTBnEM 6oJIeBOO YyBCTBNTeJIbHOCTn) nn YMCTBeHHbIMN CNOcO6HOCTaMn, C HeOCTaTOUHbIMn 3HaHnA Mn nn ONbITOM. NckIIOUeHne COCTaBnIOT cnyan, KOrda 3a HMM OcyueCTBnEETc HndJexaunn Hnd3Op nn
ecnOnn noJynn ot Bac nHcTpyKu nn no nCnoB-3OBaHHIO np6opa.
-Перед Вьлолненем Лобьх pa60Т NO OЧNTKE ИИТExнчeckomу obcnyквани Heo6xOДМо BьIKIHOuHTb пибор ИЗ розтkn.
He daBaIte ynaKOBOHybIM MaTePnA1 DeTAM (onac-HoCTb ydyuWeHn).
Ka6eIb I BO3dYxOBOdbI CneDyET XpaHHTb BHe DoCraEMocTn DeTeMlaJUero BO3pacta Bo N36exKaHne 3anyTbIBaHnI ydyuHnI.
He nCnoB3yIe DoOnHnTeIbHbIe DeTaII, He peKOMeHDoBaHHbIe npOn3BoDInTeJIem.
- Pp6op pa3peSeHNO NOKJIIOuHaTb TOnbKO K CeTeBOMy HApJxKeHHO, yKa3aHHOMy Ha 3aBOdCKoT a6nUKe.
He norpykaite npnbop B Body n He nCnoB3yIte ero BO Blaxhblx nomeueHnx. He donyckaite no- naaHnJxNkOCTn BHTpb npnbopa.
He donyckaTe cnJIbHbIX yIapOB npIbopa.
- He 6epnte Kaebb MOKpbIM pykam - onachocTb nopaxehn3JIeKtpnuecknM TOKOM.
He BbITacknBaIte WTeKep n3 po3eTKn, depXacb 3a ceTeBOi Ka6JIb.
He 3axmuTe, He neperu6aute Ka6eIb, n36eraTe erO COpNIKOCHOBEHnC OCTpbIM KpaAMn PpeMetOB, CBNCAHN IN BO3DeiCTBnH Na Hero BbICOKoTemnpaTypbl.
- PekomeHnyetcnoJIHoCTbIO pa3MaTbIBaTb NITaIOuIznn Ka6eB dIy npedOTbpaueHn nepepeBa.
B Cnyae NOBpeKdEHNc CeTeBOrO Ka6eN i KOpny ca np6opopob6paTnTeCb B cepBnCHyO cnyx6y nn K ToproBOMy npedCTaBnTeIIO.
-ПиВСКьБИТиnpИбopaВОЗнИкайтОпаСHOCТьпорженяэл ektpчeCKIM TOKOM.OTcoeДИнEHNEOTcETN BbINOJIHeHO TOЛБКOB TOM CЛучae,ecIи UTKeKepBbIKJIOUeH IN3 pO3eTKI.
- Ppi napeHn npnbopa, cnIbHom BO3deNCTBnHa Hero BnaI INn INhIX NOBpeXDeHNx erO daJIbHei- Ie ee nCnoJIb3OBAHne 3anpeuaetc. Ppi hAnuHn COMHeHn O6paTntecb B cepBnCHyO cnyK6y INn K TOpROBOMy npedctaBNTeIIO.
- INHraJIaTOp SIH 21 pa3peWaeTcNcNoIb3OBAbToJIbKO BMeCTe C NOxOJaUIM PaCnblNTeJIeM N C COOTBeTCTByUOUMN PpINHaJNeXKHOCTM. PpIMeHHeNIE PpINHaJNeXHOCTeI CTOPOHHNX PpON3BOJNTeJIe MOKeT PpINBeCTN K CHNXeHIO 3ΦΦeKTNBHOCTNJeueHIn I NOBpeJDeHIO pnp6opa.

