BD4600 - Blood pressure monitor TOPCOM - Free user manual and instructions
Find the device manual for free BD4600 TOPCOM in PDF.

User questions about BD4600 TOPCOM
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Blood pressure monitor in PDF format for free! Find your manual BD4600 - TOPCOM and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. BD4600 by TOPCOM.
USER MANUAL BD4600 TOPCOM
UK The features described in this manual are published with reservation to modifications.
Section C1-2 FuHai Industrial Zone
Fuhai Road Fuyong Town
Bao'an District
518103 Shenzhen City, Guangdong
PEOPLE'S REPUBLIC OF CHINA
Shanghai International Holding Corp. GmbH (Europe)
Address: Eiffestrasse 80, 20537 Hamburg Germany

Keep dry.
This Blood Pressure Monitor complies with the EC Directives and bears the CE Mark "CE 0197".
This blood pressure monitor also complies with mainly following standards, (including but not limited).
Safety standards
EN 60601-1 - Medical electrical equipment part 1:General requirements for safety
EMC Standard
EN 60601-1-2 Medical electrical equipment part 1-2: General requirements for safety-Collateral standard: Electromagnetic compatibility - Requirements and tests
Performance standards:
EN 1060-1 Non-invasive sphygmomanometers - General requirements
EN 1060-3 Non-invasive sphygmomanometers - Supplementary requirements for electromechanical blood pressure measuring systems.
EN 1060-4 Non-invasive sphygmomanometers - Test procedures to determine the overall system accuracy of automated non-invasive sphygmomanometers.
The declaration of conformity can be found on:
www.tristar.eu
We request that you read this information in order to quickly become familiar with this equipment and learn how to use its functions.
1 Information Regarding This User Guide
This user guide contains information on the BD-4600 (referred to as 'the equipment' in the following) and provides important instructions on the initial use, safety, proper use and maintenance of this equipment. Always keep the user guide available near the equipment. Every person responsible for operating, repairing or cleaning the equipment must read and/or consult the user guide.
Keep this user guide in a safe place and give it to the next owner of the equipment.
2 Copyright
This document is protected by copyright legislation. All rights are reserved, including rights of photomechanical reproduction, copying or distribution using specific means such as data processing, data storage media or data networks, either in full or in part, as well as technical changes and changes to the contents.
3 W a r n i n g s

CAUTION
This warning points out a potentially hazardous situation.
Injuries and/or material damage may result if the hazardous situation cannot be avoided.

Follow instructions for use.
4 General rules when using the equipment
This device uses the oscillometric method to measure systolic and diastolic blood pressure as well as your heart rate.
This product is for household use only. This product is recommended for users over the age of 18, and should not be used by children. It is not intended as a substitute for consultation with your doctor. All other forms of use are improper and can lead to hazards.
Always follow the instructions provided in this user guide.
No liability is accepted for any loss or damage resulting from improper use.
The user of the equipment is exclusively responsible for all risks.

CAUTION
- The unit should not be operated by children so to avoid hazardous situations.
- For users diagnosed with common arrhythmia (atrial or ventricular premature beats or atrial fibrillation), diabetes, poor circulation of blood, kidney problems, or for users suffered from stroke, or for unconscious users, the device might not be suitable for use. Consult your doctor in case of any doubt.
- The unit contains high-precision assemblies. Therefore, avoid extreme temperatures, humidity, and direct sunlight. Avoid dropping or strongly shocking the main unit, and protect it from dust.
- Do not press the start key when the cuff is not placed around the arm.
- Do not disassemble the main unit or cuff

To avoid inaccurate results caused by electromagnetic interference between electrical and electronic equipment, do not use the device near a cell phone or microwave oven. At least keep a maximum output power of 2 W yields and a distance 3.3m away from this equipment.

