Duo Sport 2 - Connected fitness device BLUETENS - Free user manual and instructions
Find the device manual for free Duo Sport 2 BLUETENS in PDF.
| Device type | CE medically certified electrostimulator (TÜV SÜD) with dual TENS (pain relief) and EMS (muscle) technology |
|---|---|
| Box contents | 2 Duo Sport 2 devices, 2 dual-output electrode cables 25 cm, 1 USB-A to USB-C cable 80 cm, DS2 electrode pack (4 large 100x50 mm + 8 small 50x50 mm), black soft pouch |
| Programs | 3 categories: Cure (pain relief via Gate Control and Burst modes), Relax (massage and post-workout recovery), Tone (physical preparation and muscle strengthening) |
| Split function | Run two different programs simultaneously across both devices - treat two areas or two objectives at the same time |
| Intensity | 60 levels (4 quarters of 15 levels each), adjustable via +/- buttons on the device or in the app |
| Electrical specifications | Frequency: 1 to 1,200 Hz - Output pulse: 24 to 400 µS - Voltage: 0 to 120 V (at 500 Ω) - Max power: 10 mW |
| Battery and charging | 3.7 V / 500 mAh lithium-ion polymer battery - USB-C charging 5 V/500 mA in approx. 1 hour - Orange indicator below 20% - Recharges approx. once a month with use every 2 days |
| Weight and dimensions | 33 g per device - 50.8 x 51.5 x 18 mm |
| Protection and longevity | IP22 protection rating - Minimum lifespan: 5 years - Software version V3.3 |
| Mobile app | Bluetens app on iOS 13+ and Android 10+ - Automatic Bluetooth connection - Personalised programs, guided treatments and Premium function (health professional plan within 48 h) |
| Electrodes | G607 biocompatible gel (ISO 10993-5 and ISO 10993-10 compliant) - Lifespan: 30 to 50 uses - Non-woven fabric backing - Apply to clean, dry skin |
| Warranty and support | 2 years on the device (excluding electrodes, cables and accessories) - Email support at cs@bluetens.com, response within 24 h (excluding weekends and public holidays) |
Frequently Asked Questions - Duo Sport 2 BLUETENS
User questions about Duo Sport 2 BLUETENS
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Connected fitness device in PDF format for free! Find your manual Duo Sport 2 - BLUETENS and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. Duo Sport 2 by BLUETENS.
USER MANUAL Duo Sport 2 BLUETENS
natural_image
Blue circular icon with a black open book symbol (no text or symbols)DUO SPORT

USER MANUAL
MANUEL UTILISATEUR - BENUTZERHANDBUCH
MANUAL DE USUARIO
Product model: DS2
Version: V1.3