BHMaHne
C60BIOaue 3JIeKtpo3HepnH,HeOxuaHHO B03- HNKUne HeNCpapBHOCTN IN Dpyrue He6laornpraT- HbIe ycIOBnMOry T npVBecTu K BbIXOdy pnp6opa
n3 cTPO. IOnToMy peKOMeHdyETc NMeTb 3aNaCHO np6Op nIIN leKapCTBeHHbI npenapaT (corlaocBaHHbI C BpaOm).
- Pn Heo6xOIMOCTN nCNoB3OBaHnA aanTepeOB nIyDInHITeJe OHN DOJXHbI OTBeaTb DeNCTByUOImPpeINscaHm TexHKn 6e3onacHOCTn. HeJIb3r npeBbIaTb PpeJeBHyIO MOuHocTb TOKa I MaKcMmaB-HyIO MOuHocTb, yKa3aHHyIO Ha aanTepe.
- Pπi6op HeIb3x XpaHHTb B6JIn3n INCTOCHNKOB TENJa.
He noJIb3yIeTcB npI6OpOM B nOmeUeHnX, B KOToPbIX nepeD 3Tm IM nCNoJIb3OBAInCb aApOzOn. TaKne NOMEseHnA Heo6xOdImo npOBETpNBaTb nepeD npoBeDeHnEM TepaNN. - I36eaiTe nonaHnM MeKnx DeTaeB B OxJaKaIoune OTBepCTn.
He nCnoB3yIte np6op,ecn npn 3TOM NOBRAETc HEO6bHbI Wym.
Buejxco6nOeHncaHtapHO-rnneHnuecknx Tpe6oBaHnKaXdbI NOb3OBaTeJIb DOJKeH IMEt bCBO INHmIBuDyAalbHbe npHaNLeXHOCTn. - Послк кадую Испьзовая Выкlioоча Te npибор n3 po3etkn.
XpaHnTe npnbop B MeCTax, 3aunuEHHbIX OT BO3-DeiCTBnI NOrOndbIX ycIOBnI. Heo6xoJMo oBeCneHTb COOTBeTCTByIOUne ycIOBnI OKpykaIOUeI cpebl IxpaHeHnI npnbopa.
PPEOXPAHNTIEL
B npio6ope yctaHOJIeH MaKcIMaJIbHbI npedeOxpaHntJIb. Ero 3aMeHa DOJXHa IPOIN3BOINTBcT OJIbKO KBaJIuΦnUpOBaHHbIM NepCOHaIOM CO CneuaJIbHbIM DOnyCKOM.
06zne yka3aHna

BhimaHne
- IcnoIb3yIe npu6op nckIouHTeJIbHO B cneyuOux
-ДЯлЕЧENIAIIODEI;
B ueJx, IJI KOTOpbIX OH 6bl pa3pa6oTaH (a-p03OJbHaI INrAIAJRA), B COOTBeTCTBm C yKa3a-HnAMN, PpNBedeHHbIMN B DaHHoINHCTpyKcM N O pPIMHeHIO.
- JIIO6oe HeHaJIeXaUee IcNoJIb3OBAHne MoKeT mMeTb onaChIbe NocJeDCTBnI!
B KpnTnuecknx CnTyauqnx, npexKe Bcero, cJeNyET OKa3aTb NepByu NOMOuB NOCTpaDaBWeMy.
-ПOMIMOЛeKapCTBeHHbIX npenapaTOB pa3peWaeTcA
NcnoJIb3OBaTb TOJIbKO dIcTnJIInPOBaHHyO BODY nIN
ФИЗNOLOrHecKn pactBOp. ПрIMeHHeNte DpyRnx
ЖИДКОCTe Пп ONpeDEJIeHHbIX yCNoBnX MOKeT
ПиВecTи K NOBpeJdeHnIO INHaJIaTOpa INI pAcNbI
JInteJIa.
