TheraFace Mask - Skin care device THERABODY - Free user manual and instructions
Find the device manual for free TheraFace Mask THERABODY in PDF.

User questions about TheraFace Mask THERABODY
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Skin care device in PDF format for free! Find your manual TheraFace Mask - THERABODY and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. TheraFace Mask by THERABODY.
USER MANUAL TheraFace Mask THERABODY
Powered by QX-Micro Motor Technology
Clinitally Proven LED Technology
User Manual
TABLE OF CONTENTS
Language
EN 2-14
ES 15-26
FR 27-38
IT 39-50
DE 51-62
ESMX 63-75
FRCA 76-88
SC 89-100
KR 101-112
Product Overview

TheraFace Mask
The most advanced LED mask with targeted vibration.
TheraFace Mask is the only one of its kind to help relax, restore and rejuvenate your skin. This mask combines 648 full-face Red, Blue, and Red + Infrared LED lights and has been FDA-Cleared with the Following Indications for Use:
Red Light is intended to treat full-face wrinkles.
Blue Light is intended to treat mild to moderate inflammatory acne.
Red+Infrared Light is intended to treat full-face wrinkles.
The device is safe for use on all skin types (Fitzpatrick Types 1-6).
Intended Use
Red Light is intended to treat full face wrinkles
- Blue Light is intended to treat mild to moderate inflammatory acne
Red ^+ Infrared Light is intended to treat full face wrinkles
What's in the Box:
TheraFace Mask Device
Eyeshield (assembled)
Non-chargeable Device Stand
USB-C Cable
Instruction Manual
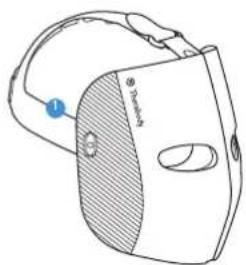
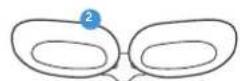
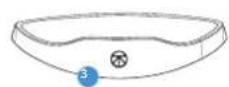



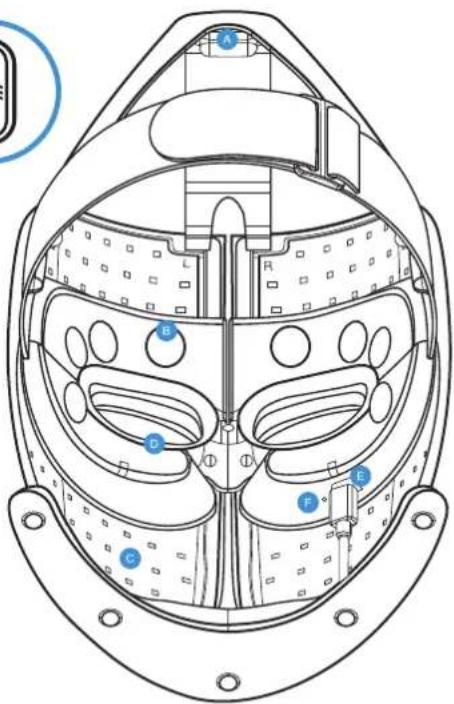
Getting To Know Your TheraFace Mask Device.
Head Massage Units
Face Massage Pads
Multi-color LEDs
Eyeshield
USB-C port
LEDindicator
LED button
Vibration button
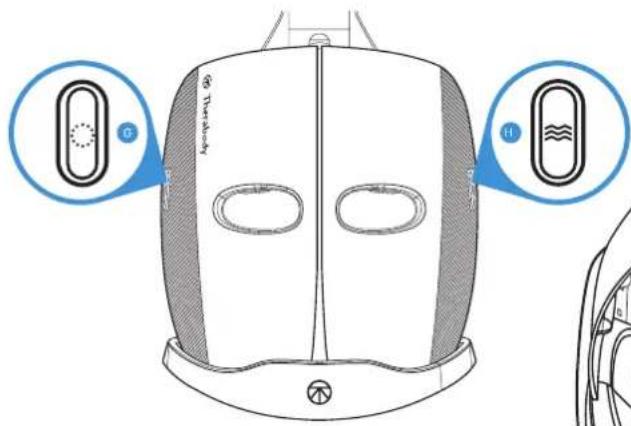
Getting Started
Powering the TheraFace Mask Device On and Off.
