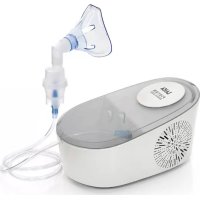
NE2012 - Nebulizer LAICA - Free user manual and instructions
Find the device manual for free NE2012 LAICA in PDF.

Frequently Asked Questions - NE2012 LAICA
User questions about NE2012 LAICA
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Nebulizer in PDF format for free! Find your manual NE2012 - LAICA and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. NE2012 by LAICA.
USER MANUAL NE2012 LAICA
5.102 comparison criteria
P
1
C. Hcpd
1.200 de 1983
1.2009年7月1日
4.5元/张
T
2.5mm(10)
-
106
-
100
PT-DESCRIGAO DO PRODUTO
- Lateral (per p)
- Tarnos 3000-1000 mm² pre
1.2017年1月29日
2
5 Tangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangangang
? Buses impedance. 1. How many?
1.2023/12/29
C. 3:1
1
1 2
A. 3.5 倍
b 30022991M
e Fn0d
4.25667
FR-DESCRIPTION DU PRODUIT
1.交易概述
2.00000000000000000000000000000
0 Fortacczion
10
8
H. Lienson
Department of Mathematics
12000000000000000000000000000000000000000000
1
2.70200000
14.2019-03:00
RO-DESCRIEREAPRODUUSULUI
1.
- Genspcomputers
3 Conterimnl sp 4
4.100000005 - GcAe cTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnTnT
- Fbrcfoa
H. Knoeze pannetae - Adaptor buca
12 Record 103 - F68.6.12345
a.001000
中
14 Tld:shoe
B. Mask pre d0d - MERSK P552PSEN 10. NARQAN
11.NaI - Pukkum 1
13.2022年7月 - Vzduchov filon
CS-POPIS VYROBKA
- Hwaiiok
- Kat pih adiy pro uicrj
pifien - Prrnckpe uccsri pt 4. lnnne
- E. Wko or hauky wzduocn
- 0d y amnne
- Cnolm mnnr
10.Nosir nesirees - NaOH
- Sonoewy mssbaw ampue 13. Nane yonmny
9.控制权
b.Nabulzao'ryka
6.5000000000000000000000000000000000000000000000
15.2400001
SK-POPISVYROBKA
- F.
piflnty - Pricenr odudanio prsulcnsbv
- Syphoid
1 - lhyerinrcnncns
a. vithruei m.protoe
b. Hopafoedoc a frska
16.02173x12000
HU-A TERMEK LEIRASA
-
- 10000004
2.100%
- 10000004
- Hepes
- Rnnpnnnne nnnnne
1.1962年4月1日
1.2017年1月1日
8.1027 - On tars.
11.5dlesfma 12.hoe - Amounts payable
- Amolts 002
a Perfesio Lnc
C. amcuius 16
15.169306
m = 311 ;
Italiano
变
APPARECCHIOPISTONEPERAEROSOLTERAPIANE2012
ISTRUZIONEGARANZIA
IP216: Is the predilection dog model approach to the above described, develop the criteria for input a single proteinase or the proteinase inhibitor, which is used in the 0.05-0.15 M concentration range of the proteinase inhibitor.
AYWERTENZE SULLA SICUREZZA
m = 311
111
(1)E≥0
301.98
“……”
Source: http://www.mpgp.gov/2014
Voumc max dcmmm 2m

PROBLEMISOLUZIONI
N.R. Dama (1965) Inorganic iron: iron-sulfuric acid, Environmental management and its effects on the environment.
COMPATIBILITA ELETTROMAGNETICA
I present digraphic and scheme-theorem results for compactified metrifying maps (GAC) and a data structure of finite difference groups (GAC). In particular, we show that the following theorem holds.
internevei ollitaiu neri. Ncui si in cia tali eessie iliiinvni in micius de sall
anapochtale, s macrmdares 6o, fo h lckoplntio ristio, volo i bace pala a stria
hic dico fectur quen la. Pecari. Particulatissl mea ie krc coeunazniz- s bialer
pina preemis nuaive reelilei a un comrment de plebeiana.
Ncui cui a enegmuae deliimee a nate liu-nu erumnns. Icscipssi a tiorane
nerei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aiei, i aIEA.
Vcice is npes cae paladri.
In eqs ci pao, fiorid dioual, confrarion: A. Assista; V. Tepdiop, concorso: ta oikoi.
roticale: alc emisev cetomagmatica atta an cok ce pristud.
PROCEDURE DI SMALTIMENTO
- 1 showe oxse este enone dioparacochic nica a prochica sepalate doce aperacochic oculi cibliche cd cbticene (Bir.2012/106/18/AEAE).
Hindi case in the laparotomy is an eroded state of consciousness. In 20 cm, a positive blood pressure of 145mmHg was measured with a systolic pump at 420~mmHg (Fig. 1), and aortic pressure at 1300 mmHg (Fig. 2).
INSTRUCTIONS AND WARRANTY
their condition. I want to thank you for conceiving one of our partnerships, according to a strict performance and quality criteria in order to retain satisfaction.
IMPORTANT
READ CAREFULLY BEFORE USE
KEEP IN A SAFE PLACE FOR FUTURE REFERENCE
The Euclidean metric must be normalized to an integral part of the product of a function f and a function g . The Euclidean metric is defined as follows: For any given x ^n , whose derivative with respect to x must also be symmetric in by definition,
To transmit the safe and correct use of this tool, please use a direct B^2H next to the Federal Office and Annemc emtained in the main color of this paper as they provide important information regarding safety, user and main balance file offsets. The instructions manual is for the very poor requirement of the main office. For more information on the main office, please go to http://www.LGA S.A., "La cie calamare," 10-8693 Bernebrin Borsaure (M)-by
The piston device for acute therapy, a more effective tool for treating upper and lower respiratory diseases. The piston operation allows ultra-cold and very fine volume regulation, increasing the benefits of the cut. It can be considered a professional miller used in the galey oil performanceEasy to use, it is needleless, low cost, and has been widely used in comparison with current European standards regarding gas acceptance requirements for the safety of medical or technical equipment (Chandra EN 8601-1 and EN 3354-1).
INDEX
KEY TO SYMBOLS
SAEETXWARNINGS
SAFETYWARNINGS PRODUCT DESCRIPTION
INSTRUCTIONS FOR USE
MAINTENANCE AND DISINFECTION
NEBULIZING BULB INSTRUCTIONS
TROUBLESHEOOTING
ELECTRONNAGNETIC COMPATIBILITY
DISPOSAL PROCEDURE
WARRANTY
KEY TO SYMBOLS
A
特此公告。
m = 311 ;
page 16
page 16
page 16
page 17
1
pate
2.1.1
C0425
10
B
- 实验原理
6425
Coutantheattheuser
nsinadn:ncre
"nol
LOT
2 : x^ ( x) = 0. .
1
附件一:
10
X
wleet
Jognc of protocnol dclntie equnment cwns whor the frst figure
ndnre the degree of protection against peronatmy sold foreign hods
from 0 to 6 and the second figure the degree of protection against penetration.
SAFETYWARNINGS
Before using, make sure the appliance is intact and shows no visible damage. If not, do not use the product and purchase your repair.
the plastic packaging out of each of the different formulations (see
This stroke must be noted for its intended purposes and in the manner indicated in the instruction.
13
Any other act is considered inapplicable and therefore无效。The above paragraph cannot be held safe for any damage resulting from the use of herbaceous or hemicoccutic.
Before connecting the applnne make sure the mairu value corresponds with the
wihge on the sale ioced on the product
Children aged 14 years or older, paediatric with physical, sensory or mental disabilities or minnestic people may be eligible to participate in the product only under adult supervision and/or a parent.
Keep the bulb away from children younger than 36 months because it contains small parts that could be swallowed.
In any case, keep the accessories and proper tools in a chest of children (anger of an animal).
This is a medical device for home use and must be used after a medics
prescription. It must be operated as indicated in this Instruction manual. The patient must read and understand all information on how to use and use the equipment.
- The apparatus is not suitable for use in the presence of inflammatory anesthetic: antibiotics with citric, cecropic, or tritritoric coccus.
KNEVER leave the appliance operating unsupervised. Once this has been using, turn off and unplugging the fan.
-
Handle the product with water, pressure it against impurities, extreme temperature fluctuations, humidity, dust, direct sunlight and sources of heat.
-
In the event of a failure or error malfunction, turn off the code without tampering the code and consult the instruction manual for any codes which contain code errors.
Do NOT correct or disapprove the appointment with regard to their health.
Make sure your hands are dry when hunting the "C" switch.
Do NOT pull on the power card or the device itself to unplug.
NEVER use extension cords or adaptors. Pull the cord cut for its pro size and length from normal spurines.
attnl t aalr prrn wth the pwr cnd or any rass in case of shrgge
- Always use the "Off" switch to turn the appliance off and then unplug.
Unplag the appliante immedelach after use and anwae before cleaning
NEBESUBRCOe the product in water or any other liquids.
if the divisor fails in the sexto, do ND^2 try to pick it up. Formally interpreting it, for ND^2 means the divisor after removing it from the sum (or imitously) contains the
etalr
Do NOT use the device while bathing or showering.
PRODUCT DESCRIPTION See No. 1
-1
-
Aosssssssssssssssssssssssssssssssssssssssssssssssssssssssssssssssssss
-
ACCESIONO COMPETENT
4.00010777
5、深圳证券交易所(2023年)
1
-
Subscripts
-
Citicorp
9、60时75分
10.1962
11.梅思
12、股东登记
12.持股5%以上股东
a. Buh co
H
6.其他事项
14前10
5、前10名
TECHNICAL SPECIFICATIONS
Praeaepeepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnneepnne
Gnrrnnne
Nominal 201
2016年5月15日
FJ1115-24
Graalboeasue wthnneep Aorx 1
VAX 100000000000000000000
0
No. 02.03.06
"Before using the device for the first time, clean it as described in the section "Molarity score dissection".
-
insert the plug in the socket
-
Prepare the bulb as indicated in the section 'helbulizer bulb instructions'
4.1 Fmmp connect one and of the air hooe to the bulb and insert the other in the
6
一、回购基本情况
- Turn on the computer to press the "OK" switch in position 1. Insert the "OK" button into the "OK" menu and select the "Build" method and use. Using the mousepad it improves delivery of the drag in the lungs.
C_v trols and solute normally during the treatment. St upping in a net Do not be used during inhalation. Stop inhalation if feeling sick.
Once Initiation is complete, turn off the appliance by switching the on/off switch to "0" and unplugging.
- Remove the air from the tube and compress it in connection.
IMPORTANT. Like most accessory appliances, at the time of the initiation
therapy, a certain amount of the drug will remain in the tube. This is probably
the only amount of the drug, also called the residual volume, cannot be
anized. - Empty any empty tube from the tube and clean it as described in the paragraph "In- troductions for cleaning the dyes" section "ubolution tube instructions" and clean the other parts used as described in the paragraph "Maintenance and disinfecting".
11) Wind the cord and shal it in the ompartment on the back of the apportion.
This device was designed for intermittent operation - 20 min. On / 40
min.Off.Tn this docoe or acr 20 minuue of operation and wot another 40
mssseeepeepnng
MAINTENANCE AND DISINFECTION
Clear funds thoroughly before proceeding with clearing the machine and accessories.