Bluetens France
5 passage Saint Bernard 75011 Paris, France.
Bluetens limited
Uni. G. 15/F. TAL Building, 49 Austin Road, Kowloon, Hong Kong
Contact:cs@bluctens.com
S4M EUROPE SAS EC REP
- rue Castellion 0100 Oyornex FRANCE regulation@0x4m europe.com
English 4
Français 23
Deutsch 42
Español 61
Other languages 87
Electrode placements 80
Description of currents output 82
Electromagnetic compatibility 84
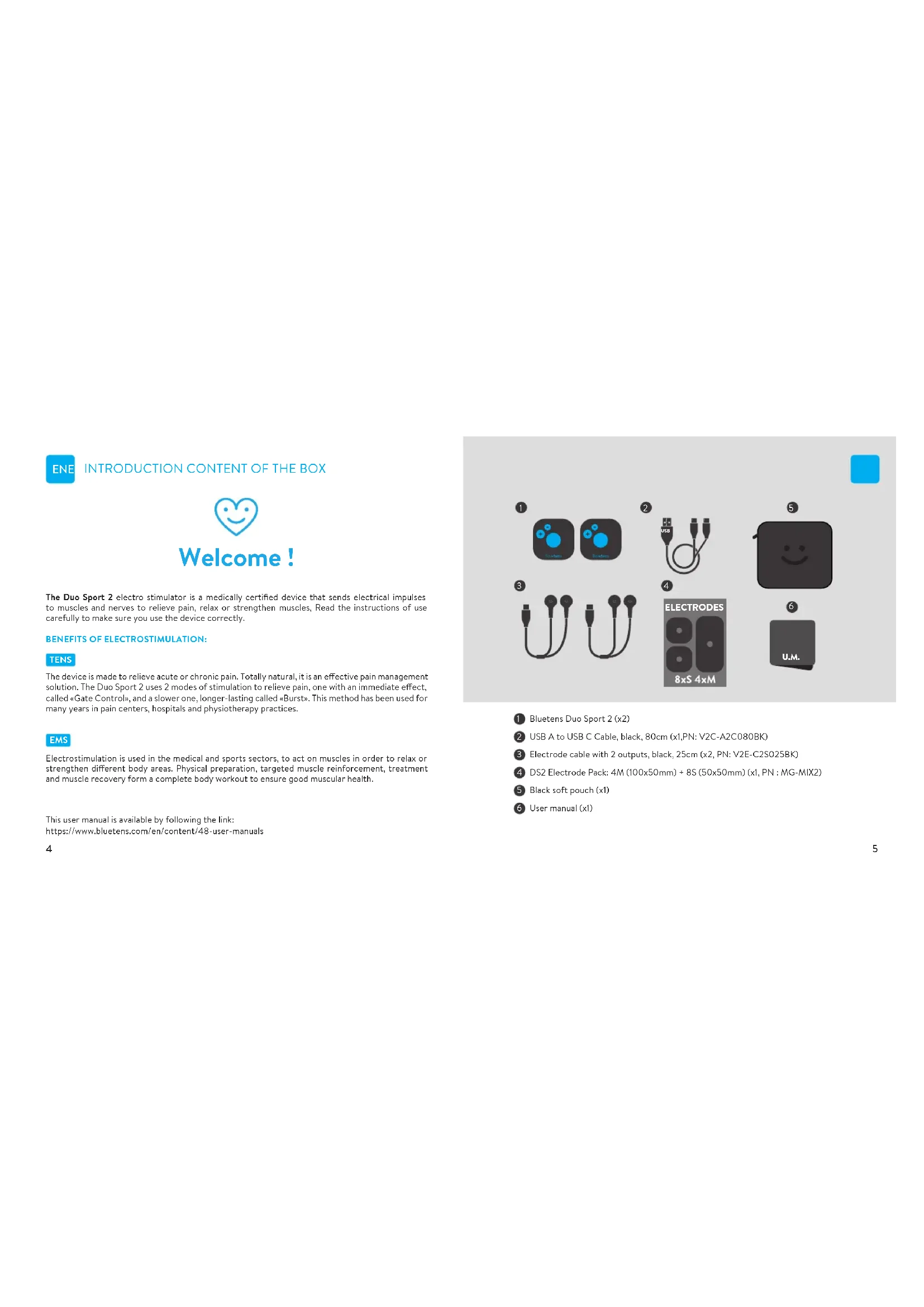
ENE INTRODUCTION CONTENT OF THE BOX

Welcome!
The Duo Sport 2 electro stimulator is a medically certified device that sends electrical impulses to muscles and nerves to relieve pain, relax or strengthen muscles. Read the instructions of use carefully to make sure you use the device correctly.
BENEFITS OF ELECTROSTIMULATION:
TENS
The device is made to relieve acute or chronic pain. Totally natural, it is an effective pain management solution. The Duo Sport 2 uses 2 modes of stimulation to relieve pain, one with an immediate effect, called «Gate Control», and a slower one, longer-lasting called «Burst». This method has been used for many years in pain centers, hospitals and physiotherapy practices.
EMS
Electrostimulation is used in the medical and sports sectors, to act on muscles in order to relax or strengthen different body areas. Physical preparation, targeted muscle reinforcement, treatment and muscle recovery form a complete body workout to ensure good muscular health.
This user manual is available by following the link:
https://www.bluetens.com/en/content/48-user-manuals
1

2

5

3

4

6

1 Bluetens Duo Sport 2 (x2)
2 USB A to USB C Cable, black, 80cm (x1,PN: V2C-A2C080BK)
3 Electrode cable with 2 outputs, black, 25cm (x2, PN: V2E-C2S025BK)
4 DS2 Electrode Pack: 4M (100x50mm) + 8S (50x50mm) (x1, PN : MG-MIX2)
⑤ Black soft pouch (x1)
6 User manual (x1)
ENE
SAFETY WARNINGSSF