- If the unit is stored in a cold place, allow it to acclimatize at room temperature before use.
- Do not take a measurement in a low (less than 41^ / 5^ ) or high (more than 104^ / 40^ ) temperature or you may get the inaccurate readings.
- Wait 30 45 minutes before measurement if you've just consumed beverages containing caffeine or smoked cigarettes.
- Rest at least 5 ~ 10 minutes before taking a measurement.
Relax at least 3 5 minutes in between measurements. - Make sure the cuff plug is attached to the monitor.
- We recommend you using the same arm (preferably the left arm) and measuring around the same time each day.
- Perform measurements in a quiet and relaxed environment at room temperature.
- Sit down comfortably and place your arm on the table with your feet flat on the floor.
- Wrap the cuff snugly around your exposed upper arm and lay your arm on a table or a flat surface at the same level as your heart. Your feet should be flat on the floor.
- Do not move or shake the device during a measurement.
- Blood pressure measurements should be interpreted by a physician or a trained health professional who is familiar with your medical history.
- Using the unit and recording the results regularly for your physician to interpret, you will keep your physician informed of the continuing changes in your blood pressure.
- If you have one of the circulatory problems as arteriosclerosis, diabetes, liver disease, kidney disease, severe hypertension, peripheral circulation......, please consult your healthcare professional before using the device.
- This product is not suitable for people with arrhythmia and pregnant women...
- Blood pressure measurements taken with this device are equivalent to those obtained by a trained observer using the cuff / stethoscope auscultation method and are within the accuracy limits prescribed by the Standard of EN 1060-4.
- This product is not a toy. Don't let children play with it.
- To avoid accidental strangulation, keep this product away from children. Don't let them wrap the tube around their neck.
5 Blood pressure
5.1 What is Blood Pressure?
Blood pressure is the pressure exerted on the artery tube while blood flows through the arteries. The pressure measured when the heart contracts and sends blood out of the heart is systolic (highest). The pressure measured when
the heart dilates with blood flowing back into the heart is called diastolic (lowest) blood pressure.
5.2 Why Measure Your Blood Pressure?
Among the various health problems afflicting modern people, problems associated with high blood pressure are by far the most common. High blood pressure's dangerously strong correlation with cardiovascular diseases and high morbidity has made measuring blood pressures a necessity of identifying those at risk.
5.3 Blood Pressure Standard
This device is equipped with a WHO Blood Pressure Indicator which classifies your blood pressure measurements into six stages (Optimal to Severe hypertension) based on WHO classification on blood pressure levels as shown in below chart.
After each measurement is completed, the LCD display will show your position automatically on the six segments of the bar indicator which corresponds to WHO Blood Pressure Indicator 2.
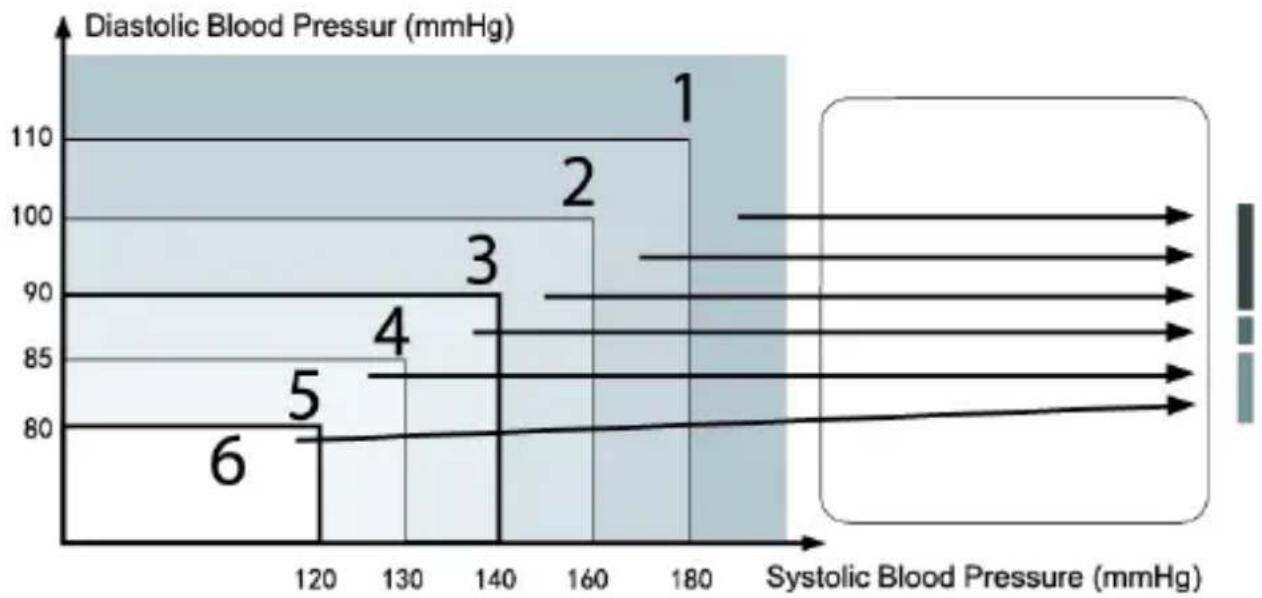
- Severe hypertension
- Moderate hypertension
- Mild hypertension
- Normal systolic value
- Normal blood pressure
- optimal blood pressure (target value)
5.4 Irregular Heartbeat Detector (IHB)
The symbol IHB will appear on the display, indicating a certain heartbeat irregularity was detected during measurement. The heartbeat rhythm that is more than or less than 25% from the average rhythm is usually defined as an irregular heartbeat rhythm. Talking, moving, shaking or an irregular pulse during the measurement can result in the appearance of this symbol.
Usually this is not a cause for concern, however if the symbol appears often, we recommend you seek medical advice. And please note that the device does not replace a cardiac examination, but serves to detect pulse irregularities at an early stage.