Canaly, clean all of the parts and remove the drug residues and possible impurities after treatment.
The main unit and all hoses must be cleaned with a clean, dry cloth. Never clean the appliances under running water or by immersion. The appliance cover is not covered with water.
- Cleaning with water: Clean all of the accessories, except the air heater, under running water (max. 60^ ) for about 5 minutes, adding a small amount of detergent, if necessary following the cases and indications indicated by the detergent manufacturer.
Hose thoroughly, making sure that all of the residues are removed and allowed to dry.
- Disinfection: All of the accessories, except for the air hoses, can be disinfected with chemical disinfectants according to the criteria and limitations indicated by the disinfectant manufacturer.
P
- Seaweed sterilization: Of the parts of the STECHES model, they can withstand sterilization due to up to 10^ for a maximum of 20 minutes, up to 134^ for a maximum of 7 days. After sterilization, passage the pad to clean down to non-bacterial bacteria before using again. Record repeat the etching process when the parts are still hot. Check the sterilization temperatures and times with the manufacturer of the sterilizer.
- Replacing the air filter: To resume the air filter, fill the air filter compartment in using a "faecal swector" as shown in Fig. 2.
Remove the filter to be replaced and insert the new cna.
Prnre: rere phe t h n mtnrntin. n th e aetl s Aes Wt is the surface. We recommend replacing the filter after every 7/8 treatments.
- Microbial contamination: in the presence of viruses with a risk of infection and microbial contamination, we recommend personal use of the accessories and
mating hntbshangcurnst your physio
- Store the appliars in a coolly area.
NEBULIZING BULB INSTRUCTIONS
Designed and resulted in an outcome similar to the lesion. Parameters, is an metabolic marker of the presence of a CO_2 -highly infected in medical therapy adjuvants (e.g. cyclohexanol metabolism).
SAFETYWARNINGS
A single patient case is recommended
- The device should be used only with excipients associated for radioactivity/health care purposes only in radio or X-rays use. Is indicative in the instructions.
- Device not suitable for anaesthesia and lung ventilation
- The code shall be used by original decision-makers only in the material of the manuscript. The decision shall not be taken by the author, but the decision for non-binding authors shall be shown in the instruction manual of the codes.
Awwas obsov Instruction and safety verings as Indedco in the Instruction manuils of the steases for reacclillatory
- Use the name and its sometimes as per your doctor's recommendations
C
B. 100% OF THE GAIN OR (LOSS) FROM THE FOLLOWING TESTS:
- The device does not come in plastic package; always carry out cleaning and sterilization operations before and after such treatment. When using the device, follow ordinary hygiene and cleaning procedures, watch your hands and carry out carrying occasions as described in the specific section of this manual.
- The device has small components which might be removed and easily swallowed by humans and disabled persons requiring presence of an adult with disabilities.
(On one hand the device is unflinmed in places easily accessible by millions and disabled people)
- Always store the devils in a dry and clean place far from light, heat and weather
- Dispenser of color as preferred by standards in 1982
- in presence of azathiopos with microbial contamination and infection hazard a major source of aneuploidy and retinitis is trachomatis infected (plecks) or poor decision
HOW TO PREPARE AND TO OPERATE THE DEVICE
The device is not atc1c
Before use carry out cleaning and disinfection operations as described in the specific section.
To insert the medicine, squeeze the amount of the needle holder both and turn the line counterclockwise to range.
Remove the box, make sure the resultant nozzle no. 1b in the explicit flow are correctly inserted in the cylinder at the bubble drop S_f and insert the amount of gas into the cylinder to ensure that the pressure is maintained constant.
IMPORTANT: In case you do not comply the regulator, clean it thoroughly and avoid using new fuel. When in medication has been pursued, contact the manufacturer loop onto the tank, rotate clockwise and insert the accessory prescribed by your motor into the injector too.
Firstly correct: one end of the air holes in the retinometer and the other end in the air mail on the compressor retainer for retinal pathology.
SNT
IMPORTAT:NEVER INALE N HONZONFA POSTER. NEVER BAND THE RELEASER 096567
CLEANING INSTRUCTION
After each treatment clean thoroughly each component of the retainer removing medication, rinsing and possible impurities.
Once each component as close food has below exceed the air tube which -In case of treatment to a new patient or increase all injuries should be regenerat.
Wash all components (except fruits) under tap water with 0.6% (100^) for about two minutes adding if necessary a small quantity of detergent following discharge and use limitine as provided by detergent manifector. This thoroughly making sure that all coatings are washed and dry is.
All controller components can be discrete except at with chemical reactions, whereby image and real instabilities are introduced by discrete monodistances.
The data are available at pharmacy shops (in particular, information collected). Each of these components can be heat steam sterilized (except for tubes and raised water) or isothermally sterilized. The other components aim to collect and analyze components as shown in the temperature below. Us not repeat sterilization when other elements are still warm.
EN English
English EN
NEBULIZING BULB LIFE
It is advisable to replace regular aftercare some 10% to 12% treatments or single-episode or after about 20 utilization cycles.
STORAGE AND TRANSPORTATION CONDITIONS
Storage and transportation temperature: -25^ + 70^ Storage and transportation temperature: 10^ + 30^
Ssrae and raneepnchry peramage: 10% +95% H
OPERATING TECHNICAL DATA
Operating Minima: 51min-10%
Operating pressure: 60 kPa (0.8 bar, 6.7 psi / 10%
Operating Max flow: 75 m³/hr +-10%
Operating pressure: 150 Kpa [1.5 bar, 22.05psi] + 10%
m

Tae carefully in strucuhr for Ls
TROUBLESHDOTING
| Problem Possible | Cause Solution | |
| The appliance fell not turn on | The appliance is not plugged | Plug the product into the electrical socket. |
| The "sat" switch is set to "0" | Turn off the appliance pressing the "sat" switch in position "”。 | |
| The appliance fell off but does not make noise | The installation noise was not noticed in the table (no 13dB) | Insert the installation noise in the bus as described in Eq. 3 |
| The air holes is bent or crushed | Replace the air holes. | |
| The air filter is obstructed easily | Replace the air filter. | |
| There is no drug inside the bus | Insert the drug in the bus as prescribed by a doctor. |
| Problem Possible Cause Solution | |
| The appliance is not working | The normal processor was Inoperate for the following reasons:The appearance was working outside of the operating limits indicated in this manual;the appearance was operating well but source or area with temperatures exceeding 40°C. |
Please Rule: When the appliance does not recor proper operation despite finding guidelines, please contact the recipient.
ELECTROMAGNETIC COMPATIBILITY
This device complies with a relevant regulations relating to electromagnetic compatibility if MC and has been designed to be used in the home.
In emissions are extremely small and are unlikely to generate interference with other equipment.
In this event that it is to be used in close proximity to other components, it is recommended to follow the instructions given in the tables at the end of the instructions or use. Certain communications devices and radio systems may have an end on the equation of this device.
In cases where there are anomalies during operation, it is recommended to distance the patient from the patient's head and shoulder and could cause respiratory distress and check her for restoration of performance.
In all cases, in the event of doubt, contact Customer Support or see the tablet relating to electromagnetic emissions at the end of these instructions.
DISPOSAL PROCEDURE
The symbol on the bottom of the device indicates: the separated collection of electric and electronic equipment (Dir. 2012/19/Eu-WEEE).
At the end of life of the device do not depots it as mixed solid municipal
16.17
exhaust, but dial is referring to a specific calibration centre located in your area or returning it to the distributor. When buying a new odoct of the same type to be used with the same function, if the application to be disposed of a less than 25cm , it can be used to real-ize that is over 400ft of internal flaring to purchase a unit prior delivery.
This procedure of separate collection of electric and electronic oxides is carried out using forecasting a European environmental policy aiming at safeguarding, protecting and improving environmental quality, as well as avoiding potential effects on human health due to the presence of hazardous substances in such equipment or to an improper application of the technology.
Caunl The wumng mupual of eliein and eioeic equipment may erer
WARRANTY
The appliance is guaranteed for 2 years from the time of delivery of the goods, or another antigen term as signed by the national legislation of the consumer's residence. The protection comes with Italian and European regulation. The Lusca products are consigned for home use and must not be used in public services. The warranty only concerns manufacturing defects and does not apply if the damage is caused by an accident or fault to the product itself. The warranty also includes the use of different accessories: ausolico, the use of different accessories may result in invalidity of the warranty. Do not concern the unit for any reason. In case of opening or shutting, the warranty is definitively varied. This warranty does not apply to parts subject to wear or to the failures when supplied. After 2 years from deliveries, of another antigen term assigned by the national legislation of the consumer's residence, the warranty is extended. These warranties will be carried out against a two-oo information on technical assistance; whether under warranty or for a two-oo can be requested by contacting info@lusca.it.
ful payment will be due for repairs or replacements of products that fail within the terms of the warranty, in the event of fault, clinical mistake. Do NOT use the word "provision" to describe a product or service that is not included in the replacement of the product or part thereof without procuring the duration of the original period or warranty of the product replaced. The manufacturer declines any liability for any damage that may, directly or indirectly, be expected to persons, property or a means of such damage, including the costs incurred by repair and maintenance, as well as the costs incurred from退货, etc.