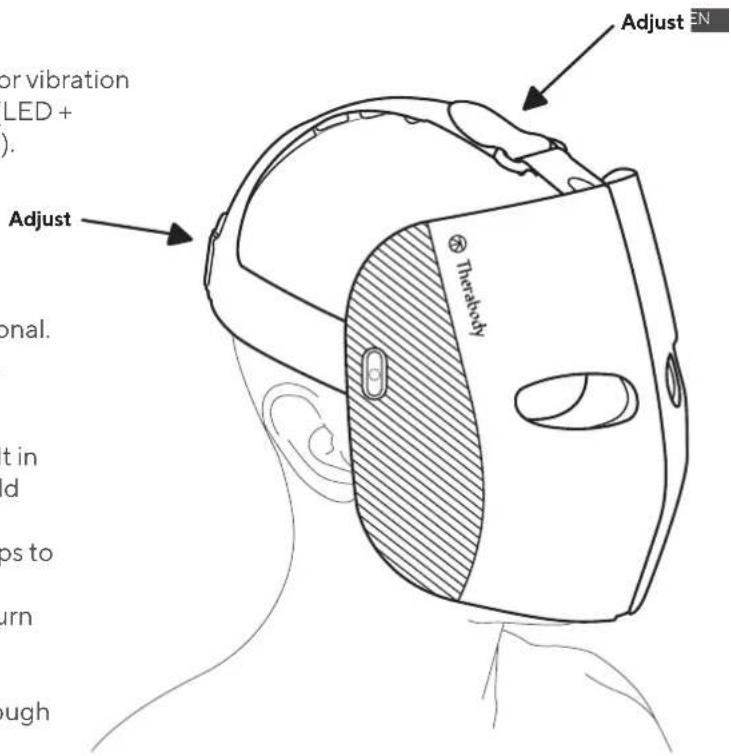
Long press the LED button (G) to power the device ON and turn on LED light and vibration massage therapy. The device will guide you through a 9 minute science-backed treatment that cycles through Red, Red + Infrared, and Blue LED light therapy in combination with unique vibration patterns. Long press the same button to power the device OFF.
Long press the vibration button (H) to power the device ON to vibration-only mode. Short press the vibration button during the treatment to change the vibration mode. Long press the same button to power the device OFF.
Automatic Shut Off
If the device is not manually powered off by long pressing the LED or vibration button, the device will automatically shut off after each treatment (LED + vibration, 9-minute treatment; vibration only, 15-minute treatment).
Basic getting started steps:
- Begin with a clean, dry face
- Slide the TheraFace Mask eyeshield into the mask through the protrusions that meet near the nose. Use of the eyeshield is optional. Using the eyeshield can reduce strain on the eyes from the light. If you choose not to use the eyeshield, keep your eyes closed throughout your treatment. Do not look into LED lights during treatment. *Normal use of TheraFace Mask eyeshields may result in redness around your eye area. Some redness is normal and should subside within 5-10 minutes of using TheraFace Mask.
- Place the device on your face, and adjust both of the Velcro straps to your desired fit
- To turn on the device, either Long press the LED button (G) to turn ON LED light + vibration, or the vibration button (H) to turn ON vibration only
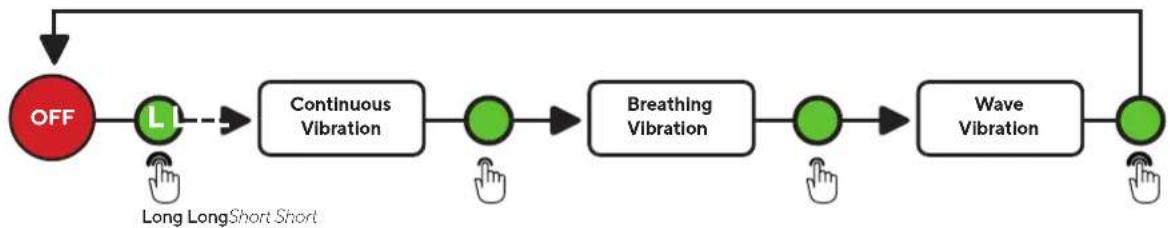
- Short press the vibration button (H) or LED button to toggle through each vibration mode (Continuous, Breathing, and Wave)
- Enjoy your desired treatment.
*Normal use of TheraFace Mask increases blood flow throughout your face, which may result in redness around your forehead, temples, and bottom of your eyes. Some redness is normal and should subside within 5-10 minutes of using TheraFace Mask.

Using TheraFace Mask Device
Full Face Light Therapy
- Place the device securely and comfortably on your face.