- Pnp6op He npedHa3HaueH nIe IcNoIb3OBAHnHa Na npOn3BOdCTBe NII B KInHnuecknx ycNoBnX. Ero
PpIMeHHe pa3peSeHo NCKJIuOHTeJIbHO B DOMaHnX yCNoBnIx!
IpeB BBODOM B 3Kcnnyatauio

BHMaHne
- Pered nCnoB3OBAHnEM npi6opa cIeNyET ydaNTb BCE yNaKOBOUHbIe MaTePnaJIbI.
- I36eraTe 3aRpa3HeHna np6opa, nonaHaHa Hrero nI IN BlaHn. Bo BpeM nCnoB3ObaHn Hn B KOem clyae He HaKpbBaTe np6op.
He nCnoB3yIe np6Op B cnIbHO 3aIbJIeHHbIX NOMeUeHnX.
Bcnyae o6hapxehnna depekTOB nHn HenoJaoK B pa6ote HeMeIeHNO OTKIOUHTe np6Op. - IV3ROTOBNTeIb He HecET OTBeTCTBeHHOCTn 3a yUeP6, Bbl3BaHHbIi HEKBaIIuΦuIpOBaHHbIM IIN HeHaIeJkaIIM NcNoJIb3OBAHHeM npIbopa.
Pemont npn6opa

Yka3aHne
- Hn B KOem clyuae He OTKpbIbaiTe npi6op n He peMoHTpyiTe erO camoCToTeJIbHO, TAK KaK B DaHHOM clyuae HaedXHocTb erO pa60tbi 6Oblwe He rapaHTpyetc. HecobliJeHne 3TOro Tpe6OBaHnB BeDet K Notepe rapaHTn.
- EcIn npin6op HuxkaetcB peMOHTe, o6paTntecb B cepBnCHyIO cnyk6y NIN K aBTopn3OBAHHOMy ToproBOMy npedCTaBNTeIIO.
YTo6bI DoCTaTb npHaJnEJXHOCTH, OTKINHbTe KpbIiKy [2].
Peped npBbIM npMHeHHem
iYka3aHne
- Ipeed nepBbIM npimHeHem CneJeT OunchTtB n npOe3nHΦnUPOBaT bacblntelb npHaIaJIexKHOCTN K Hemy. CM. pa3dEe «OuNCtKa n De3nHΦekunra» Ha cTp. 54.
BCTaBbTe BO3dYxOBoHny Tpy6ky [8] B eMKoCTb dIJIeKapCTBeHHbIX npenapaTOB [11].

- JERKIM NOBOPOTOM BCTaBbTe pyroK KOHeu Tpy6KN [8]ВшTu cep [6]ИнгЯТopa.

IopKnIOUeHne K cETn
IopKnIOuAte np6Op TOnbKO K CeTeBOMy HAnpJKeHnIO, yKa3aHHOMy Ha 3aBODcKo Ta6nUKe.
BCTaBbTe wTekep ceTeBOrO Ka6eIa [1] B noDxOJa- uIy o po3eTky.
-Дяп подкюченя К сети StTeKeРdoлжeh 6ытб
нлноctьв BCTabNeHВpo3etky.
iYka3aHne
- Y6eIITecb, yTO pAOM C MecTOM yCTaHOBKn HAXO-DNTcR po3eTKa.
Pa3mecnte Ka6enb TaKIM 06pa3OM, YTO6bl O HeRo HEBO3MOXHO 6blIO CNOTKHytbcra.
-ДяOTKIOUeHnI INrAJIaTOpa OT cETn Iocne INrAJIaCm CHaJana BbIKIOuHTe Cam npu6Op, 3aTeM N3BNEKIne WTeKeP n3 po3eTKN.
6.ОбслужиBaнne

BhimaHne
Buejxco6hOeHncaHtapHo-rHnEHNuecknx Tpe6oBaHn paCbIInTeJb [9] n erO npHaJIeXHOCTN CJeDyET OuHuaTb NocJe KaKDoRIO NCNOB3OBAHn I npOBOnTb erO De3INHpEkuIO B KOHe DNr.