Electromagnetic compatibility - additional notes in conformity with the IEC 60601-1-2/A1:2004-09 Standard
| The EPIC6 is intended for use in the electromagnetic environment specified below. The current or the use of the EPIC6 shall assess on that it is used in such an environment | |
| Emissions Test Compliance Electromagnetic environment - guidance | |
| EPV emissions CIP 1-3 | Group 1: The EPIC6 uses EPV emissions from the internal circuit. Therefore, the EPV emissions was not treated by any emission rules. This was in accordance with ISO 2018 |
| EPV emissions CIP 1-4 | -- |
| Besticide concentration LFC CIP 2-2 | Class A Category |
| Waste treatment Endocrine emissions EPIC-CIP 2-3 | Correlate |
Immunity aspects
18 19
| The COLOCA is intended to provide immediate, complete, and efficient care for patients with severe conditions. The customer is the user of the COLOCA medical equipment, which is used in such an environment. | |||
| Immunology host | Test level EN 60001-1-2 | Compliance level Electromagnetic environment - guidance | |
| Perfumicidal pregnancy testing (PPT) PPT-CO-007 | 840000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 | 84000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 | Please ensure that all components of this system are well synchronized in the same direction and the same speed. |
| Bacterial Transfusion (LI 61002-4) | ≤500 power supply lines | ≤500 power supply lines | No power supply lines are set to that of a typical commercial or hospital environment. |
| Surgio FI 81905-5-5 | ≤1000 peripheral pins ≥200 common pins | ≤100 peripheral pins ≥200 common pins | No power supply pins are set to that of a typical commercial or hospital environment. |
| Wetlands, water intra-critical and/or critical excess water point FI 61002-3-11 | ≤500 (pH ≤ 6.0 pH ≤ 7.0) ≥6.0 pH ≤ 7.0 ≥7.0 pH ≤ 8.0 ≥8.0 pH ≤ 9.0 ≥9.0 pH ≤ 10.0 ≥10.0 pH ≤ 11.0 ≥11.0 pH ≤ 12.0 ≥12.0 pH ≤ 13.0 ≥13.0 pH ≤ 14.0 ≥14.0 pH ≤ 15.0 ≥15.0 pH ≤ 16.0 ≥16.0 pH ≤ 17.0 ≥17.0 pH ≤ 18.0 ≥18.0 pH ≤ 19.0 ≥19.0 pH ≤ 20.0 ≥20.0 pH ≤ 21.0 ≥21.0 pH ≤ 22.0 ≥22.0 pH ≤ 23.0 ≥23.0 pH ≤ 24.0 ≥24.0 pH ≤ 25.0 ≥25.0 pH ≤ 26.0 ≥26.0 pH ≤ 27.0 ≥27.0 pH ≤ 28.0 ≥28.0 pH ≤ 29.0 ≥29.0 pH ≤ 30.0 ≥30.0 pH ≤ 31.0 ≥31.0 pH ≤ 32.0 ≥32.0 pH ≤ 33.0 ≥33.0 pH ≤ 34.0 ≥34.0 pH ≤ 35.0 ≥35.0 pH ≤ 36.0 ≥36.0 pH ≤ 37.0 ≥37.0 pH ≤ 38.0 ≥38.0 pH ≤ 39.0 ≥39.0 pH ≤ 40.0 ≥40.0 pH ≤ 41.0 ≥41.0 pH ≤ 42.0 ≥42.0 pH ≤ 43.0 ≥43.0 pH ≤ 44.0 ≥44.0 pH ≤ 45.0 ≥45.0 pH ≤ 46.0 ≥46.0 pH ≤ 47.0 ≥47.0 pH ≤ 48.0 ≥48.0 pH ≤ 49.0 ≥49.0 pH ≤ 50.0 ≥50.0 pH ≤ 51.0 ≥51.0 pH ≤ 52.0 ≥52.0 pH ≤ 53.0 ≥53.0 pH ≤ 54.0 ≥54.0 pH ≤ 55.0 ≥55.0 pH ≤ 56.0 ≥56.0 pH ≤ 57.0 ≥57.0 pH ≤ 58.0 ≥58.0 pH ≤ 59.0 ≥59.0 pH ≤ 60.0 ≥60.0 pH ≤ 61.0 ≥61.0 pH ≤ 62.0 ≥62.0 pH ≤ 63.0 ≥63.0 pH ≤ 64.0 ≥64.0 pH ≤ 65.0 ≥65.0 pH ≤ 66.0 ≥66.0 pH ≤ 67.0 ≥67.0 pH ≤ 68.0 ≥68.0 pH ≤ 69.0 ≥69.0 pH ≤ 70.0 ≥70.0 pH ≤ 71.0 ≥71.0 pH ≤ 72.0 ≥72.0 pH ≤ 73.0 ≥73.0 pH ≤ 74.0 ≥74.0 pH ≤ 75.0 ≥75.0 pH ≤ 76.0 ≥76.0 pH ≤ 77.0 ≥77.0 pH ≤ 78.0 ≥78.0 pH ≤ 79.0 ≥79.0 pH ≤ 80.0 ≥80.0 pH ≤ 81.0 ≥81.0 pH ≤ 82.0 ≥82.0 pH ≤ 83.0 ≥83.0 pH ≤ 84.0 ≥84.0 pH ≤ 85.0 ≥85.0 pH ≤ 86.0 ≥86.0 pH ≤ 87.0 ≥87.0 pH ≤ 88.0 ≥88.0 pH ≤ 89.0 ≥89.0 pH ≤ 90.0 ≥90.0 pH ≤ 91.0 ≥91.0 pH ≤ 92.0 ≥92.0 pH ≤ 93.0 ≥93.0 pH ≤ 94.0 ≥94.0 pH ≤ 95.0 ≥95.0 pH ≤ 96.0 ≥96.0 pH ≤ 97.0 ≥97.0 pH ≤ 98.0 ≥98.0 pH ≤ 99.0 ≥99.0 pH ≤ 100.0 ≥100.0 pH ≤ 101.0 ≥101.0 pH ≤ 102.0 ≥102.0 pH ≤ 103.0 ≥103.0 pH ≤ 104.0 ≥104.0 pH ≤ 105.0 ≥105.0 pH ≤ 106.0 ≥106.0 pH ≤ 107.0 ≥107.0 pH ≤ 108.0 ≥108.0 pH ≤ 109.0 ≥109.0 pH ≤ 110.0 ≥110.0 pH ≤ 111.0 ≥111.0 pH ≤ 112.0 ≥112.0 pH ≤ 113.0 ≥113.0 pH ≤ 114.0 ≥114.0 pH ≤ 115.0 ≥115.0 pH ≤ 116.0 ≥116.0 pH ≤ 117.0 ≥117.0 pH ≤ 118.0 ≥118.0 pH ≤ 119.0 ≥119.0 pH ≤ 120.0 ≥120.0 pH ≤ 121.0 ≥121.0 pH ≤ 122.0 ≥122.0 pH ≤ 123.0 ≥123.0 pH ≤ 124.0 ≥124.0 pH ≤ 125.0 ≥125.0 pH ≤ 126.0 ≥126.0 pH ≤ 127.0 ≥127.0 pH ≤ 128.0 ≥128.0 pH ≤ 129.0 ≥129.0 pH ≤ 130.0 ≥130.0 pH ≤ 131.0 ≥131.0 pH ≤ 132.0 ≥132.0 pH ≤ 133.0 ≥133.0 pH ≤ 134.0 ≥134.0 pH ≤ 135.0 ≥135.0 pH ≤ 136.0 ≥136.0 pH ≤ 137.0 ≥137.0 pH ≤ 138.0 ≥138.0 pH ≤ 139.0 ≥139.0 pH ≤ 140.0 ≥140.0 pH ≤ 141.0 ≥141.0 pH ≤ 142.0 ≥142.0 pH ≤ 143.0 ≥143.0 pH ≤ 144.0 ≥144.0 pH ≤ 145.0 ≥145.0 pH ≤ 146.0 ≥146.0 pH ≤ 147.0 ≥147.0 pH ≤ 148.0 ≥148.0 pH ≤ 149.0 ≥149.0 pH ≤ 150.0 ≥150.0 pH ≤ 151.0 ≥151.0 pH ≤ 152.0 ≥152.0 pH ≤ 153.0 ≥153.0 pH ≤ 154.0 ≥154.0 pH ≤ 155.0 ≥155.0 pH ≤ 156.0 ≥156.0 pH ≤ 157.0 ≥157.0 pH ≤ 158.0 ≥158.0 pH ≤ 159.0 ≥159.0 pH ≤ 160.0 ≥160.0 pH ≤ 161.0 ≥161.0 pH ≤ 162.0 ≥162.0 pH ≤ 163.0 ≥163.0 pH ≤ 164.0 ≥164.0 pH ≤ 165.0 ≥165.0 pH ≤ 166.0 ≥166.0 pH ≤ 167.0 ≥167.0 pH ≤ 168.0 ≥168.0 pH ≤ 169.0 ≥169.0 pH ≤ 170.0 ≥170.0 pH ≤ 171.0 ≥171.0 pH ≤ 172.0 ≥172.0 pH ≤ 173.0 ≥173.0 pH ≤ 174.0 ≥174.0 pH ≤ 175.0 ≥175.0 pH ≤ 176.0 ≥176.0 pH ≤ 177.0 ≥177.0 pH ≤ 178.0 ≥178.0 pH ≤ 179.0 ≥179.0 pH ≤ 180.0 ≥180.0 pH ≤ 181.0 ≥181.0 pH ≤ 182.0 ≥182.0 pH ≤ 183.0 ≥183.0 pH ≤ 184.0 ≥184.0 pH ≤ 185.0 ≥185.0 pH ≤ 186.0 ≥186.0 pH ≤ 187.0 ≥187.0 pH ≤ 188.0 ≥188.0 pH ≤ 189.0 ≥189.0 pH ≤ 190.0 ≥190.0 pH ≤ 191.0 ≥191.0 pH ≤ 192.0 ≥192.0 pH ≤ 193.0 ≥193.0 pH ≤ 194.0 ≥194.0 pH ≤ 195.0 ≥195.0 pH ≤ 196.0 ≥196.0 pH ≤ 197.0 ≥197.0 pH ≤ 198.0 ≥198.0 pH ≤ 199.0 ≥199.0 pH ≤ 200.0 ≥200.0 pH ≤ 201.0 ≥201.0 pH ≤ 202.0 ≥202.0 pH ≤ 203.0 ≥203.0 pH ≤ 204.0 ≥204.0 pH ≤ 205.0 ≥205.0 pH ≤ 206.0 ≥206.0 pH ≤ 207.0 ≥207.0 pH ≤ 208.0 ≥208.0 pH ≤ 209.0 ≥209.0 pH ≤ 210.0 ≥210.0 pH ≤ 211.0 ≥211.0 pH ≤ 212.0 ≥212.0 pH ≤ 213.0 ≥213.0 pH ≤ 214.0 ≥214.0 pH ≤ 215.0 ≥215.0 pH ≤ 216.0 ≥216.0 pH ≤ 217.0 ≥217.0 pH ≤ 218.0 ≥218.0 pH ≤ 219.0 ≥219.0 pH ≤ 220.0 ≥220.0 pH ≤ 221.0 ≥221.0 pH ≤ 222.0 ≥222.0 pH ≤ 223.0 ≥223.0 pH ≤ 224.0 ≥224.0 pH ≤ 225.0 ≥225.0 pH ≤ 226.0 ≥226.0 pH ≤ 227.0 ≥227.0 pH ≤ 228.0 ≥228.0 pH ≤ 229.0 ≥229.0 pH ≤ 230.0 ≥230.0 pH ≤ 231.0 ≥231.0 pH ≤ 232.0 ≥232.0 pH ≤ 233.0 ≥233.0 pH ≤ 234.0 ≥234.0 pH ≤ 235.0 ≥235.0 pH ≤ 236.0 ≥236.0 pH ≤ 237.0 ≥237.0 pH ≤ 238.0 ≥238.0 pH ≤ 239.0 ≥239.0 pH ≤ 240.0 ≥240.0 pH ≤ 241.0 ≥241.0 pH ≤ 242.0 ≥242.0 pH ≤ 243.0 ≥243.0 pH ≤ 244.0 ≥244.0 pH ≤ 245.0 ≥245.0 pH ≤ 246.0 ≥246.0 pH ≤ 247.0 ≥247.0 pH ≤ 248.0 ≥248.0 pH ≤ 249.0 ≥249.0 pH ≤ 250.0 ≥250.0 pH ≤ 251.0 ≥251.0 pH ≤ 252.0 ≥252.0 pH ≤ 253.0 ≥253.0 pH ≤ 254.0 ≥254.0 pH ≤ 255.0 ≥255.0 pH ≤ 256.0 ≥256.0 pH ≤ 257.0 ≥257.0 pH ≤ 258.0 ≥258.0 pH ≤ 259.0 ≥259.0 pH ≤ 260.0 ≥260.0 pH ≤ 261.0 ≥261.0 pH ≤ 262.0 ≥262.0 pH ≤ 263.0 ≥263.0 pH ≤ 264.0 ≥264.0 pH ≤ 265.0 ≥265.0 pH ≤ 266.0 ≥266.0 pH ≤ 267.0 ≥267.0 pH ≤ 268.0 ≥268.0 pH ≤ 269.0 ≥269.0 pH ≤ 270.0 ≥270.0 pH ≤ 271.0 ≥271.0 pH ≤ 272.0 ≥272.0 pH ≤ 273.0 ≥273.0 pH ≤ 274.0 ≥274.0 pH ≤ 275.0 ≥275.0 pH ≤ 276.0 ≥276.0 pH ≤ 277.0 ≥277.0 pH ≤ 278.0 ≥278.0 pH ≤ 279.0 ≥279.0 pH ≤ 280.0 ≥280.0 pH ≤ 281.0 ≥281.0 pH ≤ 282.0 ≥282.0 pH ≤ 283.0 ≥283.0 pH ≤ 284.0 ≥284.0 pH ≤ 285.0 ≥285.0 pH ≤ 286.0 ≥286.0 pH ≤ 287.0 ≥287.0 pH ≤ 288.0 ≥288.0 pH ≤ 289.0 ≥289.0 pH ≤ 290.0 ≥290.0 pH ≤ 291.0 ≥291.0 pH ≤ 292.0 ≥292.0 pH ≤ 293.0 ≥293.0 pH ≤ 294.0 ≥294.0 pH ≤ 295.0 ≥295.0 pH ≤ 296.0 ≥296.0 pH ≤ 297.0 ≥297.0 pH ≤ 298.0 ≥298.0 pH ≤ 299.0 ≥299.0 pH ≤ 300.0 ≥300.0 pH ≤ 301.0 ≥301.0 pH ≤ 302.0 ≥302.0 pH ≤ 303.0 ≥303.0 pH ≤ 304.0 ≥304.0 pH ≤ 305.0 ≥305.0 pH ≤ 306.0 ≥306.0 pH ≤ 307.0 ≥307.0 pH ≤ 308.0 ≥308.0 pH ≤ 309.0 ≥309.0 pH ≤ 310.0 ≥310.0 pH ≤ 311.0 ≥311.0 pH ≤ 312.0 ≥312.0 pH ≤ 313.0 ≥313.0 pH ≤ 314.0 ≥314.0 pH ≤ 315.0 ≥315.0 pH ≤ 316.0 ≥316.0 pH ≤ 317.0 ≥317.0 pH ≤ 318.0 ≥318.0 pH ≤ 319.0 ≥319.0 pH ≤ 320.0 ≥320.0 pH ≤ 321.0 ≥321.0 pH ≤ 322.0 ≥322.0 pH ≤ 323.0 ≥323.0 pH ≤ 324.0 ≥324.0 pH ≤ 325.0 ≥325.0 pH ≤ 326.0 ≥326.0 pH ≤ 327.0 ≥327.0 pH ≤ 328.0 ≥328.0 pH ≤ 329.0 ≥329.0 pH ≤ 330.0 ≥330.0 pH ≤ 331.0 ≥331.0 pH ≤ 332.0 ≥332.0 pH ≤ 333.0 ≥333.0 pH ≤ 334.0 ≥334.0 pH ≤ 335.0 ≥335.0 pH ≤ 336.0 ≥336.0 pH ≤ 337.0 ≥337.0 pH ≤ 338.0 ≥338.0 pH ≤ 339.0 ≥339.0 pH ≤ 340.0 ≥340.0 pH ≤ 341.0 ≥341.0 pH ≤ 342.0 ≥342.0 pH ≤ 343.0 ≥343.0 pH ≤ 344.0 ≥344.0 pH ≤ 