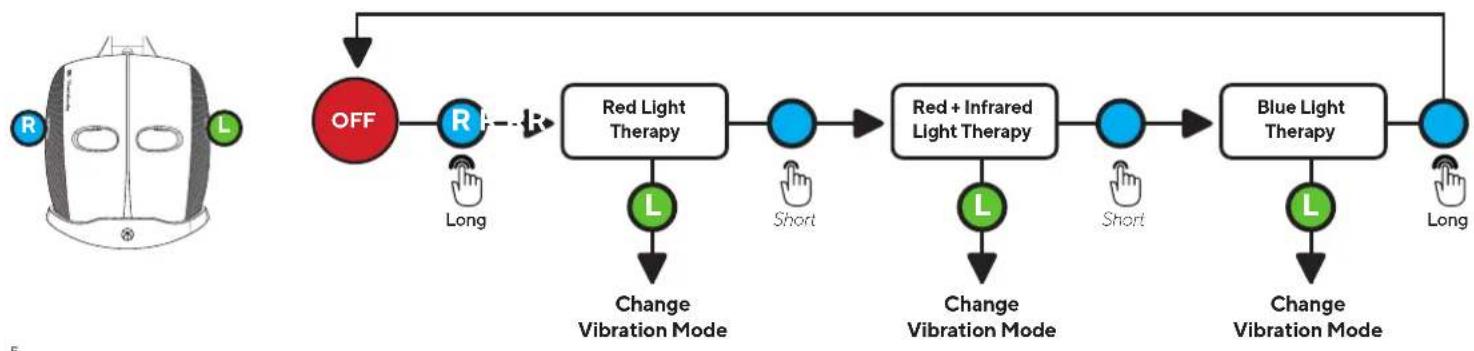
- The TheraFace Mask has three LED light therapy wavelength options: Red, Red + Infrared, Blue.
- Long press the LED button (G) to turn ON the LED light and vibration. This will turn on the 648 multi-color LED lights around the face, the eight vibration motors around the eyes, and the nine vibration motors on the top and back of the head. Face and head vibration therapy accompanies LED light treatment during Red and Red + Infrared treatment. Only head vibration therapy is available during Blue Light treatment.
- When you turn ON the device, it will begin an automatic Red Light + vibration treatment. In this mode, the device will guide you through a science-backed treatment that cycles through Red (3 minutes), Red + Infrared (3 minutes), and Blue (3 minutes) light therapy in combination with vibration patterns, for a 9-minute treatment.
- Short press the LED button (G) to toggle through each of the three LED light modes (Red, Red + Infrared, Blue). Each LED light mode is a 3-minute treatment.
- Short press the vibration button (H) to toggle through each of the three vibration pattern modes (Continuous, Breathing, and Wave).
- Long press the LED button (G) to power the device OFF.
Vibration Therapy
- Place the device securely and comfortably on your face and long press the vibration button (H) to turn ON vibration only. This will turn on the eight vibration motors around the eyes and the nine vibration motors on the top and back of the head.
- When you turn ON the device, it will begin on the Continuous mode. Short press the vibration button (H) to toggle through each of the three vibration pattern modes (Continuous, Breathing, and Wave). The treatment time is 15 minutes.
- Short press the vibration button (H) to reach the second vibration mode, Breathing. Allow the motors to guide you through a breathing exercise. First, breathe in for five seconds as the vibration intensity increases. The high-speed vibration is your cue to begin to exhale. Continue to exhale as the vibration gradually slows.
- Short press the vibration button (H) to reach the third vibration mode, Wave. Sequential vibration patterns will move from face to head during the Wave treatment.
- Long press the vibration button (H) to power the device OFF.

TheraFace Mask Device After Care & Cleaning
Device Maintenance
The following maintenance instructions are important to ensure that your device continues to work as it was designed. Failure to follow these instructions may cause your device to stop working.
Care and Cleaning
- Wipe the device clean with a disposable cloth after each use.
- Before cleaning, power the device OFF by long pressing the LED light button (G) or vibration button (H); ensure that your device is not connected to a charging cable or power source before cleaning.
- Visually inspect the device for any obvious signs of debris build-up.
- Wipe off any visible dirt with a disposable cloth moistened with a mild detergent. Be sure to wipe the interior surfaces of the mask. Next, use a soft cloth stained with 70% isopropyl alcohol to wipe and disinfect the mask for three (3) minutes, especially the inner surface of the mask. Repeat this process three times. Finally, wipe the disinfectant with a clean, dry cloth or towel until there is no visible residue. Do not submerge the device in water or apply excessive cleaning solution or disinfectant when cleaning.
- If you use the TheraFace Mask eyeshield during your treatment, wipe clean.
- After cleaning, allow the device to dry thoroughly before storing or beginning another treatment. A properly cleaned device should have no visible signs of debris or moisture.
Charging:
- To check the battery level, short press the LED light button (G) or the vibration button (H).
Each light color corresponds with a battery level:
Orange:Low-charge
Blue: Mid-charge
Green:Full-charge
Before charging the TheraFace Mask, power the device OFF by long pressing the LED button (G) or vibration button (H).
- Connect the TheraFace Mask to the USB-C cable. The port is located inside the mask.
- The battery level is displayed next to the USB-C port.
- The LED will flash to indicate that the device is connected properly and charging. This light will turn green when charging is complete.
Note: If using an alternative USB-C cable, ensure that it is from a trusted source and has not suffered any structural damage. Do not try to use the device while charging.