- No Irregular Heartbeat Detector measurement will be stored in memory.
- The pulse display is not suitable for checking the frequency of heart pacemarks. If a certain pulse irregularity is detected during measurement often, we recommend you seek medical advice.
- However, as a safeguard, we recommend that if you have arrhythmias such as atrial or ventricular premature beats and atrial fibrillation or any other special conditions you should check with your physician before using your device.
- The IHB function is not designed for use by people with arrhythmias nor for diagnosing or treating an arrhythmic problem. In order to filter the unstable status of user and avoid affecting the detection of heart rate from any movement, shaking or talking in the beginning of measurement, the method of averaging heart beat intervals for subject device is calculated with the three proper heart beat pulses detected in the beginning of measurement and that is different from a strict mathematical averaging of all recorded intervals.
- At least 3 beats with at least 25% difference from the average heart beat interval will generate the IHB icon on the screen.
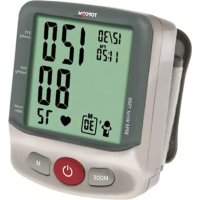
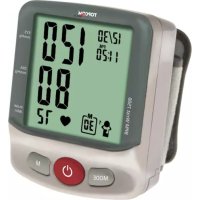
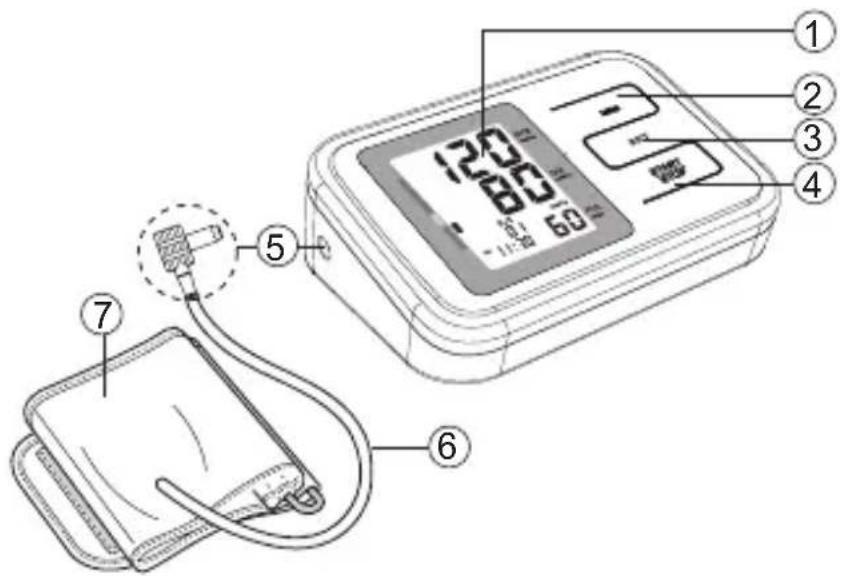
6 Product description
- Display
- MEMORY button
- SET button
- START / STOP button
- Air plug
- Rubber tube
- Arm cuff
7 Display description
- Systolic pressure
- Diastolic pressure
- Blood pressure unit
- Average indication
- Pulse rate
- Heartbeat symbol
- Date and time
- Memory
- Irregular heartbeat icon
- WHO classification indication
- Battery low icon