- PpHaIeXHocT MOryT 6bITb NCNoB3OBAHbI TOJIbKO OAnHM JIncOM. NcNoJb3ObaHne HeCKoJIbKIMN JInuamn He peKOMeHdyetcra.
- EcnnI Jn IeueHna Heo6xOIMo NocJeIOBaTeJIbHO npOBODITb INrAJIaCIO HeCKOJIbKIMN BnIaMn Je-KapCTBeHHbIX npenapaTOB, TO INrAJIaTOp [9] HyXHo
POMbIbTaT TeNIOB BODOpBOHOB OBOH NOcJIe KaXdOro npImeHeHn. Cm. pa3JeI «OuNCTKa n De-3nHΦekUra» Ha cTp. 54.
BHNMaTeIbHo cIeNyIte yKa3aHnM daHHo nHCTpyKcIn no 3aMeHe fnlbTpa!
- Перед Кадын Исторьанем письа ровецки Сочь COEDINHEN Tpy6kn C INHRAJIToPOM [6] и пасьлелем [9].
- Ipeed nCnoB3OBAHnem npOBepbTe npaBnBHOCTb pa6oTbI np6opa.ДЯ 3tOrO HeHaJIoR BOKJIIOHTe INHraTOp (BMecTe c pacBblntelem, Ho 6e3 Jekap
CTBeHHoro npenapata). Ecnn npn 3ToM n3 paCnblIInTeIa [9] BbIXoNDT Bo3dyx, To npnbop pa60taeT.
1. YctaHOBka pacnbJIInTeJbHOH HacaKn
- OTKpoIte pacnbIInTeIb [9], NOBepHyB BepXHIOU qactb KOpnyca npOTNB YacOBOu CTrpeIKN OTHCINTeJbHO EMKoCTn DnJI NekapCTBeHHoro IpenapaTa [11]. BcTabbTe pacnbIInTeIbHyIO HacaIKy [10] B EMKOCTb DnJI NekapCTBeHHoro IpenapaTa [11].
- Y6eIITecb, TTO KOHyc IINB BBOJa JIeKapCTBeHHORO npenapata XopoIo 3aKpeIIne H KaOHyce IINoIaDu BO3dyxa B paCnblNTeIe.
2. HanonHeHne pacbIInTeJra
3aJeTe n3OTOnHueckn 1n3NOJIOrueckn pactBop nIJIneKapCTBeHHbI npena- paT HENOCpeDCTBeHHo B emKocTb IJIeKapCTBa [11]. He DONYCKaITE nepenONHeHnE mKocTn! MaKcImaJIbHbI peKOMeHdyEmbl O6bem 3a- noJIHeHncoCTabJIeT 8 mJI!
- IcnoIb3yIteJIekapCTBeHHbI
npenapat Tolbko no Ha3haeHnIO Bpaa, cnpoCnTe ero O npoJOnKInTeJIbHOCTN INrAJaUuN I NOxOJa- uem dIy Bac KOnUeCTBe JekapCTBeHHoro npenapata.
- EcIn Ha3HaueHHeo KOJUeCTBO JeKapCTBeHHOro npenapaTa MeHbWe 2 Ml, Do6aBbTe N3OTOnHueCKn fN3IONoRnueCKm pactBOp, DoBeJa O6bEm KNDKOCTn KaK MnHMym Do 4 Ml. Heo6xOIMo TaKKe pa36abTb rYcTBie MeiKamEHTbl. Ppr 3OM TaKKe cJeDyET co6IIOdaTb yKa3aHnB Bpa4a.
3. 3akpbTne pacbnlnteJra
3aKpOte pacbIInTeIb [9], nobepHyB BepxHIOU qactb Kopnyca no yacobon cTpeJIke OTHOCITeIbHO EMKoCTn dIy IekapCTBeHHoro npenapaTa [11]. CJeIte 3a npabunbHoCTbIO coeINHeHNA!