TheraFace Mask ProductWarnings and Guidance (Precautions and Contraindications)
Background
Therabody products are designed to unlock the body's natural ability to achieve health and well-being. Through science and technology, the Therabody portfolio allows people to access the therapeutic benefits of different natural phenomena to meet both their needs and preferences. There will be times when it is advisable to modify how devices are used (precautions) or times when it is not appropriate to use certain devices (contraindications). Read the following safety information for TheraFace Mask in its entirety prior to use.
Important Safety Information
General TheraFace Mask Use
Read the fullWarnings and Guidance prior to using the TheraFace Mask device.
This device is intended for use by people in good health. This device is contraindicated against and should not be used by or on anyone with a history of epilepsy, seizures or cardiopathy. The TheraFace Mask device is not recommended for anyone with an electronic implanted device (such as a pacemaker), cardiac arrhythmia, tumors, or acute episodes of inflammatory diseases. The device is not recommended for those who have arteriosclerosis, thromboses, or implants in the body region being treated. The device should not be used if you have dark brown or black spots, such as large freckles, birthmarks, moles, or warts, on the area being treated. The device is not recommended if you have eczema, psoriasis, lesions, open wounds, or active infections other than mild to moderate inflammatory acne, such as cold sores, in the area being treated. Wait for the infected area to heal before using the device. The device should not be used if you have abnormal skin conditions caused by diabetes or other systemic or metabolic diseases. If you have a history of herpes outbreaks in the area of treatment, use of the device is not recommended unless you have consulted with your physician and have received preventive treatment. Please consult your physician prior to using the device if you are pregnant and/or nursing.
Immediately stop using the device at the first sign of discomfort.
Adult supervision should be provided for those under the age of 18 using this device. If you have any medical considerations, are taking any medications that cause light sensitivity, or have had any facial surgery or other surgical procedures, please consult your doctor before using the device.

Safety, Precautions, and Contraindications
TheraFace Mask LED light therapy (Red LED, Red+IR LED, and Blue LED Therapies)
These recommendations are derived from consultation with medical experts and published research regarding precautions and contraindications as of the printing date. For up-to-date information, please visit us online at https://www.therabody.com/us/en-us/precautions-and-contraindications.html.
Precautions
Due care is required in these circumstances and device use may need to be modified. Consult with a medical professional if you currently have or suspect you may have any of the following conditions or if you have any questions.
Recent injury, surgery, or facial treatment, including neurotoxin, dermal filler, microneedling, laser, and chemical peel until the skin has fully healed.
Current Herpes Simplex Virus breakout
- Broken skin
- Retinol application before use of red LED light
Contraindications
The following are circumstances where the potential risks may outweigh the benefits. Consult a medical professional before use.
- Skin rash, open wounds, blisters, local tissue inflammation, infection, bruises, or tumors
- Pregnancy/nursing
Abnormal sensations (e.g., numbness)
Cancer/tumors
Epilepsy
Cardiopathy (heart disease) - Photo allergy or disorder (e.g., Lupus, porphyria)
Medications that cause light sensitivity
Medications for severe inflammatory acne - Extreme sensitivity to light
- Melasma or hyperpigmentation (especially if exacerbated by mild warmth)
- Suspicious lesions or skin cancer - please consult your physician
- If taking or using any retinol or other sun-sensitive medications, products, or benzoyl peroxide, do not use infrared light
- Allergy to the device material (Lycra fabric and medical grade transparent TPE and silicone)
Limited Warranty
For full warranty information, please visit www.therabody.com/warranty. To request a copy of the warranty by mail, you may send a request to the following address:
Therabody-Warranty
Attn: Customer Service
6100 Wilshire Blvd. Ste 200
Los Angeles, Ca. 90048
TheraFace Mask Vibration Therapy
These recommendations are derived from consultation with medical experts and published research regarding precautions and contraindications as of the printing date. For up-to-date information, please visit us online at https://www.therabody.com/us/en-us/precautions-and-contraindications.html.
Precautions
Due care is required in these circumstances and device use may need to be modified. Consult with a medical professional if you currently have or suspect you may have any of the following conditions or if you have any questions.
Recent injury, surgery, or facial treatment, including neurotoxin, dermal filler, microneedling, laser, and chemical peel until the skin has fully healed.
Current Herpes Simplex Virus breakout
- Broken skin
Hypertension (controlled)
Abnormal sensations (e.g., numbness)
Sensitivity to pressure
Medications that may alter sensations
Contraindications
The following are circumstances where the potential risks may outweigh the benefits.
Consult a medical professional before use.
