Blood Pressure Monitor - Smartwatch ARCHOS - Free user manual and instructions
Find the device manual for free Blood Pressure Monitor ARCHOS in PDF.

User questions about Blood Pressure Monitor ARCHOS
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Smartwatch in PDF format for free! Find your manual Blood Pressure Monitor - ARCHOS and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. Blood Pressure Monitor by ARCHOS.
USER MANUAL Blood Pressure Monitor ARCHOS
- Thank you very much for selecting ARCHOS Blood Pressure Monitor.
- Please do read the user manual carefully and thoroughly so as to ensure the safe usage of this product, and keep the manual well for your further reference in case you have problems.

Manuel_BPM_book.indd 106/06/2014 10:21:20
CATALOGUE
Table of Contents
INTRODUCTION 2
Safety Information
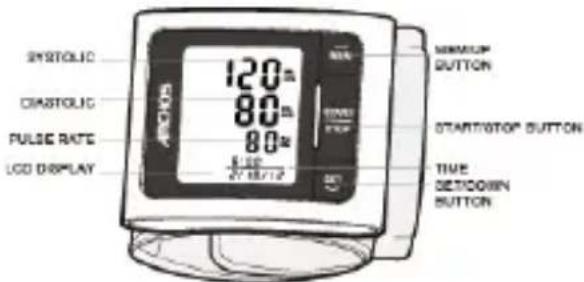
LCD Display Signal
Monitor Components
BEFORE YOU START 6
Installing and Replacing the Batteries
Setting Date and Time
MEASUREMENT 11
Tie the Cuff
Start Measurement
DATA MANAGEMENT. 13
Recalling the Records
Deleting the Records
INFORMATION FOR USER. 15
Tips for Measurement
Maintenance
ABOUT BLOOD PRESSURE. 17
What are systolic pressure and diastolic pressure?
What is the standard blood pressure classification?
Why does my blood pressure fluctuate throughout the day?
Why the blood pressure I get from the hospital is different from home?
If the result is the same if measuring on the right wrist?
TROUBLESHOOTING. 19
SPECIFICATIONS 20
CONTACT INFORMATION 21
COMPLIED EUROPEAN STANDARD LIST 21
EMC GUIDANCE 22
2
Manuel_BPM_book.indd 206/06/2014 10:21:20

EN
INTRODUCTION
General Description
Thank you for selecting ARCHOS Blood Pressure Monitor. The monitor features blood pressure measurement, pulse rate measurement and the result storage. The design provides you with two years of reliable service.
Reading taken by the Blood Pressure Monitor are equivalent to those obtained by a trained observer using the cuff and stethoscope auscultation method.
This manual contains important safety and care information, and provides step by step instructions for using the product.
Read the manual thoroughly before using the product.
FEATURES:
-
Systolic Blood Pressure
-
Diastolic Blood Pressure
-
Pulse Rate
-
Memory: Up to 60 pieces of records
Safety Information
The below signs might be in the user manual, labeling or other components. They are the requirement of standard and using.

3



| Symbol for “THE OPERATION GUIDE MUST BE READ” | Symbol for “MANUFACTURER” | ||
| CE0123 | Symbol for “COMPLIES WITH MDD93/42/ECC REQUIREMENTS” | EC REP | Symbol for “Authorized Representative in the European Community” |
| Symbol for “TYPE BF APPLIED PARTS” | Bluetooth | The Bluetooth Combination Mark | |
| SN | Symbol for “SERIAL NUMBER” Symbol | for “ENVIRONMENT PROTECTION – Waste electrical products should not be disposed of with household waste. Please recycle where facilities exist. Check with your local authority or retailer for recycling advice” | |
| = | Symbol for “DIRECT CURRENT” | ||
| Symbol for “MANUFACTURE DATE” | |||


CAUTION
Please do read this user manual carefully and thoroughly before use.
This device is intended for adult use only.
This device is intended for non-invasive measuring and monitoring of arterial blood pressure. It is not intended for use on extremities other than the wrist or for functions other than obtaining a blood pressure measurement.
Do not confuse self-monitoring with self-diagnosis. This unit allows you to monitor your blood pressure. Please start or end medical treatment basing solely on physician's treatment advice.
If you are taking medication, consult your physician to determine the most appropriate time for your measurement. Never change a prescribed medication without your physician's consent.