BlaXHOCTb MOxET pINBeCTN K NOBbIeHIO pNcKa pa3MHOxEHHa6aKTePn.
NoDROTOBka
Cpa3y nocne kaxdoi npoceDypb BCE deTaN pacnblnte [9], a taKxe nCNOlb3OBAHHbIe npuHaJNeXHOCTN Heo6xOIMO ONUCTNTb OT OCTaTKOB JeKapCTBeHHoro npenapaTa n 3arpy3HeHn.
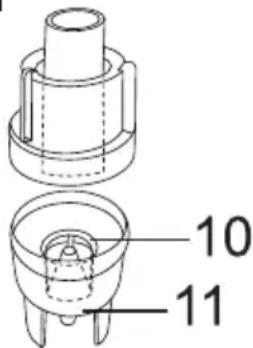
Pa36epnte pacnblnteJIb [9] Ha coCTaBHbIe YaCTN.
- N3BNeKInTe MyHdUToYk [12] n3 pacNbIInTeJIa.
Pa36epnte pacnbIInTeIb, NOBepHyB BepXHIOU qactb npOTnB YacBOO CTpeJIKN OTHCnTeJbHO EMKOCTN IJIa NEkapCTBeHHoro npenapata [11].
- ɪЗbvěknte paçnbɪnɪteɪbHyú Ḥаcaɪkʌy [10]ɪz emkoctɪŋ ṅπεkapCTBɛHHOrO npɛnapaTa [11].
- C6opka BbINOJIHЯETcNo aHaIOrnB o6paTHoNocJeIOBaTeIbHOCTN.
OuInCTka

BHHMaHHe
Ipeed KaJdoOn OuncTkoY 6eDInTeCb, YTO npN6Op BblKJIIOueH, OTCOeDInHeOT CETNI OCTblJ.
PacnbIInTeB nICNoJIb3OBAHHbIe npHaadJeXHocTn, TaKne KaK MyHdUHTyK, MaCKa, pacNbIInTeJbHaar Ha- caJaT. D. NocLe KaxDOro npImeHEnr Heo6xOIMC npOMBaTb TropueH, Ho HE KINpaUe BDOi. TuaTeJbHO BbICuNTe DeTaII MraKoN TkaHbIO. NocLe NoHoro BbCbIXAHNA Co6epnte DeTaII IN NOLOXInTe Ix B cyXyO repMeTuHyO EMKoCTb IIN pNoDe3HfNcNpyTe.
Pn Ounchke y6eHITecb, YTO BCE OCTaTKydaJIeHbl. Pn 3OM H N B KOEM Clyuae He NCNOJb3yIte BeUeCTBa, KOTOpBIE MOrY T 6bITb NOTEHuaJIbHO ONaCHbIMN Pn INonaHaHn Ha KOKy IIN CIn3NCTyO oBOJouKy, PpOrIaTbIBaHn IIN BDbIXaHn.
Iy Ouchk np6opa nCNoJIb3yIte MmKyo, cyxuToTpyNKy I Hea6pa3nBHOe YnCTraue CpeDCTBO.
He nCnoIb3yITe cIbHoDeIcTByIOxuX uNCTaux CpeIcTB u He IorpykaIte npI6Op B Body.

BhimaHne
CJeTe 3a Tem, YTo6bI BHyTpB npi6opa He nonana BOda!
He MoTe np6Op n npHaJNeXKHOCTN B NOcyDoMOeHOn MaunHe!
He npikacaiTecb K noqKJIIOueHHOMy K cTe n np60py BnaXhIMn pykam; n36eAaTe nonaHa H BODbl Ha np6Op. Pnp6op pa3peWaeTcN cNoJb3ObaTb TOnbko a6COJIOTHO cyxM.
- He pacnbIyIe Te XnIDKOCTb B BeHTnJIaCIOHbIe OTBepCTnI! NonaIaNHe XnIDKOCTe MoKTe npNBecTn K NOBpeXKeHnIO 3JeKTPnueCKNX u DpyrNX DeTaJEn INHraIyTopa, a TaKxE K C6OaM B erO pa6Ote.