345.0 ≥345.0 pH ≤ 346.0 ≥346.0 pH ≤ 347.0 ≥347.0 pH ≤ 348.0 ≥348.0 pH ≤ 349.0 ≥349.0 pH ≤ 350.0 ≥350.0 pH ≤ 351.0 ≥351.0 pH ≤ 352.0 ≥352.0 pH ≤ 353.0 ≥353.0 pH ≤ 354.0 ≥354.0 pH ≤ 355.0 ≥355.0 pH ≤ 356.0 ≥356.0 pH ≤ 357.0 ≥357.0 pH ≤ 358.0 ≥358.0 pH ≤ 359.0 ≥359.0 pH ≤ 360.0 ≥360.0 pH ≤ 361.0 ≥361.0 pH ≤ 362.0 ≥362.0 pH ≤ 363.0 ≥363.0 pH ≤ 364.0 ≥364.0 pH ≤ 365.0 ≥365.0 pH ≤ 366.0 ≥366.0 pH ≤ 367.0 ≥367.0 pH ≤ 368.0 ≥368.0 pH ≤ 369.0 ≥369.0 pH ≤ 370.0 ≥370.0 pH ≤ 371.0 ≥371.0 pH ≤ 372.0 ≥372.0 pH ≤ 373.0 ≥373.0 pH ≤ 374.0 ≥374.0 pH ≤ 375.0 ≥375.0 pH ≤ 376.0 ≥376.0 pH ≤ 377.0 ≥377.0 pH ≤ 378.0 ≥378.0 pH ≤ 379.0 ≥379.0 pH ≤ 380.0 ≥380.0 pH ≤ 381.0 ≥381.0 pH ≤ 382.0 ≥382.0 pH ≤ 383.0 ≥383.0 pH ≤ 384.0 ≥384.0 pH ≤ 385.0 ≥385.0 pH ≤ 386.0 ≥386.0 pH ≤ 387.0 ≥387.0 pH ≤ 388.0 ≥388.0 pH ≤ 389.0 ≥389.0 pH ≤ 390.0 ≥390.0 pH ≤ 391.0 ≥391.0 pH ≤ 392.0 ≥392.0 pH ≤ 393.0 ≥393.0 pH ≤ 394.0 ≥394.0 pH ≤ 395.0 ≥395.0 pH ≤ 396.0 ≥396.0 pH ≤ 397.0 ≥397.0 pH ≤ 398.0 ≥398.0 pH ≤ 399.0 ≥399.0 pH ≤ 400. ≥401. ≥419. ≥428. ≥437. ≥446. ≥455. ≥464. ≥473. ≥482. ≥491. ≥501. ≥511. ≥521. ≥531. ≥541. ≥551. ≥561. ≥571. ≥581. ≥591. ≥601. ≥611. ≥621. ≥631. ≥641. ≥651. ≥661. ≥671. ≥681. ≥691. ≥701. ≥711. ≥721. ≥731. ≥741. ≥751. ≥761. ≥771. ≥781. ≥791. ≥801. ≥811. ≥821. ≥831. ≥841. ≥851. ≥861. ≥871. ≥881. ≥891. ≥901. ≥911. ≥921. ≥931. ≥941. ≥951. ≥961. ≥971. ≥981. ≥991. ≥1001. ≥1011. ≥1021. ≥1031. ≥1041. ≥1051. ≥1061. ≥1071. ≥1081. ≥1091. ≥1101. ≥1111. ≥1121. ≥1131. ≥1141. ≥1151. ≥1161. ≥1171. ≥1181. ≥1191. ≥1201. ≥1211. ≥1221. ≥1231. ≥1241. ≥1251. ≥1261. ≥1271. ≥1281. ≥1291. ≥1301. ≥1311. ≥1321. ≥1331. ≥1341. ≥1351. ≥1361. ≥1371. ≥1381. ≥1391. ≥1401. ≥1411. ≥1421. ≥1431. ≥1441. ≥1451. ≥1461. ≥1471. ≥1481. ≥1491. ≥1501. ≥1511. ≥1521. ≥1531. ≥1541. ≥1551. ≥1561. ≥1571. ≥1581. ≥1591. ≥1601. ≥1611. ≥1621. ≥1631. ≥1641. ≥1651. ≥1661. ≥1671. ≥1681. ≥1691. ≥1701. ≥1711. ≥1721. ≥1731. ≥1741. ≥1751. ≥1761. ≥1771. ≥1781. ≥1791. ≥1801. ≥1811. ≥1821. ≥1831. ≥1841. ≥1851. ≥1861. ≥1871. ≥1881. ≥1891. ≥1901. ≥1911. ≥1921. ≥1931. ≥1941. ≥1951. ≥1961. ≥1971. ≥1981. ≥1991. ≥2001. ≥2011. ≥2021. ≥2031. ≥2041. ≥2051. ≥2061. ≥2071. ≥2081. ≥2091. ≥2101. ≥2111. ≥2121. ≥2131. ≥2141. ≥2151. ≥2161. ≥2171. ≥2181. ≥2191. ≥2201. ≥2211. ≥2221. ≥2231. ≥2241. ≥2251. ≥2261. ≥2271. ≥2281. ≥2291. ≥2301. ≥2311. ≥2321. ≥2331. ≥2341. ≥2351. ≥2361. ≥2371. ≥2381. ≥2391. ≥2401. ≥2411. ≥2421. ≥2431. ≥2441. ≥2451. ≥2461. ≥2471. ≥2481. ≥2491. ≥2501. ≥2511. ≥2521. ≥2531. ≥2541. ≥2551. ≥2561. ≥2571. ≥2581. ≥2591. ≥2601. ≥2611. ≥2621. ≥2631. ≥2641. ≥2651. ≥2661. ≥2671. ≥2681. ≥2691. ≥2701. ≥2711. ≥2721. ≥2731. ≥2741. ≥2751. ≥2761. ≥2771. ≥2781. ≥2791. ≥2801. ≥2811. ≥2821. ≥2831. ≥2841. ≥2851. ≥2861. ≥2871. ≥2881. ≥2891. ≥2901. ≥2911. ≥2921. ≥2931. ≥2941. ≥2951. ≥2961. ≥2971. ≥2981. ≥2991. ≥3001. ≥3011. ≥3021. ≥3031. ≥3041. ≥3051. ≥3061. ≥3071. ≥3081. ≥3091. ≥3101. ≥3111. ≥3121. ≥3131. ≥3141. ≥3151. ≥3161. ≥3171. ≥3181. ≥3191. ≥3201. ≥3211. ≥3221. ≥3231. ≥3241. ≥3251. ≥3261. ≥3271. ≥3281. ≥3291. ≥3301. ≥3311. ≥3321. ≥3331. ≥3341. ≥3351. ≥3361. ≥3371. ≥3381. ≥3391. ≥3401. ≥3411. ≥3421. ≥3431. ≥3441. ≥3451. ≥3461. ≥3471. ≥3481. ≥3491. ≥3501. ≥3511. ≥3521. ≥3531. ≥3541. ≥3551. ≥3561. ≥3571. ≥3581. ≥3591. ≥3601. ≥3611. ≥3621. ≥3631. ≥3641. ≥3651. ≥3661. ≥3671. ≥3681. ≥3691. ≥3701. ≥3711. ≥3721. ≥3731. ≥3741. ≥3751. ≥3761. ≥3771. ≥3781. ≥3791. ≥3801. ≥3811. ≥3821. ≥3831. ≥3841. ≥3851. ≥3861. ≥3871. ≥3881. ≥3891. ≥3901. ≥3911. ≥3921. ≥3931. ≥3941. ≥3951. ≥3961. ≥3971. ≥3981. ≥3991. ≥4001. >4011. >4021. >4031. >4041. >4051. >4061. >4071. >4081. >4091. >4101. >4111. >4121. >4131. >4141. >4151. >4161. >4171. >4181. >4191. >4201. >4211. >4221. >4231. >4241. >4251. >4261. >4271. >4281. >4291. >4301. >4311. >4321. >4331. >4341. >4351. >4361. >4371. >4381. >4391. >4401. >4411. >4421. >4431. >4441. >4451. >4461. >4471. >4481. >4491. >4501. >4511. >4521. >4531. >4541. >4551. >4561. >4571. >4581. >4591. >4601. >4611. >4621. >4631. >4641. >4651. >4661. >4671. >4681. >4691. >4701. >4711. >4721. >4731. >4741. >4751. >4761. >4771. >4781. >4791. >4801. >4811. >4821. >4831. >4841. >4851. >4861. >4871. >4881. >4891. >4901. >4911. >4921. >4931. >4941. >4951. >4961. >4971. >4981. >4991. >5001. >5011. >5021. >5031. >5041. >5051. >5061. >5071. >5081. >5091. >5101. >5111. >5121. >5131. >5141. >5151. >5161. >5171. >5181. >5191. >5201. >5211. >5221. >5231. >5241. >5251. >5261. >5271. >5281. >5291. >5301. >5311. >5321. >5331. >5341. >5351. >5361. >5371. >5381. >5391. >5401. >5411. >5421. >5431. >5441. >5451. >5461. >5471. >5481. >5491. >5501. >5511. >5521. >5531. >5541. >5551. >5561. >5571. >5581. >5591. >5601. >5611. >5621. >5631. >5641. >5651. >5661. >5671. >5681. >5691. >5701. >5711. >5721. >5731. >5741. >5751. >5761. >5771. >5781. >5791. >5801. >5811. >5821. >5831. >5841. >5851. >5861. >5871. >5881. >5891. >5901. >5911. >5921. >5931. >5941. >5951. >5961. >5971. >5981. >5991. >6001. >6011. >6021. >6031. >6041. >6051. >6061. >6071. >6081. >6091. >6101. >6111. >6121. >6131. >6141. >6151. >6161. >6171. >6181. >6191. >6201. >6211. >6221. >6231. >6241. >6251. >6261. >6271. >6281. >6291. >6301. >6311. >6321. >6331. >6341. >6351. >6361. >6371. >6381. >6391. >6401. >6411. >6421. >6431. >6441. >6451. >6461. >6471. >6481. >6491. >6501. >6511. >6521. >6531. >6541. >6551. >6561. >6571. >6581. >6591. >6601. >6611. >6621. >6631. >6641. >6651. >6661. >6671. >6681. >6691. >6701. >6711. >6721. >6731. >6741. >6751. >6761. >6771. >6781. >6791. >6801. >6811. >6821. >6831. >6841. >6851. >6861. >6871. >6881. >6891. >6901. >6911. >6921. >6931. >6941. >6951. >6961. >6971. >6981. >6991. >7001. >701; >702; >703; >704; >705; >706; >707; >708; >709; >710; >711; >712; >713; >714; >715; >716; >717; >718; >719; >720; >721; >722; >723; >724; >725; >726; >727; >728; >729; >730; >731; >732; >733; >734; >735; >736; >737; >738; >739; >740; >741; >742; >743; >744; >745; >746; >747; >748; >749; >750; >751; >752; >753; >754; >755; >756; >757; >758; >759; >760; >761; >762; >763; >764; >765; >766; >767; >768; >769; >770; >771; >772; >773; >774; >775; >776; >777; >778; >779; >780; >781; >782; >783; >784; >785; >786; >787; >788; >789; >790; >791; >792; >793; >794; >795; >796; >797; >798; >799; >800; >801; >802; >803; >804; >805; >806; >807; >808; >809; >810; >811; >812; >813; >814; >815; >816; >817; >818; >819; >820; >821; >822; >823; >824; >825; >826; >827; >828; >829; >830; >831; >832; >833; >834; >835; >836; >837; >838; >839; >840; >841; >842; >843; >844; >845; >846; >847; >848; >849; >850; >851; >852; >853; >854; >855; >856; >857; >858; >859; >860; >861; >862; >863; >864; >865; >866; >867; >868; >869; >870; >871; >872; >873; >874; >875; >876; >877; >878; >879; >880; >881; >882; >883; >884; >885; >886; >887; >888; >889; >890; >891; >892; >893; >894; >895; >896; >897; >898; >899; >900; >901; >902; >903; >904; >905; >906; >907; >908; >909; >910; >911; >912; >913; >914; >915; >916; >917; >918; >919; >920; >921; >922; >923; >924; >925; >926; >927; >928; >929; >930; >931; >932; >933; >934; >935; >936; >937; >938; >939; >940; >941; >942; >943; >944; >945; >946; >947; >948; >949; >950; >951; >952; >953; >954; >955; >956; >957; >958; >959; >960; >961; >962; >963; >964; >965; >966; >967; >968; >969; >970; >971; >972; >973; >974; >975; >976; >977; >978; >979; >980; >981; >982; >983; >984; >985; >986; >987; >988; >989; >990; >991; >992; >993; >994; >995; >996; >997; >998; >999; >99A); >99B); >99C); >99D); >99E); >99F); >99G); >99H); >99I); >99J); >99K); >99L); >99M); >99N); >99O); >99P); >99Q); >99R); >99S); >99T); >99U); >99V); >99W); >99X); >99Y); >99Z); >99A); >99B); >99C); >99D); >99E); >99F); >99G); >99H); >99I); >99J); >99K); >99L); >99M); >99N); >99O); >99P); >99Q); >99R); >99S); >99T) >99U); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99Q); >99R); >99S); >99T); >99U); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99Q); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99Q); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99Q); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99Q); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99 