This unit is not suitable for continuous monitoring during medical emergencies or operations.
If the pressure of the cuff exceeds 40kPa (300 mmHg), the unit will automatically deflate. Should the cuff not deflate when its pressure exceeds 40kPa (300 mmHg), detach the cuff from the wrist and press the START/STOP button to stop inflation. Do not use the monitor under the conditions of strong electromagnetic field (e.g. medical RF equipment) that radiates interference signal or electrical fast transient / burst signal.
The maximum temperature that the applied part can be achieved is 42.5 日 while the environmental temperature is 40日 .
The device is not AP/APG equipment. It is not suitable for use in the presence of a flammable anesthetic mixture with air (or oxygen, nitrous oxide).
Please keep the unit out of reach of infants or children, since inhalation or swallowing of small parts is dangerous or even fata.
Please use ACCESSORIES and detachable parts specified / authorized by


MANUFACTURER. Otherwise, it may cause damage to the unit or danger to the user / patient.
Manufacturer will make available on request circuit diagrams, component parts listed.
Sensitive people, including pregnant women and those who implanted medical electronic
Instrument, should avoid using the unit whenever possible.
This unit is not suitable for continuous monitoring during medical emergencies or operations.
After the cuff inflated long time, the patient's wrist and fingers will is insufficient, anesthesia, distending pain and ecchymosis.
Please use the device under the environment which was provided in the user manual.
Otherwise, the performance and lifetime of the device will be impacted and reduced.
During using, the patient will contact with the cuff. The materials of the cuff have been tested and found to comply with requirements of ISO 10993-5:2009 and ISO
10993-10:2010. It will not cause any potential allergic reaction or contact injury.
The device has been evaluated clinically used manual cuff/ stethoscope auscultation as the reference.
The device doesn't need to be calibrated in two years of reliable service.
When the device was used to measure patients who have common arrhythmias such as atrial or ventricular premature beats or atrial fibrillation, the test result may occur deviation. Please consult your physician about the result.
This device is contraindicated for any female subject who may be suspected of, or is pregnant.
Besides provided inaccurate readings, the effects of this device on the fetus are unknown.
6
Manuel_BPM_book.indd 606/06/2014 10:21:20


7
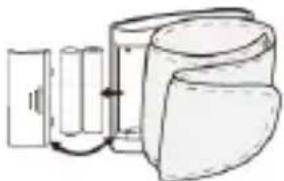
Monitor Components
Component List:
- PCBA; 2. Air Pipe; 3. Pump; 4. Valve; 5. Cuff.

List
- Wrist Blood Pressure Monitor 2. Two AAA-size batteries 3. User Manual
BEFORE YOU START
Installing and Replacing the Batteries
. Open the battery door.
-Insert the batteries according to the polarity indications. (Always select the authorized / specified battery: Two LR03 AAA-size batteries).
- .Close the battery door.
8
Manuel_BPM_book.indd 806/06/2014 10:21:21

EN
Battery Life: Approx. 57 days
(Battery capacity: 600 mAH. If measured 3 times per day, each measurement takes 30s, and memory checked once per day, each checking takes 60s. The current for measurement is 350 mA, and that for records display is 50 mA while the current when shutdown is 25 uA.)
Charging the power under following circumstances:
displays on the LCD
The LCD display dims
When powering on the monitor, the LCD doesn't light up.
CAUTION
*Removebatteriesifthedeviciso notlikyto be usedforsometime.
* Worn batteries are harmful to the environment. Do not dispose with daily garbage.
* Remove the old battery from the device following your local recycling guidelines.
*Do not dispose of batteries in fire. Batteries may explode or leak.
Setting Date and Time
Please proceed to time setting before your initial use so as to ensure each piece of record is labeled with a time stamp. (Year Range: 2012-2052; Time Format: 12 Hours) 1. When the monitor is OFF, press and hold "SET" button for 3 seconds to enter Time Setting Mode.