Skin rash, open wounds, blisters, local tissue inflammation, infection, bruises, or tumors
Active inflammatory acne breakout
Bone fracture or myositis ossii
Hypertension (uncontrolled)
- Acute or severe cardiac, liver, or kidney disease
- Neurologic conditions resulting in loss or altered sensation
- Bleeding disorders
Recent surgery or injury
Connective tissue disorders
- Peripheral vascular insufficiency or disease
Medications that thin the blood or alter sensations
- Direct placement over surgical site or hardware
Extreme discomfort or pain
- Pacemaker, ICD, or history of embolism
Please note, this is not a return address or a retail location. No Therabody products or packages will be accepted at this location.
Customers who are in need of product support should visit https://www.therabody.com/us/en-us/support/support.html for the available contact methods.
FDA-Cleared
Limited Warranty Only With Authorized Retailer Purchase
©2023 Therabody, Inc. All Rights Reserved.
Patents and Pending Patents at www.therabody.com/patents.
Distributed by:
Therabody International Limited, Marine House, Clanwilliam Place, Dublin, Ireland
Therabody UK Limited, 55 Ludgate Hill, 2nd Floor, London EC4M7JW
Therabody Australia Pty. Ltd., The Commons South Yarra, 11 Wilson Street, South Yarra VIC 3141 australia@therabodycorp.com

UNITWARNINGS
BEFORE USING OR CHARGING THE TheraFace Mask, READ ALL INSTRUCTIONS AND CAUTIONARY MARKINGS IN THIS MANUAL, ON THE CHARGER, AND THE TheraFace Mask. If your device doesn't turn on or the battery indicator displays a low battery level, please charge before first use. The TheraFace Mask is intended for over-the-counter use.
This TheraFace Mask is not intended to diagnose, cure, or prevent diseases. Therabody strives to make the TheraFace Mask as safe for intended use as possible. This is an advanced mechanical tool with electric components. If the TheraFace Mask and its accessories are not used or maintained properly, there is a risk of fire, electric shock, or injury. When using the TheraFace Mask, the following basic precautions should always be adhered to:
-
USE ONLY AS INstructed.Use the TheraFace Mask as described in the TheraFace Mask User Manual only.Use only Therabody recommended accessories and replacement parts.Do not carry out any maintenance other than as advised by Therabody.
-
NOT FOR CHILDREN, The TheraFace Mask and charger are not intended for use by young children or persons with reduced physical, sensory, or reasoning capabilities, or lack of experience and knowledge. The TheraFace Mask is not to be used as a toy. Do not play with bend, or pull the electrical components. Advise children not to play with the TheraFace Mask or charger.
-
CHARGING LOCATIONS. Charge the TheraFace Mask with a USB-C charger. The TheraFace Mask should be charged indoors in a well-ventilated, dry location. Do not charge the TheraFace Mask outdoors, in a bathroom, or within 10 feet (3.1 meters) of a bathtub, shower, or pool. Do not use the TheraFace Mask or charger on wet surfaces, and do not expose the charger to moisture, rain, or snow. Do not use the TheraFace Mask or its compatible charger in the presence of explosive atmospheres (gaseous fumes, dust, or flammable materials). Sparks may be generated, possibly causing a fire.
-
DO NOT OVERCHARGE. Do not leave the device connected to the charger for more than one hour after the battery has been fully charged. The battery includes a protection system to avoid the risk of overcharging. However, overcharging may reduce its life over time.
-
DONTCRUSH, DROP, OR DAMAGHTHE DEVICE OR CHARGER. Do not use a charger that has received a sharp blow, been dropped, run over, or damaged in any way.
-
BATTERY CHEMICALS CAUSE SERIOUS BURNS. Never allow the internal battery to come into contact with the skin, eyes, or mouth. If a damaged battery leaks chemicals, use rubber or neoprene gloves to dispose of it. If skin is exposed to battery fluids, wash with soap and water and rinse with vinegar. If eyes are exposed to battery chemicals, immediately flush with water for 20 minutes and seek medical attention. Remove and dispose of contaminated clothing.
-
DO NOT SHORT CIRCUIT. A battery will short circuit if a metal object makes a connection between the positive and negative contacts on the battery or the 16V connector. Do not place a battery near anything that may cause a short circuit, such as coins, keys, or nails in your pocket. A short circuited battery may cause fire and personal injury.
-
DEVICE DISPOSAL. This device contains a lithium-ion battery, and care must be taken upon disposal of the device. Before disposal of this device, please review your local laws and requirements surrounding Lithium Ion Battery disposal. The preferred method of disposal is recycling the whole device.
-
SERVICE. If the device is not working properly, has received a sharp blow, or has been dropped, damaged, left outdoors, or dropped into water, then do not use it. Do not attempt to repair or disassemble the device which may result in an electric shock or fire.
t treatment immediately and remove the device. Discontinue use of the device if it overheats or becomes uncomfortably hot.
is not responsible for damages that may occur due to the use of third-party chargers.
-
DONT OPERATE UNDER BLANKET AND PILLOW. Excessive heating can occur and cause fire, electric shock, or injury.