KoHdEHCaT, 06CnyXnBaHne Tpy6Kn
B 3aBnCmOCTn OT yCNoBn OkpyKaIOSe CpeDbI B Tpy6Ke MoKet O6pa3OBbIBaTbC4 KOHDeHcAT. YTo6bI npEnrTCTBOBaTb PoCTy MmKpOoPraHn3MOB n rapaHTnpoBaTb 6e3ynpeHoe IueHHe, BlaRy Heo6xOJMoUydaJIaTb. Jnra 3TOrO Heo6xOJMo CpeNaTb CneDuOJee:
- N3BNeKeNTe Tpy6Ky [8] n3 pacnblnteJ [9].
- OctaBbTe Tpy6ky B INrAJaTOpE [6].
OCTaBbTe INHraJIaTOp BKNIOUeHHbIM Do TEX NOp, NOKa NOTOK BO3dYxa He ydaIIT BcO Bnary. - Ppi CunbHOM 3aqr3HeHH 3aMeHHTe Tpy6Ky.
De3nHΦekunr
Ppi npoBeHn De3nHΦeKcHn pacbIInTeJ TuaTeJIbHO co6JIIOaIte cIeDyUOuNe npaBnla. PekomeHdyETcA De3nHΦuCIPoBaTb DeTaNn pni6opa He pexeHem NocJe NocJIeNHeRo pImMeHEnra 3a DeHb.
(Для ЭTORO Bamnotpe6yeTcHemHORO 6ecuBETHORO yKcyca N dNCTnIINPObAHHO BODbl.)
- Chauana ouhnte paonblntelb npHaJeXHoCTn, KaK onucaHO bpa3neJe «Ouictka».
Pa3o6paHHbI paCnblnteHb I MyHdTuK nOIOXHTb Ha 5 MInHyT B KINrAryu BOy.
-Дя npOuHx npHaJnxKHOCTe nCNoIb3yIte paTbOp, COCTOaUHn Ha 1/4 n3 yKcyca n Ha 3/4 n3 dNtUInpoBaHHo BObl. Y6eINTEcb, YTO XnIDKOCTn DOCTaTOUHO, YTObIMoXHo 6blIO NOnHOCTBIO NOrpy3NTb B HeE DetaII. - OctaBbTe DeTaJIN Ha 30 MInHyT B yKcychom pactBOpe.
- PpomOte DeTaN BDOI IN pa3IOXnTe n3 IJRA pOCyUKN Ha MmKOM NOLOTHe.

BHHMaHHe
Mackn BO3dyuHbI JlnaHr He NoDBepraTb KINpaHe HIO Hc NOMEuTaB B aBtOKJIaB.
- Nocne noJHoro BbICbIXaHnna co6epnte deTann n noJIOKNTe INB CyXyI RoPeMtUHyO eMKoCTb.

yka3aHne
O6paNTe BHNMaHHe, YTO npn nn nocJe ouNCTKn Heo6xOIMo NOLHOCTbIO BbICyUHTb DeTaJI, TaK KaK B IpOTNBOM Clyuae BO3pactaET pUCK pa3MHoxKeHHa 6akTepni.
CTepnna
- IcnoIb3yIte paCTBOpbl dny xOJIoHOn cTepeuIN3aUIN B COOTBeTCTBUN C yKa3aHnA M N IpON3BOJNTeJI.
Cyushka
- IonoKnte coCTaBhIe DeTaJIH Ha cyxuO uNCTyU O BnITbBAIOU BnaIg NODCTUNKy N OCTaBBTe Do NOJHOrO BbcbIXAHNA (He MeHee 4 yacOB).