8 Installing batteries

CAUTION
Failure to use batteries according to instructions can be hazardous.
Always use Alkaline batteries of the same type.
- Ensure that batteries are installed with correct polarity. Damage to the batteries and the equipment can result from installation with incorrect polarity.
- Never toss batteries into a fire, due to the risk of explosion.
- Never allow battery terminals to come into contact with each other or with metal objects, either intentionally or accidentally. This can cause overheating, fire or explosion.
- Keep batteries out of the reach of children. Consult a doctor immediately in case of swallowing.
- Fluid from leaking batteries can cause permanent damage to the equipment. Take particular care when handling damaged or leaking batteries, due to the risk of corrosion. Wear safety gloves.
- Remove the batteries if the equipment will not be used for an extended period.
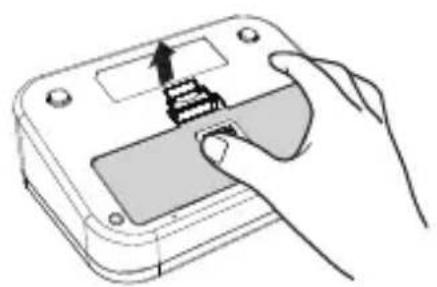
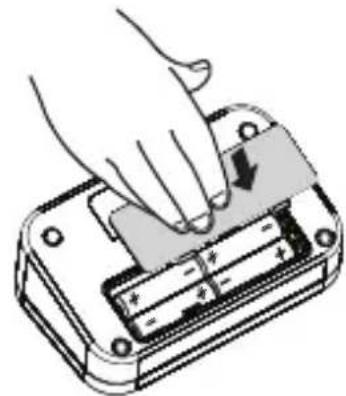
- Open the battery compartment by pulling the cover away on the bottom of the unit.
- Insert 4 AA Alkaline batteries. Follow the polarity as indicated in the battery compartment.
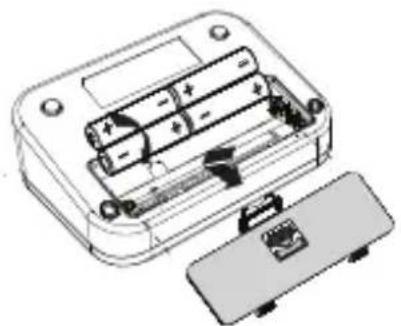