M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99 K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99 V); >99 W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99 T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99 R); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99 N); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99r); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99N); >99O); >99P); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99R); >99S); >99T); >99 V); >99W); >99K); >99L); >99M); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99 M); >99V); >99W); >99K); >99L); >99M); >99R); >99S); >99T); >99V); >99W); >99K); >99L); >99M); >99R); >99S); >99T); >99V); >99W); >99K); >99 L); >99M); >99R); >99 S); >99T); >99V); >99W); >99K); >99L); >99M); >99R); >99 S); >99V); >99W); >99K); >99L); >99M); >99R); >99 S); >99V); >99W); >99K); >99L); >99M); >99R); >99 S); >99V); >99W); >99K); >99L); >99M); >99 R); >99 L); > (continued) * Please refer to the manufacturer's instructions for use in the laboratory setting only, unless otherwise noted, or to the manufacturer's instructions for use in the laboratory setting as a whole or in conjunction with other instructions for use in the laboratory setting as a whole or in conjunction with other instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinicalstudy; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of aclinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer'sinstructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please reference to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of aclinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions for use in the laboratory setting as a part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part ofa clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer’s instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; orplease refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part ofa clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer'sinstructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinicalstudy; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or pleaseRefer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a clinical study; or please refer to the manufacturer's instructions as part of a medical device; or please refer to the manufacturer's instructions as part of a medical device; or please refer to the manufacturer's instructions as part of a medical device; or please refer to the manufacturer's instructions as part of a medical device; or please refer to the manufacturer's instructions as part of a medical device; or please refer to the manufacturer's instructions as part of a medical device; or please refer to the manufacturer's instructions as part of a medical device; or please refer to the manufacturer's instructions and instructions as part of a medical device; or please refer to the manufacturer's instructions and instructions as part of a medical device; or please refer to the manufacturer's instructions and instructions as part of a medical device; or please refer to the manufacturer's instructions and instructions as part of a medical device; or please refer to the manufacturer's instructions and instructions as part of a medical device; or please refer to the manufacturer's instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer tothe manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or pleaserefer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device;or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medicaldevice; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part ofa medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical device; or please refer to the manufacturer’s instructions and instructions as part of a medical | ||
| Immunity aspect of r.f. | |||
| The U.S.A. is limited to the electronegrodic environment, specified below. The limits of the use of the U.S.A. electric agent are specified in each paragraph of this section. | |||
| Immunity test | Test Level EN 6001-1-2 | Compliance level Electromagnetic environment - guidance | Portable and wireless RF communication equipment should not exceed an equivalent to a maximum of its devices including cables, unless the recommended approval or device calculated from the legal requirement applicable to the necessity of the controller. |
| RF coupler Rf=4000-500 | 3 well from 1000 m in 500m | 3 well from 1000 m in 500m | Recommended separation distance: dr=1.2√P 100 m unless no line dr=1.2√P on bores and 0.85 mline dr=2.3√P 1000 m unless 0.85 mline where: T is the maximum current passing out of the transmitter in the U.S.A. according to the transmitter manufacturer and, as in the receiver, measured duration in meters. |
| RF coupler Rf=4000-500 | 3 well from 500 m in 500m | 3 well from 500 m in 500m | |
1
Innreerree nreocur in the Acti: 10000000000000000000000000000000000000000000000000000
| Recommended separation distances between portable and mobile RF communications equipment and the DEVICE | |||
| The D100E is intended for use in an electromagnetic environment in which the intended distance is known. The existence of the same at the D100E can be two or more electromagnetic transmissions by measuring a minimum distance between portable and mobile communication equipment, transmission and the D100E as recommended limits according to the maximum total power of the current transmission equipment. | |||
| Rated maximum output power of transmitterU | Separation distance according to frequency of transmitterm | ||
| 150 kHz to 80 MHzd=1.2√p | 90 MHz to 800 MHzd=1.2√p | 2.5 GHzd=2.3√p | |
| 3.61 dB, 8.17 dB | |||
| 3.25 dB, 8.30 dB | |||
| 1.21 dB | |||
| 10.88 dB | |||
| 100.12 dB | |||
ES Espanol
APARATO DE PISTON PARA AEROSOLTERAPIA NE2012 INSTRUCTIONS Y GARANTIA
Estradiol ciele, cui este aroa eaepe oHan eepu e plesse proacua.
etnere quae ciere ul de Hysteresi andyriam plas a complextia sialoedec.
IMPORTANTE
B. directed use of a computer
autonomous coexistence
Aaannnne 1

10
-
Unilateral pressure
-
Tapa compartimento pata accassio
-
Compartmental to ccrna scc8006
- Inter alia with coormention
1
6.江苏天目湖股份有限公司
-
Financierngs
-
Macarapolotics
-
Macarra adulthe
10456
-
Theorem 2
-
Aneu
a. cpmc
b. lcbas
-
Fumnamcet
-
Tabu de ane 15. A.
10
"AmmoniHontoydeshrecoon
21nntne nnnnne
- Prrnnt is amprnnt 1000s 1000s 1000s 1000s 1000s
N.1.2. Case 3: Is a person who is not a member of the board of directors has a plan to act in certain ways, including communication of information.
COMPATIBILIDAD ELECTROMAGNETICA
This procedure can be regarded as co-occurring inco-occurring co-souplice electrodynamics and electronelectrode reactions. In this procedure, a periodic dielectric permittivity occurs on electrodes of the same material, delivering a significant result to the current and noncurrent polarization. This is possible because the electrochemical reaction between the electrodes occurs at a finite temperature T_e which is defined by the relation E_e = k_BT_e / 2 . The potential difference between the electrodes is therefore proportional to the difference in the electrochemical reaction rate:
CARANTIA
O estatue a poe para bermicnias inductres e um cas inermeas rana atae.
ca n tatee ao das abecas cae was reectiaticns abe pesis.
nuchoncrprrsperolstocrnprrurarccgagculrraepckfinaamontane esbcnofo de tlatratoe.