9
Manuel_BPM_book.indd 906/06/2014 10:21:21
- As pictured in the right, the blinking numeral "6" representing [HOUR]. Press "MEM" button to change the numeral. Each press will increase the numeral by one in a cycling manner.

- Press "SET" button again to confirm [HOUR]. Then the numeral representing [MINUTE] blinks.
- Repeat step 2 and 3 to confirm [MINUTE].


- Repeat step 2 and 3 to confirm [MONTH], [DAY] and [YEAR].
- After confirming [YEAR], the LCD will display "DONE" and the monitor will shut off.

10
Manuel_BPM_book.indd 10 06/06/2014 10:21:21

EN
Tie the Cuff
-
Remove all accessories (watch, bracelet, etc.) from your wrist. If your physician has diagnosed you with poor circulation in your wrist, use the other wrist.
-
Roll or push up your sleeve to expose the skin.
-
Apply the cuff to your wrist with your palm facing up.
-
Position the edge of the cuff about 1 - 1.5cm
-
Fasten the wrist cuff around your wrist, leaving no extra room between the cuff and your skin. If the cuff is too loose, the measurement will not be accurate.
Resting for 5 minutes before measuring.
Wait at least 3 minutes between measurements. This allows your blood circulation to recover.
For a meaningful comparison, try to measure under similar conditions. For example, take daily measurements at approximately the same time, on the same wrist, or as directed by a physician.


Pair-up the Blood Pressure Monitor with Your Device
- Turn on Bluetooth and the app. Make sure both are ON when pair-up is proceeding.
- When the monitor is OFF, press and hold the START button for 2 seconds to start pair-up. The symbol and the symbol will be shown on the LCD alternatively, indicating pair-up is proceeding.
If SUCEED, symbol will be shown on the LCD.
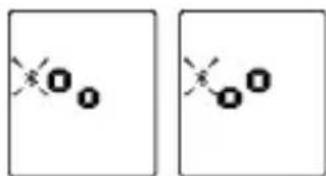
11


If FAIL, symbol will be shown on the LCD.
- The monitor will shut off automatically after Pair-up process is complete.
MEASUREMENT
Start Measurement
- After correctly positioning the cuff, press START button to turn on the monitor, and it will complete the measurement process automatically. Adjust to zero.



Inflating and measuring.
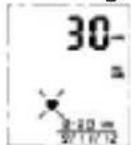
12
Manuel_BPM_book.indd 12 06/06/2014 10:21:21

The mobile or portable equipment, which are equipped with Bluetooth function in line with BLE Technical Specifications as well as BLP Protocol established by global organization Bluetooth SIG, are capable to receive the health data, including measurement time, systolic pressure, diastolic pressure, etc.
DATA MANAGEMENT
Recalling the Records
- Press "MEM" button to access the memory.
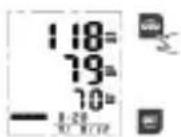
- Press "MEM/UP" button or "SET/DOWN" button to rotate the history records. "MEM/UP" to go forward; "SET/DOWN" to go backward.

Deleting the Records
When you did not obtain the accurate measurement, you can clear all the measuring results by following below steps.
- Under Memory Recalling Mode, press and hold both the "MEM" button and the "SET" button for 3 seconds.
- The LCD will display "dEL dONE", indicating that memory clearing is complete.
- If you wish to give up clearing, press "START/STOP" to turn off the monitor.
- When there is no memory in the monitor, if you press the "MEM" button to look up
History, the LCD will display as pictured to the right.

INFORMATION FOR USER
Tips for Measurement
It can cause inaccuracy if the measurement is taken in the following circumstances.

Maintenance
To obtain the best performance, please follow below instructions.