When the batteries are low, batteries must be replaced.
is displayed. Low
9 Setting date and time
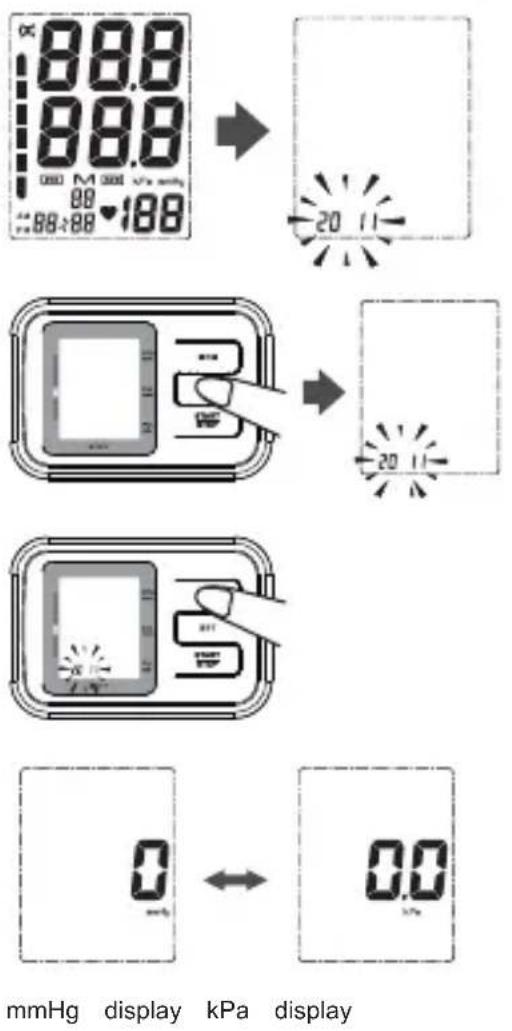
To adjust the date, time and pressure unit:
- Press the SET button for 3-4 seconds. The year will blink on the display.
- Press the MEM button successively to change the year (2010-2030).
- Press the SET button to confirm the year. The month will blink on the display.
- Press the MEM button successively to change the month.
- Press the SET button to confirm the month. The day will blink on the display.
- Change the day, hours and minutes as described above, using the MEM button to change and the SET button to confirm every setting.
After setting the minutes, press the MEM button to change the unit (KPa or mmHG)
After setting the unit, the device is ready to use.
10 Applying the cuff
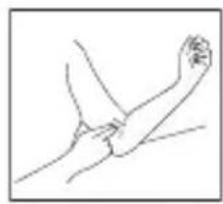
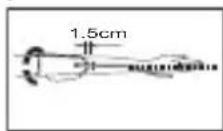
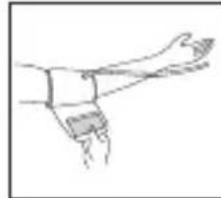
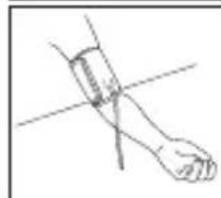
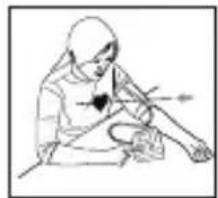
- Remove all watches, jewellery, etc. prior to attaching the Arm cuff.
- Press your brachial artery by two fingers approximately 2,5cm above the elbow on the inside of your left arm to determine where your strongest pulse is.
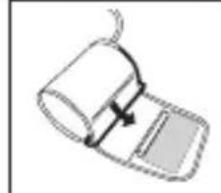
- Slide the end of arm cuff furthest from the tube through the metal ring to a loop. The smooth cloth should be on the inside of the cuff. The Velcro will be on the outside of the cuff.
- Put the left arm through the cuff loop. The bottom of the cuff should be approx. 1,5 cm above the elbow. The cuff should lie over the brachial artery on the inside of the arm.
- Pull the cuff so that the top and bottom edges are tightened around your arm.
- When the cuff is positioned properly, press the Velcro firmly against the pile side of the cuff. The metal ring will not touch your skin.
- Relax your entire body, especially the area between your elbow and fingers. Place your elbow on a table so that the cuff is at the same level as your heart. Avoid leaning back while the measurement is being taken.
11 Taking measurement
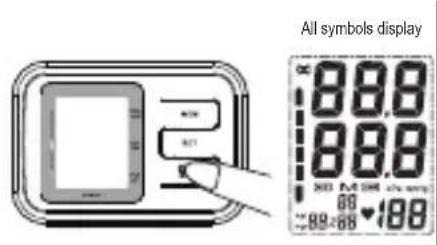
When the batteries are installed.
- Press the START/STOP button to activate the device. All segments will be displayed.
- The measurement will start automatically. The cuff will inflate.
After the initial inflation of the cuff, the pressure will slowly decrease and when a pulse is detected, the heart rate symbol will start flashing.


"kPa" display

"mmHg" display
This monitor will re-inflate automatically to more pressure if the system detects that your body needs more pressure to measure your blood pressure.
- When the measurement is finished, the systolic pressure, diastolic pressure and WHO indicator will be displayed alternating with puls rate.
- When an irregular heartbeat is detected, ' ' is IHB displayed.
- When a mistake was detected during measurement the Error icon 'Err' appears on the display (See chapter 13)
To interrupt the measurement, you may press the START/ STOP button. The cuff will deflate immediately after a button is pressed.
The monitor will automatically shut off after 2 minutes if no button is pressed.
12 Memory function
12.1 Storing data
The BPM Arm BD-4600 can store up to 60 measurements including the date and time The result will be stored automatically after the measurement.
12.2 Recalling data
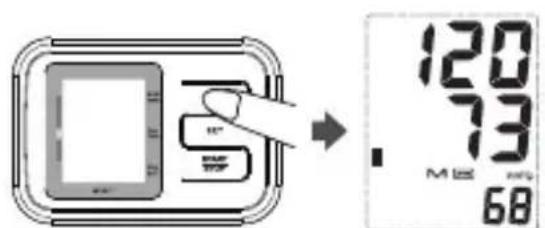
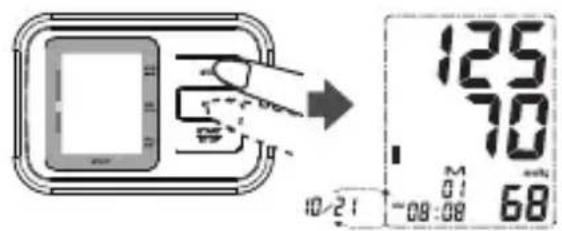
To recall measurement results: When the clock is displayed.
- Press the MEMORY button to enter memory mode.
The LCD will display the average of the last 3 measurement results. - Press the MEMORY button repeatedly for prior values stored the average of the last 3 measurement results.
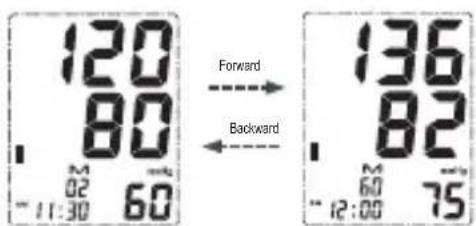
After the oldest record is displayed, the unit will display again the switch back to normal time.
To erase all data from one memory zone:
- Press the MEMORY button for 4 6 seconds.
- Press the START/STOP button to confirm