MAIN DEVICE:
- The electrode is treated as a BF-type applied part.
• Device name: Duo Sport 2 - Product model: DS2
• Weight: 33 grams
• Battery voltage: 3.7V 500mAh lithium-ion polymer battery
• USB charging: Type C 5V/500mA
• Frequency: 1Hz to 1200Hz - Output pulse: 24 to 400uS
• Output voltage: 0V to 120V (at 500Ω load)
• Output power: 10mW (r.m.s) MAX - Operating environment:
Temperature: 5°C to 35°C
Humidity: 30%RH to 80%RH
Atmospheric pressure: 700 to 1060hPa - Storage environment:
Storage temperature: 5°C to 25°C
Humidity: 30%RH to 80%RH
Atmospheric pressure: 700 to 1060hPa
• Product dimensions: 50.8mm X 51.5mm X 18mm
• Minimum lifespan: 5 years
• Software version: V3.3 - Product dimensions: 50.8mm X 51.5mm X 18mm
- Minimum lifespan: 5 years
- Software version: V3.3
ELECTRODES:
- Support material: non-woven fabric
- Safety: Biocompatibility of the G607 gel used in this product is compliant with ISO 10993-5, ISO 10993-10 standards
- Lifespan: sealed storage for 3 years, to be stored at room or cool temperature (5°C to 25°C) and protected from direct sunlight.
CONTRAINDICATIONS
-
Do not use this device if you have a pacemaker, implanted defibrillator, or any other implanted metallic or electronic device. Such use could cause electric shock, burns, electrical interference, or death.
-
The device should not be used when cancerous or other lesions are present in the treatment area.
-
Stimulation should not be applied over open wounds, rashes, or swollen, red, infected, or inflamed areas (e.g., phlebitis, thrombophlebitis, varicose veins, obliterative arteriosclerosis, etc.).
-
Electrode placements should be avoided in the carotid sinus region (front of the neck) or transcranially (across the head).
-
Anxious users - the use of electrical stimulation requires user cooperation; therefore, the procedure should not be attempted in users with communication disabilities or mental incapacity.
-
Users with cerebrovascular issues - individuals with a history of aneurysm, stroke, and transient ischemia should not undergo electrical stimulation, as it stimulates peripheral blood flow, which could be fatal in such cases.
-
Epileptic users - Electrical stimulation impulses have the potential to trigger a seizure.
-
Cases of acute pain/pain of unknown origin - using an electrical stimulator in undiagnosed cases may hinder diagnosis.
-
Acute inflammation and swelling of the genitourinary system, tumors, urinary stones, history of nervous system diseases, and other illnesses.
-
Do not use during pregnancy, especially during the first trimester.
-
Presence of a severe systemic disease, such as scleroderma, diabetes mellitus with severe complications, myopathy, multiple sclerosis, Parkinson's disease, myelopathy, or serious diseases of the liver, lungs, kidneys, hematological system, or other comorbidities.
WARNINGS
The use of the device should be discontinued in the following circumstances:
-
If you feel unwell or uncomfortable, stop immediately. If there are any abnormalities in the skin or body, consult your doctor and follow their instructions.
-
If you wish to move the electrode to other parts of the body during operation, please turn off the power before moving it. Failure to do so may result in an electric shock.
-
Do not use this device while wearing other electronic devices.
This could cause malfunction of these devices (e.g., malfunction of an electronic watch).
ENE
- As subcutaneous use presents a high risk to humans or other devices, the use of this device is prohibited.
- Use under supervision mandatory for children (over 7 years old) and disabled persons. Risk of accident or discomfort.
- Do not use this appliance in a bathroom or a place with high humidity. Do not use while bathing or showering. Strong electric shock could occur.
- Do not use while sleeping. This could damage the product, and the displacement of the electrode could cause discomfort.
- Do not use while driving. It could cause an accident due to the strong stimulation.
- Do not keep the electrodes on your body all the time. This could cause skin irritation.
- Do not use metal to touch the electrode. This could cause an electric shock.
- Application near the thoracic region could cause cardiac discomfort. Seek the advice of a doctor beforehand.
- If in doubt, consult your doctor.
- Discontinue use and do not increase the intensity level if you experience discomfort during use.
- The electrode cable must not encircle your neck to avoid strangulation.
- Do not modify the equipment. Please use original accessories, it will cause an unusable effect if you use third party accessories on the market.
- Keep out of reach of children to avoid inhalation or ingestion of small parts.
PRECAUTIONS FOR USE
-
If you have a skin allergy, you should stop using it immediately.
-
Stimulation must not be applied through or over the head, directly to the eyes, covering the mouth or on the front of the neck (especially the carotid sinus).
SIDE EFFECTS
- Users may experience skin irritation and a burning sensation under the stimulation electrodes applied to the skin.
- Users may experience pain or discomfort during or after application.
Note: Users should stop using the device and consult their doctor if they experience any adverse reactions.
GETTING STARTED