Cleaning: Dust environment may affect the performance of the unit. Please use the soft cloth to remove the dirt before use.
Please make sure the unit functions safely and it is in proper working conditions before use.
Please follow the instructions for correct replacement of interchangeable or detachable parts specified by SERVICE PERSONNEL of MANUFACTURER as "Replaceable".
Disposal: Degraded sensors or loosened electrodes may degrade the unit's performance or even cause other problems. Please dispose of ACCESSORIES, detachable parts, and the ME EQUIPMENT according to the local guidelines.
ABOUT BLOOD PRESSURE
What are systolic pressure and diastolic pressure?
16
Manuel_BPM_book.indd 16 06/06/2014 10:21:22

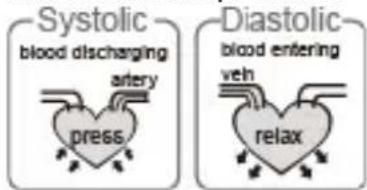
EN
When ventricles contract and pump blood out of the heart, the blood pressure reaches its maximum value in the cycle, which is called systolic pressure. When the ventricles relax, the blood pressure reaches its minimum value in the cycle, which is called diastolic pressure.
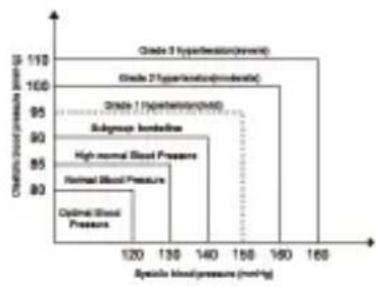
What is the standard blood pressure classification? The blood pressure classification published by World Health Organization (WHO) and International Society of Hypertension (ISH) in 1999 is as follows:
CAUTION Only a physician can tell your normal BP range. Please contact a physician if your measuring result falls out of the range. Kindly note that only a physician could tell whether your blood pressure value has reached a dangerous point.
17
Manuel_BPM_book.indd 17 06/06/2014 10:21:22
| Level BP (mm Hg) | Optimal Normal Hypertension | |||||
| G1 G2 G3 | ||||||
| SYS <120 121-130 131-140 141-160 161-180 ≥180 | ||||||
| DIA <80 81-85 86-90 91-100 | 101-110 ≥110 | |||||
Irregular Heartbeat Detector
This Wrist Blood Pressure Monitor is equipped with an intelligent function of Irregular Heartbeat (IHB) Detector. During each measurement, this equipment records the heartbeat intervals and works out the standard deviation. If the heartbeat intervals compare with the average intervals, the deviation more than 3 is over 25% or more than 5 is over 15% , this equipment will light up the IHB symbol on the screen when displaying the measuring result.
CAUTION
The appearance of the IHP icon indicates that a pulse irregularity is consistent with an irregular heartbeat was detected during measurement. Usually this is NOT a cause for concern. However, if the symbol appears often, we recommend you seek medical advice. Please note that the device does not replace a cardiac examination, but serves to detect pulse irregularities at an early stage.
Why does my blood pressure fluctuate throughout the day?
- Individual blood pressure varies every in one day, it also affected by the way you t i.e. your cuff and your measurement position, so please take the measurement at the same condition.
- The varies of the pressure is greater if the person take medicine.
- Waiting at least 3 minutes for another measurement.

Why the blood pressure I get from the hospital is different from home?
The blood pressure is different even during 24 hour because of the weather, emotion, exercise etc., especially the "white coat" in hospital which makes the results are higher than the ones at home.
If the result is the same if measuring on the right wrist?
It is ok for both wrists, but there will be some different results for different person, so suggest you measure the same wrist every time.

The attention need to pay when you measure you blood pressure at home:
If the cuff is tied properly.
If the cuff is too tight or too loose.
If the cuff is tied on the wrist.
If you feel anxious pressured.
You had better take deep breath 2-3 times before beginning.
19
Advice: adjust yourself for 4-5 minutes until you calm down.
TROUBLESHOOTING
This section includes a list of error messages and frequently asked questions for problems you may encounter with your wrist blood pressure monitor. If the products not operating as you think it should, check here before arranging for servicing.
| PROBLEM SYMPTOM CHECK THIS REMEDY | |||
| No power Display will not light up. | Batteries are exhausted. Replace with new batteries | ||
| Batteries are inserted incorrectly. | Insert the batteries correctly | ||
| Low batteries | Display is dim or Display | Batteries are low. Replace with new batteries | |
| Error massage | * shows | Data communication has failed | Check if the APP is on or not, try data transmission again. |
| Error 1 shows Inflation is slow or the cuff is not secure. | Refasten the cuff and then measure again. | ||
| Error 2 shows The cuff is very tight Readjust the cuff, not too loose or too tight and then measure again. | |||
| Error 3 shows The pressure of the cuff is excess. | Refasten the cuff and then measure again. | ||
20
Manuel_BPM_book.indd 20 06/06/2014 10:21:22