13 Symbol indication / Troubleshooting
| Display symbol | Condition/Cause Corrective action | |
| The flashing heart will appear when the pulse is detected during a measurement. | Measurements in process. Do not talk or move. | |
| Lo | Appears when the battery voltage is excessively low or the position of batteries is incorrect. | Replace all batteries with new ones. Make sure the +/- polarity is correct. |
| IHB | Appears for 1 minute when the user was talking, moving or shaking or an irregular heart beat was detected during measurement. | Repeat the measurement. Note that you be relaxed for at least 5 minutes and sit comfortably and quietly before you restart a measurement. |
| Err | Pressure system is unstable before measurement Fail to detect systolic pressure Fail to detect diastolic pressure | Wrap the cuff correctly and tightly. |
| Ensure the connection is close between the air plug and the unit. | ||
| Inflate over again after ensuring. | ||
| When measurement fails. Do not move you | ur arm and body, and keep quiet. | |
| Measure over again according to correct way. |
14 Technical specifications
| Model | BD-4600 |
| Power source | 4 x AA (1,5V) - Alkaline batteries |
| Measurement zone | Pressure: 0~299 mmHg |
| Pulse: 40~180 beats/minute | |
| Accuracy | Pressure: ±3 mmHg |
| Pulse: within ±5% of reading | |
| Pressure sensor | Semi-conductor |
| Inflation | Automatic - Pump driven |
| Deflation | Automatic Pressure release valve |
| Memory capacity | 60 Memories |
| Auto-shut-off | 2 minute after last key operation |
| Operation environment | +5°C~+40°C (41°F ~ 104°F)- Humidity 15% ~ 93% R.H. |
| Storage/transportation environment | -25°C ~ 70°C (-13°F ~ 158°F) Humidity ≤ 93% R.H. |
| Weight | +/- 355g (without batteries) |
| Cuff Arm circumference | 22~32 cm (8,7~12,6 inch) |
| Limited patient | Over 18 years old |
| Classification | Internally powered equipment |
| Classification of type BF(applied part) | |
| IPX0 | |
| Not suitable for use in presence of flammable anaesthetic mixture with air or with oxygen or nitrous oxide | |
| Continuous operation with short-time loading |
15 Maintenance
- Use a piece of cloth with water or mild cleansing agent to wipe the device and dry it immediately with a dry cloth.
- Do not use detergent or any strong chemicals to clean the device.
- Use only a dry cloth to wipe the cuff.
- If the device is not to be used for a long time, please remove the batteries from the device (leaking of battery acid can cause the device to malfunction).
- Do not twist the cuff.
- Do not drop the product and avoid any strong impacts.
- Do not attempt to disassemble or change any parts of the monitor, including arm cuff, due to substitution of a component different from that supplied might result in measurement error.
- Always store the unit in the pouch after use.
- Do not place the device directly under sunlight, in high temperature, or in humid or dusty places.
- Do not store the device in extremely low (less than -25^ / -13^ ) or high (more than 70^ / 158^ ) temperature.
16 Disposal (environmental protection)