Pare set nurn un rurrenti proffeslonai quaidhfo denepernn. Foal de dirin, e mupar a ou cnsion.
Ful. realis, ro de e le kaii tobio, tibicato o ceno da certa em aropora e do inchoal, 104-105; 106-107; 108-109; 109-110; 110-111; 112-113; 114-115; 116-117; 118-119; 120-121; 122-123; 124-125; 126-127; 128-129; 130-131; 132-133; 134-135; 136-137; 138-139; 140-141; 142-143; 144-145; 146-147; 148-149; 150-151; 152-153; 154-155; 156-157; 158-159; 160-161; 162-163; 164-165; 166-167; 168-169; 169-170; 171-172; 173-174; 175-176; 177-178; 179-180; 181-182; 183-184; 185-186; 187-188; 189-190; 190-192; 193-194; 195-196; 197-198; 200-202; 203-204; 205-206; 207-208; 209-210; 209-209
INDICE
LEGENDA RELATIVA AOS SIMBOLOS AVISOs DE SEGURANCA DESCIÁNÇO DO PRODUCTU DE ACESSORIES AO MANUTENÇO E DESINFESTAMENT
INSTRUÇOES AMPOLA NEBULIZAZORA
PROBLEMAS E SOLUCIOSA
COMPATIBILDADE ELECTRONMAGNETICA
PROCESSO DE ELIMINAGUARIA
GARANTIA
PAGIÇA 33
LEGENDA DOS SIMBOLOS
Achenericn Sinoce de "Iue e peas apracaes"
Pobigc C0425 Corrodeos cor a cieplopia.
Aetioe i aenimmele cupea o am nate do ctioeis mncias
Asinurtei dase I LOT Nurnen utre pretoe
Inntcruterigace intnrucor contigae
Nan utlter o aspche
dutie o tido nui
dute
Sta as inuus, dretioe coagens sIinapcpe pumae cemocpn aithi
Todas na pape do bolte de inolubulacion podra ser celululosic a vapor o from a esquezao do tóta e a as enacristado a 120^ (Ori cr). Cl = 134^ /V. No. Na tóe da ineligenda de aneterne a deposir o das varas pares, a temperaturam antibintecuores ou ruc. Noto pinto o cido de escoltracion consecutiva.
VIDA UTIL DO DISPOSITIVO
Reccionario a subscrito da interruptor de ourofacio apso maxima acipoes 100-120 ou depresa da cera de 20 cicles da atehartrac.
CONDIÇOÉS DE ARMAZENAMIENTO E TRANSPORTE
De transpoe or amea ramento o de tempo hua-25oC a 70oC Percentao de la mida doce estro e a meaneamento e transpoae:10% a 35%
DADOS TECNICOS DE OPERACAO
Proces: 003.000000000000000000000000000000
Assume, no case of poisoning occurs, it remains to be investigated. Consequently, the risk of anemia is assumed to be 10% . The risk of anemia is estimated by the following ecchymoprophylaxis: a ventricular ejection fraction < 50% and a pulmonary arterial blood flow rate < 60 ml/s.
5733
Fum cie de tles coiubacn 3eepo de apoeon no cielae cu corruea en labees reccntes de emiclcs cectmagnificas que o cno tram a acugir de preeintbntuotics
PROCESSDE ELIMINACAO
| Aquelos acumen de imunidade na r.l. | |||
| Culpez/SPINV dovelo ou para l'auxiliares ou reviracles de eminente(s) eicospecuarias de assasçao, o cabovo ou utilizamem de SPAGG - AVV covers assacn que os cabovo ou assacn do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do assesdo asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do assemento do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses da amissão do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses duas aposseio do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses das amissão do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses da amissao do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses dos asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do assaydo asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses daamisão do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do assles do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses do asses DO ASSESSMENTO DE ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACESSORATED ASSESSMENT OF ACCESSORIES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSEDAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENTDAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENT DIAS ASSESSES DO ASSESSMENTDIAS ASSESSES DO ASSESSMENT DiAs: | |||
h h t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t t s e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e e c a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a a
As distincas de asparao recomendado para equpamento de comuicaoes de rF portale mole e DisPOSITIVO
Pare T. Parneson and C. Diederich
p.pare.t.parneson@cs.ucla.edu
C. Diederich
Department of Mathematics, University of California, Los Angeles
C. Diederich
Department of Mathematics, University of California, Los Angeles
C. Diederich
Department of Mathematics, University of California, Los Angeles
C. Diederich
Department of Mathematics, University of California, Los Angeles
kose
10.12.1968, 345-347, 348, 349, 350, 351, 352, 353, 354, 355, 356, 357, 358, 359, 360, 361, 362, 363, 364, 365, 366, 367, 368, 369, 370, 371, 372, 373, 374, 375, 376, 377, 378, 379, 380, 381, 382, 383, 384, 385, 386, 387, 388, 389, 390, 391, 392, 393, 394, 395, 396, 397, 398, 399, 400
DE Deutschland
1 KOLBENVORrichtUNG FÜR AEROSOLTHERAPIE NE2012 ANWISENIGS UND GARANTIE
A Symbol of "Typical Anatomical Images"
LOT Number of Product/Package

。

NEENHIN
Description: The text structure is 20,000 characters. In a text file, the text is saved as .tex.gz. The text is saved as .tex.gz in the directory C: and it is saved as .tex.gz in the directory C: . The command is: Export the command
-
Dic Vorrhundert cannot in der Zuschl. verwandt wocrn das in den Fehrenmesserungen aufgeliefen.
-
The International Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited Liability Limited liability.
eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae eae
- Wroten's Snus firdn an rana loane and aen (1a
Sormenter, Namoucuon and Simpeytaehor Ernssen geschgtt. Das Gert
muse crsprochord don chshlaggen Yoschnrter entsort wondn.
Luechng be Lgagnirans 10% - 85% HH
FusreenderBefiehlmalnk:150kPa75bar,29.85m17-0%
AirdasstImenge:2mHbchattlImenge 12 m
1 Leesie 1
8.4.15.10.11.12.13.14.15.16.17.18.19.20.21.22.23.24.25.26.27.28.29.30.31.32.33.34.35.36.37.38.39.40.41.42.43.44.45.46.47.48.49.50.51.52.53.54.55.56.57.58.59.60.61.62.63.64.65.66.67.68.69.70.71.72.73.74.75.76.77.78.79.80.81.82.83.84.85.86.87.88.89.90
dianrher hicee aee
St Dbrnncrbcuay airdo ampuokcnd la pae dair du comprescur.
10.2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018 2018
noncommutative iff the fibre is an affine subspace in every two horospheris (i.e. lies on the Tr_Z at a definitional fibre to a complement under its base of equivalence, hence h is coficlus). In fact, the noncommutative iff the fibre is a complement under its base of equivalence, hence h is coficlus. In particular, the noncommutative iff the fibre is a fiber of its son of cohomology if and only if it has a subfibration onto the surface. These notions of complement or fibre endow us with a new definition.
c. base in - Frobenz Apparatus (2042/63) c. cotext reference document (1 applications of the species, references, q. persu) one additional reference document
INDICATIONS DE SECURITE
INDICATIONS POUR LE NETTOYAGE DE L'APPAREIL
Aaiee eae aee aeae aee eae ee eae eae
the information that the members of the membership group can use to make a contribution to
FR Francais
#
in the middle of the summer
IPOEIIOIHSEI T A THN AEAIA
NEPIPPAΦH TOY NPOIONTO
OAHNE XPHN
SYNTHPHESH KAI ANOAYMANSEH
0eAia52
eai52
0ekla53
a54
eai6a54
OAHHEAMNOYAAEKNEΦQTH 55
NPOBAHMAKAI ENIAYEH
HAEKTPOMAFNHTIKH SYMBATOTHTA
IAIAKAZIA ANOPPIVH
EETYH2H
AEZANTEE SYMBOAON
10000000
Anewon
C0425
00
10000000000000000000000000000
同……
Tunzun Knnpoc
Karnofsky functor
10
L0F
□□

yepnncn
aonrnoe
013x10C
1
A
2
100
IPOEIOIOHEIEI THN AΦAIA
y
eepn. To mnnn nnnn nn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn nnnn
paepia amo mkiKaKuTuV 36 npnuuv npi npaupeuvi pkp npq K
unipexivouocKoTOnOng.22088nHnLp12500110
nep
- auroevi evi erpoeyovikipnivdikskn jpnn npeh
xepnpsnsfai katoen lortpkc ouyvek
Eepnreepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnepnep
Ee e
e 1
H
M
P
EGH
- 2e nepnncn Pdne nnnnns aenrnnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne ennnnne
aouoae aouoouoetto ooepepeepnepne fie cokpckene
M
- 1
MnK nppe nnt to aaleo ooloe h m ouen y w o opepet
MHNypononoeHnne noocnoe, npopooecic, eruHt to kdo o do
mnnnne nnnnne nnnnne nnnnne
Hn Hn npepnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nnnnne nannnee
P
- 21pae nnoe rnnnne anr noonnnn Cn nn nnnn neanen ne aee
中
Bto 1000000000000000000000000000000000000000
P07T05060473012
K
E
1
040011111111111111111
MHN xpnojmoznt in nuckun o npnvi ng vnu
NEPIPPAΦHTOYPIONTOE@TEIK.1
1.
1.
-
RORINNEEIEECAPP
-
Genotyping protocol
6.10000000000000000000
7.2018年1月26日
8.12009/03
8.102.103
10.1F
11.5
17.2017年1月26日
12.2006p9 410
13.A.F.CUO BRW
A01F56
Aepnnepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepepe pe
1.135600000000000000000000000000
15.2019-03
13.48(20)
TEYNIKA XARAK
E
C054-9817,1982
10
012
Pn
030MAX:05Kpa-5624
EL EAnvIká
Pn 1234567890
- Isolation of two unboundable (covalent) bonds into the amine group was allowed in a reaction with H2O and CO2 . The reaction was carried out in the presence of the catalysts, oxygen or anhydrous base. What is needed for the synthesis of these
papoeepoeepneppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppeeppe
Aum un cumcun exci ecbdoarai laeauoay aoe bdooraiu taum Karmu on 40 karmu On. 19 crs mncn cscr 20 amu kaeuy
SYNTHPHSEH KAI ANOAYMANEN
- Armchair/Chair/sofa/bed: Any of the above categories (e.g. sofa, bed) should be included in the category.
- Equipments to KITCH: Any of the following categories (e.g. kitchen, office, bedroom, kitchen/dining room, dining table, etc.) should be included in the category.
- Equipments to EK2: Any of the following categories (e.g. kitchen, office, bedroom, kitchen/dining room, dining table, etc.) should be included in the category.
- Equipments to EK3: Any of the following categories (e.g. kitchen, office, bedroom, kitchen/dining room, dining table, etc.) should be included in the category.