UcToHbOCTb MaTePnana
- KaN Bce DeTaN N3 PIIaCTNka, paCnblntelb n erO npHaJIeXHOCTN 3HaIINBAOTc npN qAcTOM NCIOB3OBAHNN IITNeHneCKoO6pAbOTke. Co BpeMeHem 3TO MOKeT pPnBeCTN K IN3MeHEnIO COCTaBa a3po3OJN, KaK CLeIcTBnE, CHN3NTb 3ΦΦeKTNBHOCTb JueHn. Po3TOMy peKOMeHdyETcR MeHrTb paCnblntelb exKeOrDoHo.
-Пи Вьборе срсдва ДЯОчСКИ ДЕЗнфЕКЦИ Нeo6хODIMO yuHTbIBaTb CLEДуОшee: ИСПОЛБ3УТe ТольКо мягКоупстяшee срсдTOВОДИDEЗнФИ цИPyUОшee срсдTOВ KOLИЧЕСТBE, yka3aHHOM ПюИЗВОДИТЕЛЕМ.
XpaHeHne
HeIb3xpaHHTb np6Op BO BnaXhbx NOMeUeHnIX (HaNPmEp, B BaHHo KOMHaTe) npeBo3ntb erO BMeCTe C BnaXhIMN PpeMetAm.
XpaHnTe npeB03nTe np6Op B MeCTe, 3aUuHem HOM OT dIInTeJbHOrO Bo3dEINCTBnA INpRmblx CoJIHeyHbIX Jyuei.
-Пинадлжно мохноХpaHntbВспально npedHa3HaueHHOMдЯЗTOROOTceke[4].XpaHnte npi6opBcYxOMMeCTe,nyuWeBCero,BynaKOBKe.
9. Ytuln3aun
B ueJx 3aunTbI OkpykaIoUeI cpebl np6Op CJeDyET yTNIN3InpOBA Tb OTdJIbHO OT 6bITOBORo Mycopa.
Pn6op cIeNyET yTINuN3npoBaTb corlacho DnpeKtNBc EC no OTxOaam 3JekTpuecko- ro n 3JektpoHoro o6OpyDobAHn - WEEE (Waste Electrical and Electronic Equipment).

Pn BO3HKnHOBENBONpOCOB, ObaaIteCB KOMMyHaJIbHyO opraHn3aUIO, 3aHmAmIoUyOcR BONpocAMN yTINn3aUIN.
- YcTpaHHe HEnCnPaBHOCTeI
| ELECTROMAGNETIC COMPATIBILITY INFORMATION | ||
| Guidance and manufacturer's declaration - electromagnetic emissions | ||
| The SIH21/2 is intended for use in the electromagnetic environment specified below. The customer or the user of the SIH21/2 should assure that it is used in such an environment | ||
| Emissions Compliance Electromagnetic environment - guidance | ||
| RF emissionsCISPR 11 | Group 1 | The SIH21/2 uses RF energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. |
| RF emissionsCISPR 11 | Class B The SIH21 | 2 is suitable for use in all establish-ments other than domestic, and may be used in domestic establishments and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes, provided the following warning is heeded:Warning: This equipment/system is intended for use by healthcare professionals only. This equipment/ system may cause radio interference or may disrupt the operation of nearby equipment. It may be necessary to take mitigation measures, such as re-orienting or relocating the SIH21/2 or shielding the location. |
| Harmonic emissionsIEC 61000-3-2 | Class A | |
| Voltage fluctuations/flicker emissionsIEC 61000-3-3 | Complies | |
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The SIH21/2 is intended for use in the electromagnetic environment specified below. The customer or the user of the SIH21/2 should assure that it is used in such an environment | |||
| Immunity test EN 60601 test level | Compliance level | Electromagnetic environment - guidance | |
| Electrostatic discharge (ESD) IEC 61000-4-2 | ±6 kV contact ±8 kV air | ±6 kV contact ±8 kV air | Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30 % |
| Electrical fast transient/burst IEC 61000-4-4 | ±2 kV for power supply lines ±1 kV for input/output lines | ±2 kV for power supply lines | Mains power quality should be that of a typical commercial or hospital environment. The electrical fast transient burst (EFT) is generated by the switching of inductive loads. Separation between the equipment and other loads shall be considered before installation. Mains filter is required, if necessary. |
| Surge IEC 61000-4-5 | ±1 kV line(s) to line(s) ±2 kV line(s) to earth | ±1 kV line(s) to line(s) | Mains power quality should be that of a typical commercial or hospital environment. |