-
BATTERY. There is only one correct USB-C insertion position on the device. Do not force the cable into place. The Therabody logo should be facing upright when inserted.
The TheraFace Mask, batteries, or charger where temperatures may exceed 40^ / 104^ such as in direct sunlight, in a vehicle, or in a metal building during the summer.
- DEVICE CARE. The TheraFace Mask is NOT waterproof. The device is not machine washable. Do not place or store the device where it can fall or be pulled into a tub or sink. Do not place in or drop into water or other liquid. Do not reach for an appliance that has fallen into or come into contact with water. Unplug immediately. Clean the device according to the instructions found in the "After Care and Cleaning" section above. DO NOT DISASSEMBLE. Disassembly or incorrect reassembly may result in the risk of electric shock, fire, or exposure to battery chemicals. The warranty will be void if the device, batteries, or charger are damaged.
disassembled or if any parts have been removed.

Troubleshooting
| ISSUE | POSSIBLE CAUSE CORRECTIVE ACTION | |
| Device does not power on 1. Battery level is low Charge the device | ||
| 2. It still can't be turned on Contact Therabody | ||
| Vibration motor does not turn | 1. Motor or connection has been damaged | Try charging the device fully, removing the charging cable and then pressing left button for 3 seconds. If problem persists, contact Therabody |
| The battery won't charge | 1. The ambient temperature is too high or too low | Charge the device when the ambient temperature is between 5°C and 35°C |
| 2. Possible defective battery | Contact Therabody | |
| 3. Defective charging cable | Try using a different usb-c cable and adapter | |
Labels
| IP 22 | Degree of protection against ingress of water On rating label |
| Read instructions before use On rating label | |
| Level of protection type BF applied part On rating label | |
| CE | In accordance with Directive 2014/30/EU electromagnetic compatibility On rating label (only in the EU) |
| UK CA | In accordance with UK Regulations On rating label (only in the EU) |
| Therabody, Inc. On rating label Los Angeles, CA 90048 |
Labels
| SYMBOLS DESCRIPTION LOCATION | |
| Unique Device Identification (UDI) On rating label | |
| Separate collection for waste electrical and electronic equipment. Note: For more information about disposal of equipment, its parts and accessories, please contact your local distributor. | |
| RCM Mark On rating label | |
| Operating /Storage environment: Temperature, Operating: 0 ~ 35 °C Relative Humidity, Operating: ≤85% Temperature, Storage: -10 ~ 50 °C Relative Humidity, Storage/Shipping: ≤85% Atmospheric Pressure: 70.0 kPa _106.0 kPa |
Guidance and manufacturer's declaration - electromagnetic emission - for all EQUIPMENT AND SYSTEMS

GUIDANCE AND MANUFACTURER'S DECLARATION - ELECTROMAGNETIC EMISSION
| The TheraFace Mask is intended for use in the electromagnetic environment specified below. The customer or the user of TheraFace Mask should assure that it is used in such an environment. | ||
| EMISSIONS TEST COMPLIANCE ELECTROMAGNETIC ENVIRONMENTENT - GUIDANCE | ||
| RF emissions CISPR 11 | Group I | The TheraFace Mask uses RF energy only for its internal function. There for its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. |
| RF emissions CISPR 11 | Class B | The TheraFace Mask suitable for use in all establishments, including domestic establishments and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes. |
| Harmonic emissions IEC 61000-3-2 | Class A | |
| Voltage function / flicker emissions IEC 61000-3-3 | Complies | |
GUIDANCE AND MANUFACTURER'S DECLARATION - ELECTROMAGNETIC IMMUNITY
| The TheraFace Mask is intended for use in the electromagnetic environment specified below. The customer or the user of the TheraFace Mask should assure that it is used in such an environment. | |||
| IMMUNITY TEST IEC 60601 TEST LEVEL | COMPLIANCE LEVEL ELECTROMAGNETIC ENVIRONMENT - GUIDANCE | ||
| Electrostatic discharge (ESD)IEC 61000-4-2 | ±8 kV contact±2 kV, ±4 kV, ±0 kV, ±15 kV air | ±6 kV contact±2 kV, ±4 kV, ±8 kV, ±15 kV air | Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30%. |
| Electrostatic transient / burstIEC 61000-4-4 | ±2kV for power supply lines±1kV for input/output lines | ±2kV for power supply lines Mains power quality should be that of a typical commercial or home environment. | |
| SurgeIEC 61000-4-5 | ±1kV differential mode±2kV common mode | ±1kV differential mode Mains power quality should be that of a typical commercial or home environment. | |
| Voltage dips, short interruptions and voltage variations on power supply input linesIEC 61000-4-11 | 0% UT, 0.5 cycle g), At 0°, 45°, 90°, 135°, 180°, 225°, 270° and 315°0% UT, 1 cycle and 70% UT, 25/30 cycles Single phase at 0°0% UT, 250/300 cycle | 45°, 90°, 135°, 180°, 225°, 270° and 315°0% UT, 1 cycle and 70% UT, 25/30 cycles Single phase at 0°0% UT, 250/300 cycle | Mains power quality should be that of a typical commercial or home environment. If the user of the TheraFace Mask requires continued operation during power mains interruptions, it is recommended that the TheraFace Mask be powered from an uninterruptible power supply or a battery. |
| Power frequency (50/60 Hz magnetic fieldIEC 61000-4-8 | 30 A/m, 30 A/m Power frequency magnetic fields should be at level | characteristic of a typical location in a typical commercial or home environment. | |
| NOTE U1is the a.c. mains voltage prior to application of the test level. | |||
The TheraFace Mask is intended for use in the electromagnetic environment specified below. The customer or the user of the TheraFace Mask should assure that it is used in such an environment.