EN
| Error massage | Error 5 or Error 6 shows System error occurred. | Retake the measurement. If the problem persists, contact the retailer or our customer service department for further assistance. Refer to the warranty for contact information and return instructions. |
| Error 10 or Error 11 shows | The monitor detected motion, talking or the pulse is too poor while measuring. Relax for a moment and then measure again. |

| Error massage | Error 20 shows The measu | rement process does not detect the pulse signal. | Loosen the clothing on the wrist and then measure again. |
| Error 21shows on the display. | The treatment of the measurement failed. | Relax for a moment and then measure again. |

SPECIFICATIONS
| Power supply 2*AAA batteries | |
| Display mode Digital LCD V.A.36x41mm |
21




| Measurement mode Oscillographic testing mode | |
| Measurement range Pressure:0kpa-40kpa 0mmHg-pulse value:(40-199)beat/minute | 300mmHg pulse value:(40-199)beat/minute |
| Accuracy Pressure: | 5-40 within±0.4kpa(3mmHg)0-45 (out of 5-40)within±0.7kpa(5mmHg) pulse value:±5% |
| Normal working condition Temperature:5-40 | |
| Relative humidity ≤80% Atmospheric pressure: 86 kPa to 106kPa | |
| Storage & transportation condition Temperature:-20 to 60 Relative Humidity: 10% to 93% RH | |
| Measurement perimeter of the wrist About 13.5cm-21.5cm | |
| Net Weight Approx.120g(Excluding the dry cells) | |
| External dimensions Approx.80×65×22mm | |
| Attachment 2*AAA batteries, user manual | |
| Mode of operation Continuous operation |
22

EN
| Degree of protection Type BF applied part | |
| Protection against ingress of water IP22 | |
| Software version V01 | |
| Device classification Internally Powered ME Equipment |
WARNING: No modification of this equipment is allowed.
CONTACT INFORMATION
Contact Information
For more information about our products, please visit www.archos.com you can get customer service, usual problems and customer download, ARCHOS will serve you anytime.
Manufactured by: GUANGDONG TRANSTEK MEDICAL ELECTRONICS CO., LTD
Company: GUANGDONG TRANSTEK MEDICAL ELECTRONICS CO., LTD
Address: Zone A, 5/F., Investment Building, No. 12, Huizhan East Rd., Torch
Development District, Zhongshan, Guangdong, 528437, China
Authorized European Representative:
Company: MDSS - Medical Device Safety Service GmbH
Address: Schifflgraben 41, 30175 Hannover, Germany
23
Complied European Standards List
Table 1 – Guidance and MANUFACTURER'S declaration – ELECTROMAGNETIC EMISSIONS – for all ME EQUIPMENT and ME SYSTEM
| Risk Management EN/ISO 14971:2007 | |
| Labeling EN 15223:2012 | |
| User Manual EN 1041:2008 | |
| General Requirements for Safety | EN 60601-1:2006/AC2010 |
| EN 60601-1-11:2010 | |
| Non-invasive Sphygmomanometers General Requirements | EN 1060-1:1995+A2:2009EN 1060-3:1997+A2:2009EN 1060-4:2004 |
| Electromagnetic Compatibility | EN 60601-1-2:2007/AC:2010 |
| Software Lifetime EN 62304:2006/AC:2008 | |
| Usability EN 60601-1-6:2010 | |
EMC Guidance
24

Table 2 - Guidance and MANUFACTURER'S declaration - electromagnetic IMMUNITY - for all ME EQUIPMENT and ME SYSTEMS
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that it is used in such an environment. | |||
| IMMUNITY test IEC 60601 test level Compliance level Electromagnetic environment - guidance | |||
| Electrostatic discharge (ESD)IEC 61000-4-2 | ±6 kV contact±8 kV air | ±6 kV contact±8 kV air | Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30%. |
| Electrical fast transient / burstIEC 61000-4-4 | ±2 kV for power supply lines±1 kV for input / output lines | Not applicable Mains | power quality should be that of a typical commercial or hospital environment. |
| SurgeIEC 61000-4-5 | ±1 kV line(s) to line(s)±2 kV line(s) to earth | Not applicable Mains | power quality should be that of a typical commercial or hospital environment. |