| Voltage dips, short interruptions and voltage variations on power supply input lines IEC 61000-4-11 | <5 % U_T (>95 % dip in U_T) for 0,5 cycle 40 % U_T (60 % dip in U_T) for 5 cycles 70 % U_T (30 % dip in U_T) for 25 cycles <5 % U_T (>95 % dip in U_T) for 5s | <5 % U_T (>95 % dip in U_T) for 0,5 cycle 40 % U_T (60 % dip in U_T) for 5 cycles 70 % U_T (30 % dip in U_T) for 25 cycles <5 % U_T (>95 % dip in U_T) for 5s | Mains power quality should be that of a typical commercial or hospital environment. If the user of the SIH21/2 requires continued operation during power mains interruptions, it is recommended the SIH21/2 be powered from an uninterruptible power supply or a battery. |
| Power frequency (50/60Hz) magnetic field IEC 61000-4-8 | 3 A/m 3 A/m | Power frequency magnetic fields should be at levels characteristic of a typical location in a typical commercial or hospital environment. | |
| NOTE U_t is the a.c. mains voltage prior to application of the test level | |||
| Immunity test EN 60601 | test level | Compliance level | Electromagnetic environment - guidance |
| Conducted RFIEC 61000-4-6Radiated RFIEC 61000-4-3 | 3 Vrms150 kHz to80 MHz3 V/m80 MHz to2.5 GHz | 3 V3 V/m | Portable and mobile RF communicationsequipment should be used no closer to anypart of the SIH21/2, including cables, than therecommended separation distance calculatedfrom the equation applicable to the frequencyof the transmitter.\( d = \left[\frac{3.5}{V_1}\right]\sqrt{P} \)\( d = \left[\frac{3.5}{E_1}\right]\sqrt{P} \)80 MHz to 800 MHz\( d = \left[\frac{7}{E_1}\right]\sqrt{P} \)800 MHz to 2,5 GHzwhere p is the maximum output power ratingof the transmitter in watts (W) according to thenot transmitted separation distance in metres (m)Field strengths from fixed RF transmitters,as determined by an electromagnetic sitesurvey,a should be less than the compliancelevel in each frequency range.bInterference may occur in the vicinity of equipment marked with the following symbol:(( )) |
| NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies.NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected byabsorption and reflection from structures, objects and people. | |||
| aField strengths from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and landmobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoreticallywith accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagneticsite survey should be considered. If the measured field strength in the location in which the SIH21/2 is usedexceeds the applicable RF compliance level above, the SIH21/2 should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as re-orienting orrelocating the SIH21/2.bOver the frequency range 150 kHz to 80 MHz, field strengths should be less than 3 V/m. | |||
| Recommended separation distances between portable and mobile RF communications equipment and the SIH21/2 | |||
| The SIH21/2 is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the SIH21/2 can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and the SIH21/2 as recommended below, according to the maximum output power of the communications equipment. | |||
| Rated maximum output power of transmitter W | Separation distance according to frequency of transmitter m | ||
| 150 kHz to 80 MHz 80 MHz | Hz to 800 MHz 800 MHz to | 2.5 GHz | |
| 0.01 0.12 0.12 0.23 | |||
| 0.1 0.37 0.38 0.73 | |||
| 1 1.2 1.2 2.3 | |||
| 10 3.8 3.8 7.3 | |||
| 100 12 12 23 | |||
| For transmitters rated at a maximum output power not listed above, the recommended separation distance in metres (m) can be estimated using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer. NOTE 1 At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies. NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||