| IMMUNITY TESTIEC 60601 TEST LEVEL COMPLIANCE LEVEL ELECTROMAGNETIC ENVIRONMENT - GUIDANCE | |||
| Conducted RFIEC 61000-4-6 | 3 Vrms150kHz to 80MHz6 V in ISM and amateur radio bands between 0.15 MHz and 80 MHz | 3 Vrms150kHz to 80MHz6 V in ISM and amateur radio bands between 0.15 MHz and 80 MHz | Portable and mobile RF communications equipment should be used no closer to any part of the Thera Face Mask, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter.Recommended separation distance: \(d=\left[\frac{4.5}{V_1}\right]\sqrt{P} \)\(d=\left[\frac{12}{V_2}\right]\sqrt{P} \)\(d=\left[\frac{3.5}{F}\right]\sqrt{P} \)\(d=\left[\frac{2}{F}\right]\sqrt{P} \)80 MHz to 800 MHz 800 MHz to 27 GHz |
| Radiated RFIEC 61000-4-3 | 10V/m80MHz to 2.5GHz385MHz-5785MHz Test specifications for ENCLOSURE PORT IMMUNITY to RF wireless communication equipment (Refer to table 9 of IEC 60601-4-22014) | 10 V/m80MHz to 2.5GHz385MHz-5785MHz Test specifications for ENCLOSURE PORT IMMUNITY to RF wireless communication equipment (Refer to table 9 of IEC 60601-4-22014) | where p is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and d is the recommended separation distance in metres (m).Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey, should be less than the compliance level in each frequency range. Interference may occur in the vicinity of equipment marked with the following symbol:NOTE1 At 80 MHz and 800 MHz, the higher frequency range applies. NOTE 2 These guidelines may not apply in all situations. Electromagnetic is affected by absorption and refraction from structures, objects and people. |
The ISM (industrial scientific and medical) bands between 150 MHz and 80 MHz are 6.765 MHz to 6.795 MHz, 13.553 MHz to 13.567 MHz; 26.957 MHz to 27.263 MHz and 40.66 MHz to 40.70 MHz. The amate- radio bands between 0.15 MHz and 80 MHz are 1.8 MHz to
- Field strengths from fixed transmitters, such as base stations for radio (filarial/convex/telecommunications and land mobile radars), antenna to radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic
environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the TheraFace Mask is used exceeds the applicable RF compliance level above, the TheraFace Mask should be observed to
Vrrrnnn rnrnnn nnnnne eannnnnne aennnnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne
C. E = ( 12E) A + ( 12E) B + ( 12E) C
Rereomendseparatiosbetweeputableand mobileRFcommunlcationseepmentandtheEQUIPMENTorSYSTEM-forEQUIPMENTandSYTEM
Reommendationarion distance between roiable and mkiie RF rroscnlation unireent and the TheraFace Mark
| The TheraFace Mask is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the TheraFace Mask can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitter) and the TheraFace Mask as recommended below, according to the maximum output power of the communications equipment. | |||||
| Rated maximum output of transmitter W | Separation distance according to frequency of transmitter m | ||||
| 150 kHz to 80 MHz outside ISM and smateur radio bands d=[3.5/V1] √P | 150 kHz to 60 MHz in ISM and smateur radio bands d=[12/V2] √P | 80 MHz to 800 MHz d-[12/V2] √P | 80 MHz to 800 MHz d-[7/E1] √P | ||
| 0.01 | 0.20 | 0.035 | 0.07 | ||
| 0.1 | 0.30 | 0.33 | 0.10 | 0.21 | |
| 1 | 0.2 | 0.35 | 0.25 | 0.70 | |
| 10 | 3.8 | 6.32 | 1.10 | 2.21 | |
| 100 | 12 | 20.00 | 35 | 70 | |
For transmitters rated at a maximum output power not listed above the recommended separation distance d in metres ( m ) can be estimated using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer.
NOTE I At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies.
NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people.
Reporting adverse events to FDA
MedWatch is the Food and Drug Administration's (FDA) program for reporting serious reactions, product quality problems, therapeutic inequivalence/failure, and product use errors with human medical products, including drugs, biologic products, medical devices, dietary supplements, infant formula, and cosmetics.
If you think you or someone in your family has experienced a serious reaction to a medical product, you are encouraged to take the reporting form to your doctor. Your health care provider can provide clinical information based on your medical record that can help FDA evaluate your report.
However, we understand that for a variety of reasons, you may not wish to have the form filled out by your health care provider, or your health care provider may choose not to complete the form. Your health care provider is not required to report to the FDA. In these situations, you may complete the Online Reporting Form yourself.
You will receive an acknowledgement from FDA when your report is received. Reports are reviewed by FDA staff. You will be personally contacted only if we need additional information.
Submitting Adverse Event Reports to FDA
Use one of the methods below to submit voluntary adverse event reports to the FDA;
Report Online at www.accessdata.fda.gov/scripts/medwatch/index.cfm?action=reporting.home
Consumer Reporting Form FDA 3500B. Follow the instructions on the form to either fax or mail it in for submission. For help filling out the form, see MedWatchLearn. The form is available at
www.fdo.gov/downloads/aboutFDA/reportsmanualsforms/forms/ucm349464.pdf
Call FDA at 1-800-FDA-1088 to report by telephone.
- Reporting Form FDA 3500 commonly used by health professionals. The form is available at http://www.fda.gov/facts/health/162819.pdf
www.fdo.gov/download/adoa/bdout-DA/reportmanualistforms/forms/ucm183919.pdf
FCC Caution:
This device complies with Part 15 of the FCC Rules. Operation is subject to the following two conditions: (1) this device may not cause harmful interference, and (2) this device must accept any interference received, including interference that may cause undesired operation.
ES
Therabody Australia Pty. Ltd., The Commons South Yarra, 11 Wilson Street, South Yarra VIC 3141 australia@therabodycorp.com
Attn: Customer Service
6100 Wilshire Blvd. Ste 200
Los Angeles, CA 90048
Therabody Australia Pty. Ltd., The Commons South Yarra, 11 Wilson Street, South Yarra VIC 3141 australia@therabodycorp.com
Attn: Customer Service
6100 Wilsshire Blvd, Ste 200
Los Angeles, Ca. 90048

Therabody Australia Pty. Ltd., The Commons South Yarra, 11 Wilson Street, South Yarra VIC 3141 australia@therabodycorp.com
Therabody International Limited, Marine House, Clanwillian Place, Dublin, Ireland Therabody UK Limited, 55 Ludgate Hill, 2nd Floor, London EC4M7JW
Therabody Australia Pty. Ltd., The Commons South Yarra, 11 Wilson Street, South Yarra VIC 3141 australia@therabodycorp.com

Therabody Australia Pty. Ltd., The Commons South Yarra, 11 Wilson Street, South Yarra VIC 3141 australia@therabodycorp.com
www.fdo.gov/downloads/aboutFDA/reportsmanualsforms/forms/ucm349464.pdf
www.fdo.gov/downloads/aboutfdo/reportmanualsforms/forms/ucm163919.pdf
Precaución FCC:
Therabody Australia Pty. Ltd., The Commons South Yarra, 11 Wilson Street, South Yarra VIC 3141 australia@therabodycorp.com
MISES EN GARDE DE L'UNITE
Temperature, entreposage: -10 - 50 °C
www.fda.gov/downloads/aboutFDA/reportsmanualsforms/forms/ucm349464.pdf
Therabody International Limited, Marine House, Clanwilliam Place, Dublin, Ireland
Therabody UK Limited, 55 Ludgate Hill, 2nd Floor, London EC4M7JW
Therabody Australia Pty. Ltd., The Commons South Yarra, 11 Wilson Street, South Yarra VIC
3141
australia@therabodycorp.com
SC
装置警告
TeLaPteIeIaS MaStKJinTng TeLaPit
©2023 Therabody, Inc. All Rights Reserved.
默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默默
Therabody International Limited, Marine House, Clanwilliam Place, Dublin, Ireland
Therabody UK Limited, 55 Ludgate Hill, 2nd Floor, London EC4M7.JW
Therabody Australia Pty. Ltd., The Commons South Yarra, 11 Wilson Street, South Yarra VIC 3141
australia@therabodycorp.com
結果將會呈現
KR TaeLapoeAaMstAsKsIeLsAagHgLaCnTtJF,ChTg,ChTg,BeTcEgAeGtJeAnEnlAeJyJiAeJy.
本
1.
TheraFace Mask by Therabody
Born in Los Angeles, CA.
Designed for everybody.




@Therabody
 EasyManual
EasyManual 