THE DEVICE
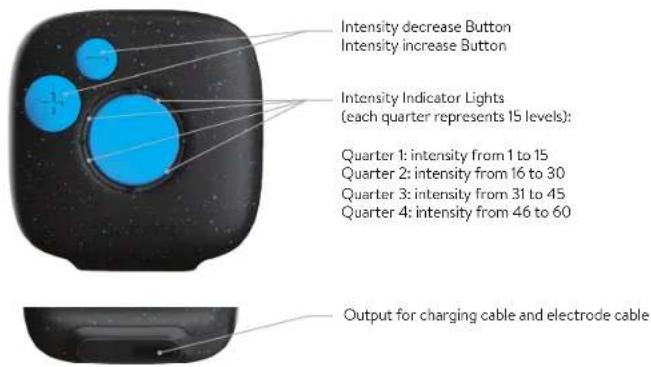
The Duo Sport 2 turns on and off by pressing the blue button in the center of the device. Here is an explanation of the inputs, outputs, and buttons of the device.
text_image
Intensity decrease Button Intensity increase Button Intensity Indicator Lights (each quarter represents 15 levels): Quarter 1: intensity from 1 to 15 Quarter 2: intensity from 16 to 30 Quarter 3: intensity from 31 to 45 Quarter 4: intensity from 46 to 60 Output for charging cable and electrode cableENE
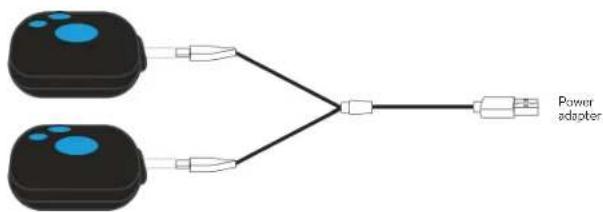
CHARGING THE BATTERY
The Duo Sport 2 is charged using the USB-C cable included in the box.
Charging time is approximately 1 hour.
When the battery is below 20%, the charge indicator light flashes orange.
This indicator light will remain orange during the charging time and will go out once the
battery is full.
We recommend charging the device with its USB-C cable connected to a computer.
If you wish to use a mains adapter, please ensure that the power is compatible.
flowchart
graph TD
A["Two devices"] --> C["Power adapter"]
B["Two devices"] --> C["Power adapter"]
The battery recharges on average once a month, if the device is used once every 2 days.
Note:
• This device cannot be used while charging.
- The mains adapter must comply with standard IEC62368-1, respect at least 2 MOOP and have an output of 5V/1A.
CONNECTING THE ELECTRODES
Here's how to connect the electrodes to the device:

flowchart
graph TD
A["Device with blue eye icon"] --> B["Sensor"]
C["Sensor"] --> D["Sensor"]
D --> E["Sensor"]
style A fill:#000,stroke:#fff,color:#fff
style C fill:#000,stroke:#fff,color:#fff
style B fill:#000,stroke:#fff,color:#fff
style D fill:#000,stroke:#fff,color:#fff
style E fill:#000,stroke:#fff,color:#fff
ENE THE APPLICATION
DOWNLOAD AND INSTALLATION
The Bluetens Application is available on Google Play and Appstore.