OAHHIEAAMNOYAAEKNEOTH
H yomn o hynnnn nnye kanaonnneauu o uoouy ou tou the npnoanr aonnnnnaa Kaaonnnnnaa anen nnnae naannnnnnaa a a cennnnnnaa
CAHJETAFAEIA
Pn 1
1. Do you know the name of your partner who you are married to? Is your partner who you are married to?
Aaonnnoanrnnnnae anen enne nnnnne
H 100
O m i g n a t e s you o u b s e y i n c l a wed . k a r j p a rn 100000000000000000000000000000000000000000000000000000000000000000000000000000
Xipoparotne mero ino 1000000000000000000000000000000000000000000000000000000000000000
My prpaponee in bafidn oce n paoopn opn eno an andp aipnane, bradn daovnnn ne expenr dbrnne. C kathoocm
H 1000000000000000000000000000000000000000000000000000000
thar a. napopovskiy na tajnaya napiyaknye krasnoy dvoi hovni yu noyu yu nopechuk vaskhuya ukiy, ukiyra konrozhny, ukiyra mirokhny in groupy okshev, noyek, noyek noyek noyek noyek noyek noyek, noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek noyek
Kothe ine ern mki kollig jmu, joukoi omui quin sice ene yuie eepenponti. 104-105. 236-237. 238-239. Anapis n in Bredni 24:496-497.
YIOYEIEE JIA TIN HIN POITOMAIKA KAI AHTEYOPIA THE DIATHEAE H Donny Fie Thoe anemoparot, ye loe aoa hau, rpo in apopopnepo, npapopne p aokipopop o kai oanopopop quin eonuubovatn aenoropopop. O To ploite to opopop, oroue the apopopv evovatn kai npopvivp oponpdo mto raouk opeo qayu vavdokop. Apogote to me opop tse opop to apopop to vovatdokop (sp. 10) To cunmmpovtioe okei kai sckadocn on faoe eoee eae ene ovee n yqumuk, lalit Eek. Iromonoty the mnnnnoe of kai, ty. Auynopopovtioe o kai oos, oine nme nyqumovs, oevoyf.
TIPOXOH 2 mmpnne nnnnne pnnnnnne aenee ne ane ene eanenr. Kaipokpni kai noaannnne nanaeknne. Moe nnononnne to daoane, Paoae lae ta nnae npaae nae aene e ane ae ene eanenr poae nae oannpnne rnae npaae nane ene ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane ane aannee aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annnee aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene annne aene
YDOAEFEETIA TON KABAPIMNO THE ADATA=
The TIRADE KORA GYON, KOJIAPACTE ME YO KOBOPKU TAY LAKAMPOV TAY KOLAMPOV, XECKMOK, XECKMOK OXII, XECKMOK OXII, XECKMOK OXII, XECKMOK OXII, XECKMOK OXII, XECKMOK OXII, XECKMOK OXII, XECKMOK OXII, XECKMOK OXII, XECKMOK OXII, XECKMOK OXII, XECKMOK OXII.
Hepre 10000000000000000000000000000000000000000000000
EL EAAyIka
Aur 1.2.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000
Pnepnnn nnnnnnne nee eannnnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne enennne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne ennnne enannnee
EETYH2H
H napoloon nucn fauo 2x anr any n aynr taananae
naa nnnnnae nnnae naan nnae nnae nnae nnnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae nnae n.
gangjohn tnpapok, Xpapopopopioe an aepopopopioo no apopopopio no popopopioe n gong jhpn oopopopoe oopopopoe evoo mpeppie Tnv ootane eynocypic ympovoyers thou cmoa kyoos knoepn no npovvds in the 10000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000.
gangjohn tnpapok ooiy i ykooi kyoos knoepn no npovvds in the 11111111111111111111111111111111111111111111111111111111111111
gangjohn tnpapok ooiy i ykooi kyoos knoepn no npovvds in the 23333333333333333333333333333333333333333333333333333333333333333333333333333333333333333333333333333
Haeepoyeyntk oupbernta - emnpo0Betec onieeoeae ouipoppon ie to npotuno IEC 50601-1-2/A1:2004-09
ΔHQY2EVIH p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p# p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#p#
## # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ###### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ]### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ### ##### ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## ## # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # # ## |
Ttgeia aipoioc o poaeouyovntc
| H 2Y2EYEH m#p#o#e #y p#y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y # #y#m##n#i#e o# em#l#me#y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y # y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y #y # y # #x#m#n#n#i#e EN 6901-1-2 | Em#me# E#p#y EN 6901-1-2 | Em#me# E#p#y EN 6901-1-2 | H#m#p#y#m#r#d m#p#y ##y#m#y |
| A#y#u#p# p#p#o#p#t#s EN 6901-1-4 | 3 V#m #m 1500 L#z #c# 500Hz | 3 V#m #m 1500 L#z #c# 500Hz | C#p#p#o#p#k e#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#0# #o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o# o# #o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o# D#v##m##on#om#m#p#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#O d=1.2VfB 200MHz d=1.2VfB 200MHz d=2.3VfB 200MHz d=2.3VfB |
| A#m#p#o#t##s p#p#o#t##s EN 6901-1-3 | 3 V#m #m 5000 L#z #c# 2.50Hz | 3 V#m #m 5000 L#z #c# 2.50Hz | C### P#p#o#o#p#k o#p#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#0# T## P#p#o#o#p#k #m#p#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o# T## P#p#o#o#p#k #m#p#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#0# T## P#p#o#o#p#k #m# p#p#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#0# T## P#p#o#o#p#k #m# p#p#o#o# o#p#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#o#0# T## P#p#o#o#p#k #m# p#p#o#o# o#p#o#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#0# T## P#p#o#o#p#k #m# p#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#0# T## P#p#o#o#p#k #m# p#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# O T## P#p#o#o#p#k #m# p#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o# T## P#p#o#o#p#k #m# p#p#o#o# o#p#o#o# o#p#o#o# o#p#o#o# o# T## P#p#o#o#p#k #m# p#p#o#o# o# T## P#p#o#o#p#k #m# p# T## P#p#o#o#p#k #m# T## P#p#o#o#p# T## P#p#o#o#p# T## P#p#o#o#p# T## P#p#o#o#p# T## P#p#o#o#p# T## P#p#o#o#p# T## P#p#o#o#p# T## P#p#o#o#p# T## P#p#o#o#p# T##P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T## P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T##P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P T## P |
580
RO Romaná
APARAT PENTRU AEROSOLTERAPIE CU PISTON NE2012 INSTRUCTIONU SI GARANITE
- Uritate principal
-
Contractual obligations
-
Commitment scope
-
Introrupator "O" pmonrcpno
6.Capac compartment hirc act 7. Fivax folit
-
Muscle pediatry
-
Maece adult
-
Adopter based
-
Report date 2. Record date
-
Foli nebulizare
-
van hove 2. Pudlak
B. Bata hova
-
Tuesday
-
Filtrud ezer
SPECIFICATI TEHNICE
Numele produsui. Elecromperoei piono cu depoziln de protelcie porhia
Denurine commercial: NE2012
Româna
Tensunr nominala:
Tensiure (230V 50Hz 150 VA)
Dispositivily a first project port to function area intermittently.
Disposizn 10 min pcrity 0 nort Crti disziitui dura 20 de minu
728mm, pomm-40, 6pin, 6pin disjoalva dupo 20 de 2015.
treatment
INTREINERE SI DEZINFECTARE
: 1
000
Gurati qu attic trats compapertcs si indepartet i zduilis ds produs
farnaccutis si posblicis impuriti dupa fiscare tratament.
Uritalra prinieai si hul de, aer yor li eurati s u s sreuiu
curata. Nu spalsti niodata operatul cu apa curenta gau introducandu-1
in ap. Aparal cu carcasa neprolejala la palrunderea lichidelor.
Curalarecu ap: Curatali loale accosorile cu exceptia tubuli de
acr cu apacurrenta (max. 60°C) circa 5 minute adlaugand dae cste
ncocar o cantitato mica de detcrgnt respectand dozarcay imitolo
prevzute de produciorul de detengent. Clati atent astiel inciat sa
Indapartat toate rezicunle iass sa sa uuce
- Dezinfectare: Toale accesonle, cu excepta lacului de aer, pol
n dozirtoate du ozirtoctant chmic foosind dozie limitee
A. p53 is a key to the cell cycle
Zakaz
C€0425 legnosticz zefraegnstickc
y
Vyrrobco
Pozzi Pocziela is a professor of rubro k. puziau.
Caley vycbri Earze
Prrrty
1
Stiday's prnse
Napuzjivite pfissto
Sino
P21. Stupen oghanye alekniiksh zafizn kotein, kke poyi fisiie
cnduie stupen ohrany protvniknul pavnych cizich 10
a2) a drunclisco stupon conrany prown khrjkapain 10 az 81
BEZPECNOSTNI UPOZORNENI
P Fred pouzitim wyobku zkontroluite, ze je pfistro neporulory boz
vidtaiych znamek pokozani. V pfipadpoohybnosti vrobek
68.60
Czech CS
nopou2vno a cbrfle so na svno prodcoo.
UdZjue plastoy aak kory je soclst obalu, mmo doah dcl: nabopoduducel.
Tenta vrybok mus by pauzivan yhrndn k dolu pro kery by
sretroj, spisibn brrnacrnn n vahu k palitn lakeki
Pra prae - praeovrana - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vaprevo - vapreva
nndnoedoe ordynoe pozill. Ploep zopoeim pflstroe o dccktixse sie so uistte, 20 odag
napeli na hypovem stilku na vryobku shoduji s idaj pouzie elektricke s16.
Provoz a Uorba Tohola Vryobkui meheu byt provodcys demi atarsi 14 let a, osebokai o amosaninii tukunai, rupovskoi nao, duvouki
f a oobn b 100000000000000000000000000000000000000000000000000000000000000000000
fadenym dohlemdoepeo asoby. Deti nesmls pfistrogm hr.
lubeynivit, amnuol mme doahd tni pldj36 min
protoze aboahme laje kaste, titeré, chez motly spkhnout. V klaiden prpidauchovcje pfluslenetvi a elektroky kabim meno dccti h
Toto je zdrazovtornyicki prostroj prot domaci pouziti a musi blut pouzii na priladji laskatkiho plozniu. Musi blut pravnozvov tao,ink in
[1] Zakiakae Iokaikensko predugosl, Musti by, provedovan tuk, jek je uvededeno v tomo navodu k pouchit. Je dulezte, aby si paci sar
precell a pochopil Informace pro pouziti a udribu jednotky. V prinadie jahnekoliy dolazu se phrata na synhe prodejfc.
Prisirj nem vhdyn pr pozilv primmns hafve anestelicke
sne 2dchm. kni hnd oxidn dusyn.
yprne 3 yylnne 2 elekricke site.
Zerabanej a Vbopn opei, orrants j prd nary. extramini zmami Irnol, vibkost, precnem, sllon, slunpim, zafipn
C. 100% complete, 100% complete, 100% complete, 100% complete, 100% complete, 100% complete, 100% complete
Vipada puroch yane bospa nfnkosti pnirsi vprelte. nepanipula snil a palidnla do prawot koupil Proyorye
oeracero vzy na svno prodcjc.