EN
| Voltage dips, short interruptions and voltage variations on power supply input lines IEC 61000-4-11 | ☑5% UT (☑95% dip in UT) for 0.5 cycle 40% UT (60% dip in UT) for 5 cycles 70% UT (30% dip in UT) for 25 cycles ☑5% UT (☑95% dip in UT) for 5 s | Not applicable Mains | power quality should be that of a typical commercial or hospital environment. If the user of device requires continued operation during power mains interruptions, it is recommended that device be powered from an interruptible power supply or a battery. |
| Power frequency (50/60 Hz) magnetic field IEC 61000-4-8 | 3 A/m 3 A/m Power frequency | magnetic fields should be at levels characteristic of a typical location in a typical commercial or hospital environment | |
| NOTE UT is the a.c. mains voltage prior to application of the test level. | |||

27
Manuel_BPM_book.indd 27 06/06/2014 10:21:22




Table 3 - Guidance and MANUFACTURER'S declaration - electromagnetic IMMUNITY - for ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that it is used in such an environment. | |||
| IMMUNITY test | IEC 60601 test level | Compliance level | Electromagnetic environment - guidance |
| Portable and mobile RF communications equipment should be used no closer to any part of the device, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter. | |||
| Portable and mobile RF communications equipment should be used no closer to any part of the device, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter. Recommended separation distance | |||

| Conducted RFIEC 61000-4-62.333 | 3 Vrms150 kHz to 80MHz | Not applicable | d=[3.5/Vt]√F d=[3.5/Vt]√F d-[1/2]√F = MHz-2.5 GHz d-[7/2]√F 400 MHz-2.5 GHz d=[1/2]√F 400 MHz-2.5 GHzwhere P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and d is the recommended separation distance in meters (m). Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey, a should be less than the compliance level in each frequency range. b Interference may occur in the vicinity of equipment marked with the following symbol: |
| Radiated RFIEC 61000-4-3 | 3 V/m80 MHz to 2.5GHz | 3V/m | |
| NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies.NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||
| Field strengths from fixed transmitters, such as base stations for radio (cellular / cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the device is used exceeds the applicable RF compliance level above, the device should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as re-orienting or relocating the device. b Over the frequency range 150 kHz to 80 MHz, field strengths should be less than[V1] V/m. | |||

Table 4 - Recommended separation distances between portable and mobile RF communications equipment and the ME EQUIPMENT or ME SYSTEM - for ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
| Recommended separation distances between portable and mobile RF communications equipment at the device The device is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the device can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and the device as recommended below, according to the maximum output power of the communications equipment. | |||
| Rated maximum output power of transmitter (W) | Separation distance according to frequency of transmitter (m) | ||
| 150 kHz to 80 MHzd= [3.5/V]√T d= [3.5/V]√T | 80 MHz to 800 MHzd= [3.5/V]√T d= [3.5/V]√T | 800 MHz to 2.5 GHzd= [3.5/V]√T d= [3.5/V]√T | |
| 0.01 Not applicable 0.1 | 7 0.233 | ||
| 0.1 Not applicable 0.369 | 0.738 | ||
| 1 Not applicable 1.167 | 2.333 | ||
| 10 Not applicable 3.690 | 7.378 | ||
| 100 Not applicable 11.67 | 23.33 | ||

C∈0123
Version:1.0
ARCHOS

Adresse: Zone A, 5/F., Investment Building, No. 12, Huizhan East Rd., Torch
Development District, Zhongshan, Guangdong, 528437, China
Adresse: Zone A, 5/F., Investment Building, No. 12, Huizhan East Rd., Torch
Development District, Zhongshan, Guangdong, 528437, China

Dirección: Zone A, 5/F., Investment Building, No. 12, Huizhan East Rd., Torch
Development District, Zhongshan, Guangdong, 528437, China
Development District, Zhongshan, Guangdong, 528437, Cina