Dispos of the device in accordance with EC Directive 2002/96/EC-WEEE (Waste Electrical and Electronic Equipment). If you have any queries, please contact your local body responsible for waste disposal. Batteries should be disposed of in an environmentally friendly manner in accordance with local regulations.
17 Tristar Warranty
17.1 Warranty period
The Topcom units have a 24-month warranty period. The warranty period starts on the day the new unit is purchased. There is no warranty on Alkaline batteries (AA/AAA type).
Consumables or defects causing a negligible effect on operation or value of the equipment are not covered.
The warranty has to be proven by presentation of the original or copy of the purchase receipt, on which the date of purchase and the unit-model are indicated.
17.2 Warranty exclusions
Damage or defects caused by incorrect treatment or operation and damage resulting from use of nonoriginal parts or accessories are not covered by the warranty.
The warranty does not cover damage caused by outside factors, such as lightning, water and fire, nor any damage caused during transportation.
No warranty can be claimed if the serial number on the units has been changed, removed or rendered illegible.
Any warranty claims will be invalid if the unit has been repaired, altered or modified by the buyer.
To ensure precise measurement reading, re-calibration of the device is recommended after 2 years from the date of purchase. Shipping plus handling cost and recalibration service fee shall be charged accordingly.
For more information, check www.tristar.eu
Besteklant,
| Guidance and manufacturer's declaration - electromagnetic emissions | ||
| The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that it is used in such an environment. | ||
| Emissions test | Compliance | Electromagnetic environment - guidance |
| RF emissionsCISPR 11 | Group 1 | The device uses RF energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. |
| RF emissionsCISPR 11 | Class B | The device is suitable for use in all establishments, including domestic establishments and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes. |
| Harmonic emissionsIEC 61000-3-2 | Not applicable | |
| Voltage fluctuations/flicker emissionsIEC 61000-3-3 | Not applicable | |
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that it is used in such an environment. | |||
| Immunity test | IEC 60601 test level | Compliance level | Electromagnetic environment - guidance |
| Electrostatic discharge (ESD) | ±6 kV contact | ±6 kV contact | Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30 %. |
| IEC 61000-4-2 | ±8 kV air | ±8 kV air | |
| Power frequency (50/60 Hz) magnetic field | 3 A/m | 3 A/m | Power frequency magnetic fields should be at levels characteristic of a typical location in a typical commercial or hospital environment. |
| IEC 61000-4-8 | |||
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that it is used in such an environment. | |||
| Immunity test IEC 60601 test level | Compliance level | Electromagnetic environment - guidance | |
| Conducted RF | 3 Vrms | Not Applicable | Portable and mobile RF communications equipment should be used no closer to any part of the device, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter. |
| IEC 61000-4-6 | 150 kHz to 80 MHz | Recommended separation distance | |
| Radiated RF | 3 V/m | 3 V/m | d = 1,2 √P 80 MHz to 800 MHz |
| IEC 61000-4-3 | 80 MHz to 2,5 GHz | d = 1,2 √P 800 MHz to 2,5 GHz | |
| where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and d is the recommended separation distance in metres (m). | |||
| Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey, should be less than the compliance level in each frequency range. | |||
| Interference may occur in the vicinity of equipment marked with the following symbol: | |||
| NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies. NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||
| a Field strengths from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the device is used exceeds the applicable RF compliance level above, the device should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as reorienting or relocating the device. b Over the frequency range 150 kHz to 80 MHz, field strengths should be less than 3 V/m. | |||
| Recommended separation distances between portable and mobile RF communications equipment and the device | |||
| The device is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the device can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and the device as recommended below, according to the maximum output power of the communications equipment. | |||
| Rated maximum output power of transmitterW | Separation distance according to frequency of transmitterm | ||
| 150 kHz to 80 MHzNot applicable | 80 MHz to 800 MHzd=1,2 √p | 800 MHz to 2,5 GHzd=2,3 √p | |
| 0,01 | Not applicable | 0,12 | 0,23 |
| 0,1 | Not applicable | 0,38 | 0,73 |
| 1 | Not applicable | 1,2 | 2,3 |
| 10 | Not applicable | 3,8 | 7,3 |
| 100 | Not applicable | 12 | 23 |
| For transmitters rated at a maximum output power not listed above, the recommended separation distance d in metres (m) can be estimated using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer. NOTE 1 At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies. NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||
TOPCOM
BD-4600
visit our websitewww.tristar.eu