NEZAPOJITE neho nepapojite phtroj, pukid mate mokne nto
Pn pouzivni ypinae Oit se ujiste, ze maie suehe ruce.
Pip odpoopnivztreky zelektnckzisunkyNETAHIEJTE zpiVodni kabel an zu prist
NIKDy npouzivc produzovac kaboly nobc adaxry, ovmno
kabo po cek [ohodeku audru toe] daleko od zrodto topla.
S Pozer NEZAHAUJE z 2adneho duvoju na ektrickem kabolu. V. p
Pristi j zdy vypriie ypimacn O'at pale cdpjle zistkz
ZASUNKY.
Chamzlo po pouzili. a pred slatenim, yjmaba zastrok piistroje ze
NIKDY neponofuits vrobok do vody api jinych tutit.
Spadre l pistr dvo de NPEKOUKSEJte se je yvloit, ae opepts in pe prrnne e kkrkite sile NEPCUZWE Tznu gfrsnt po
vynndn vocny (nneh kontkdtute sveho prodeje).
NEPOUZIVEJTE pnstroj pn koupani ci sprchovani.
POPISSYROBKAU (W208r.1)
1. Hlavni Jadnotka
- Kryt prakhady pro lozuien prialusenii 3. Bihdovce pro lozhen plaiuianii
- Yenacn'OJ: zannul/synvnul
- Privad vzduchu kompresora
3.Vao prihidky zuduchovehe fillr - Dorn ample 8. Oblique map
- Oblique massa pro dos
- Nosni nasiavec
- Nausiek 12. Glaes
- Sodovac histamine amplici e 13.NeulizapcI sample
a.Horni stn ampls
b. Nebulizacntryska
E. Spedničšć and Žižek - Vartapetko and Hulick
15.2008-03
10.12.2017, No. 1
TECHNICKE UDAJE
Naezyvynbuk-Elekricky piatoyk kmpresor sapehnoochranou Chmchni 1920
100000000000000000000000000000000000000000000000000000000
NapSt (230V 50H)
PojskaT1.6A-250V
- Sporobite videa vypnicae: VOT is a palum opodae adie.
- Sporobites sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp. sp.
- Sporobites videa vypnicae: VOT is a palum opodae adie.
Ak zahedere spodne so voly, nezhering zu in neu dossi ale hined ywhilzirah zohane zu zauwily. NEPOUVAJTE zahedere po aybilr a 100000000000000000000000000000000000000000000000
NEPOUZIVAJTE zarladenie pri kupani alebo sprchovani.
POPIS VYROBKAU Nd abzak 1
- Hlawni jednata
- Kryt prieslora ma ockladane prisusensiva 8. Euphysa e aodilatd nis
4.1 - yinfei On Zaphy, yinfei
- Enwzxd zydushu de kongresa
- Kryt uozenya zydushoyeha jilra
7.Uchytenie rozprasovada - Maska pre do
- Masa are dan
- Nomenclature
11.Novak - Prigika na mzprafoy
- Rozprasovic
a. vronna, das rozprasovc. b. Ropotravounkis.
D. OZPASOWA/YSK 2. Spodnt Asf mzocat - Hacička pravodu vžižchu
- Vzouchov filter
TECHNICKESPECIEKACIE
Nazov ymbu: Elektrickiy picey kcmpressor s topchou sharenoy
Oboshny nazov: NE2012
Meraville napelle
Nap90(230V50Hz150VA)891Ku(1.6A-350V)
1088(11.0A·200V)
Slovensky
Provadzov taki (n zoprasovacm): Pritl2ne 0,6 bar
Maximinyink: phti2e 185 kpc
Maximun yck p120 165
19824250376 1. HUANG, 58 dBA
Roxprasovanie:0.35ml/min
MMAD:2,61 m
GSD:2,75
Aerosoloy vysub: 1431 Wirtnupa hndpata:671
yysipda hnsoc 67p1 Kilisadn prsill 2040 min ONOFF
Tieda nebezpeansly, Ia
Zvotnosf pou2lvaneho vrybku:5 rokov
Provadzov podmieniy
Teptata: min. 10^ max. 40^ , vlkasi zvduchu: min. 10% max. 95% . Podminnysk skladovagin:
Tepala min.25°C max.70°C yihksgyduchu min.10% max.95%
Almosteriky tlik pri prevaczei skladovani: min. 590 hPa; max 1060 hPa.
NAVOD NAPOUZITIE
POUZTIE
1) Pred prvym pou2lrm zaradenia zaradenia vyclette spcsobom
popisarym y odaeku "Cstonic a dozintokcia
2Vlozto zastroku do elektrickej zauwyk.
3) Praprile Rozprasivac podfa potkyev v cach Navad na peuzhie rozprasivaca".
4) Pevne propojedejen konie vzduchovej hadice k Rozprazovacu a
4. Brutite, vionet, pfalnfert, maw, pfuht, alba, paevr
5) POZIO ZVOCHE PICHSSIO, IN: padstayes, npila sokrynx Ikhara
6) Zariadenie zopnite, silaenim ypinina "OIT do palohy T.Vlozte
nuslo do 1s alebo zalite msku na Ivar, pricorn sa uistile, 2e
Zakryve Lusie & ro. Pouzic rausiku ziepejne prived loxu 60 pruc Tio Rees Jefu sa poeine nadvchula a vovnivulna. Sodex u
polone so vpranenou homcu castou tola. Pocas inhalela si
nelahnite.Vcrpde choroby irinalciu prurosto.
8) Po inhalaci yypnite raviadene otafenim vypina 120
O-3V1HnE Z5sREKO 2028Nk
9) Opejote Vzduchov radu od Rozarsobova a privosu Vzduchu do kompressura.
DOLEZITE: Ako pri vacine zariadeni na aerosolov terapiu,ajtu
pocokonsni lnalace zostans v rozpragovacr urche mnozstvo
kee to ie upine homane. 100 hnozwo leu, nazyvare nez
Vgrzinite zostavjuci lijk z mzprasayata a pristite hp codla
pokynov popisanych v odseku "Pokyny na Ostenie zarladenis" v
cstlNavod na pouzite rozoresovaca a ocla
pod'apokynov v chsti"UdZroa a dozintekla 11).Zvifte kifelavuivne hdo zinruiyne bnoi
11) 219630026 no prnshno presio h 22878 stn
Tolo zariadenie je urcene na prerušovanu prevadzku 20 min.
712aphut 40 min. yphut, P8 20 min. tach prevadzky zaneodr
UDRZBA A DEZINEFKCJA
Pred yvonanim operaci distania a dezintefke zariadena i starostlvo amrue ruky.
Po kazdo msetre lstarstivv ydsite vetykmpenenty a
B
Flwia jnndk 2VzssbHs hbs je pbeBc cun cibl. Wmdd handickou, Spotepik nkiy neuoyaloe pod tegucu yodu alba
ponorcnim do vody. Zviadnic s obalom nochrancim proti prcnikaniu
kvapalin
Cistenie vodou: Vstko prisulcstvo okrem vzushchov nadlo
Ministry of Health, 2014
MaximSry abem dpnria: 12 ml

Pozome si precalaje nayd na puzie
PROBLEMY A RISENIA
| Problème Możña príncia Riesénie | |
| Spatétrèc sa nezapine. | Zătróku rîe i zapiopojna do Zărśćy. |
| Vypnáč "O'IT je v poite "Or". | |
ELEKTROMAGNETICKA KOMPATIBILITA
Fyegelert! Fyegelensen 10000000000000000000000000000000000000000000000000000000000
II kategoriakedes
LOT Gyanas tioe zma
Kapacso bilokapacso
Kapaclo kkaposiva
Nehaszindzki a kusierlendkler vorge 1
Vatcainr
IP21 Elektronos kósiázkoc csemogaiāsān ČVdVcTeSg Ioka, ahol az ázó šelämçyö a kújó szilfart teok baskovával szemón (0-6), mig i másöridská šelämçyö, a hyärdélké behatalást val szeničné vědětej tókán (0-8) jelői.
8485
Magyar HU
Poriasz14:0.35mIperc
MMAD:2.61μm
GSD:2.75
Aerzssr Kihner 143 1
Deiigles haszrolal 2040 aeg ONOFF
Kockazl oezaly. Ia
- hasznall termek varhato eettartama: 5 ay
Ozim tehich: himikhtat.
Tansierer 10-0, max. 40 C, paralrurant Fm. 10% Max 95%
barmerseklet: min. -25°C; max. 70°C, paralatalom: min. 10% max. 95%
Uzemi: tarsolasi lagryomae: min. 590 hPa: max 1060 hPa.
HASZNALATUTASITASOK
HASZNALAT
- Az eszkoz elso haznolata eloI empell liscilbari a Karbanlartis es
NOS: A2 a9301000000000000000000000000000000000000000000000000000000000000
FIO
menniyseg, melyet maradek mennylsegnek is neveznek, nam
gorasztnato
Akapson obilsken le, laivikskon el tejeses vozel es hagiyak Biai.
Baiyak kaiyak, 120-121.
Baiyak kaiyak, 120-121.
Baiyak kaiyak, 120-121.
Baiyak kaiyak, 120-121.
Baiyak kaiyak, 120-121.
Baiyak kaiyak, 120-121.
Baiyaik kaiyak, 120-121.
Baiyaik kaiyak, 120-121.
Baiyaik kaiyak, 120-121.
Baiyaik kaiyak, 120-121.
Baiyaik kaiyak, 120-121.
Baiyaik kaiyak 120-121.
Baiyaik kaiyak, 120-121.
Baiyaik kaiyak, 120-121.
Baiyaik kaiyak, 120-121.
Baiyaik kaiyak, 120-121.
Baiyaik kaiyak, 120
AZ ESZKÖZ HASZNOS ELETTARTAMA
TABOLASLESZALLUTASLEETETELK
Teles as zalitas fhtalek-25°C:70°C
Te
UZEMIMUSZAKI ADATOK
Porasuri ampulla minimans diminuta 5 (pete: 10% Porasuri ampulla minimans 694 (2.6 bar, 22.000)
Porasuri ampulla minimans 75 (pete: 10% Rehita (oligo hydramn.) 150~Xpa^ [1.5 bar, 22.000] × 10% Mollusca hydramn. (oligo hydramn.) 150~Xpa^ [1.5 bar, 22.000] Maxilla (sulcus) intercalata: 12ml
Figolmoson ovasek ois heszonal urasitacokat
PROBLEMAK ES MEGOLDASOK
| Problema Lehretseges ok Megoldas | |
| A kozilskatn nonkapoizl be. | A cassiaradzugü nizic a falla caskoikzbs lilevst. |
| Az "O" kaskoizl "O" alisken viz. | |
| A kozilskat be van kapsoke, de rem porafis#. | |
| Nizic an ampula beltebeien a parsiazukovia (13b). | |
| A levegizcak megnejot i neksejzernyod. | |
| A laskoizl alagut vizg kozits. | |
| Az ampula beltebeien nizic gyogazier. | |
| Az kozilskat non mlokidik. | |
| Az alabii okoizaki badavkaiztai hóvedeizem: | |
| A kozilskie a kozlónyden telitfnihta (izam) hatakrudnik koniul mlokidik. | |
| A kozilskie hórdássok kozelétský O'Kl'tc-meghainu námskáktlki hólvskozen kizollm. | |