The Application is compatible with Smartphones and Tablets:
iPhone : iOS 13 and above
Android : Android 10 and above
(Modified Android versions may make the Application incompatible)
Download and install the Application according to your Smartphone.
Notes:
Bluetooth must be activated to use the Application.
Android smartphones must also authorise access to the location service, to the Application.
The Bluetens Application does not have access to any data. This condition is required by Android versions to stabilise Bluetooth.
text_image
BlueTens SIGN UP WITH FACEBOOK SIGN UP WITH GOOGLE SUBSCRIBE You already have a count! CONNECTION >I will register laterFIRST USE
Launch the Application and select your device from the available options.
Log in using the options provided or create an account.
A tutorial will introduce you to the Application and its features.
ENE
text_image
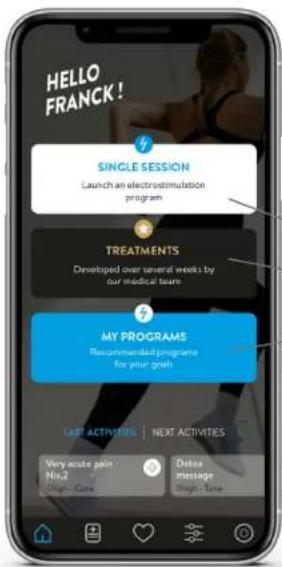
HELLO FRANCK! SINGLE SESSION Launch an electrostimulation program TREATMENTS Developed over several weeks by our medical teams. MY PROGRAMS Recommended programs for your goal LAST ACTIVITIES | NEXT ACTIVITIES Very acute pain Niv2 High - Care Delta message High - TubePRESENTATION AND USE
Choose from the main functions on the home page:
SIMPLE ELECTROSTIMULATION SESSION
TREATMENTS: generic treatments for common pathologies
MY PROGRAMS: recommended programmes to achieve the goals you have set.
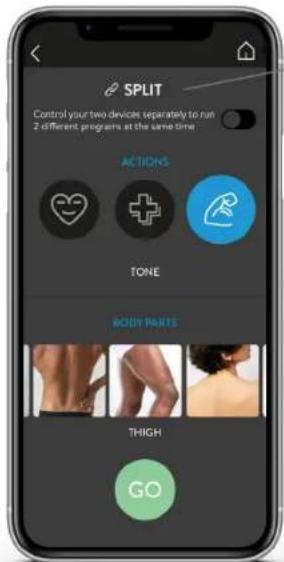
text_image
SPLIT Control your two devices separately to run 2 different programs at the same time ACTIONS TONE BODY PARTS THIGH GOSINGLE SESSION: START A PROGRAM
SPLIT FUNCTION
Activate the Split function to run two different programs simultaneously.
Select the action and the body part

CURE
Different programmes to relieve all types of pain.

RELAX
Muscle massage programmes and post-physical exertion recovery.

TONE
Physical preparation and muscle strengthening programmes.
Choose the body parts according to your needs.
Press «GO» to access the list of programmes.
ENE
PROGRAM DESCRIPTION
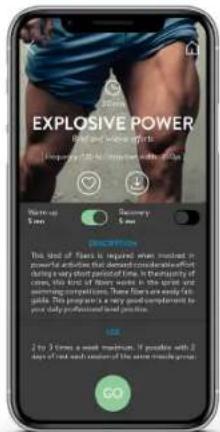
text_image
EXPLOSIVE POWER Burst and double spirits Frequency: 100 gm / 500 gm width: 5.0 gm Start-up 5 mm Discovery 5 mm ORDER: This kind of fires is required when involved in powerful activities that demand consideration of during a very short period of time. In the vicinity of cases, this kind of fires works in the sport and will be replaced by the same time. There are two weekly intervals. This year's one-year season is completed by your daily professional level position. 2 to 3 times a week maximum. If possible with 2 days of last week compared to the some seconds ago GOELECTRODES PLACEMENT
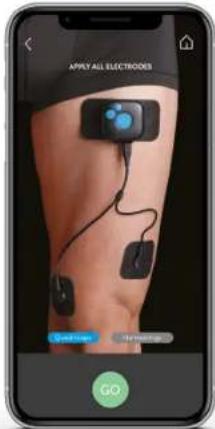
text_image
APPLY ALL ELECTRODES Goodwill Help GOTURN YOUR
DEVICE ON

text_image
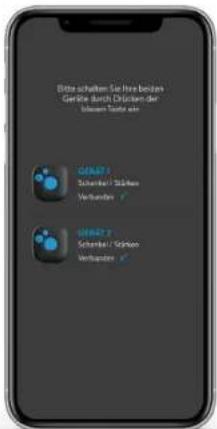
Please turn your two devices on: By pressing the blue button. DEVICE 1 High Time Connected ✓ DEVICE 2 High Time Connected ✓
The connection between the Application and the device is established automatically at this stage. Do not pair manually!
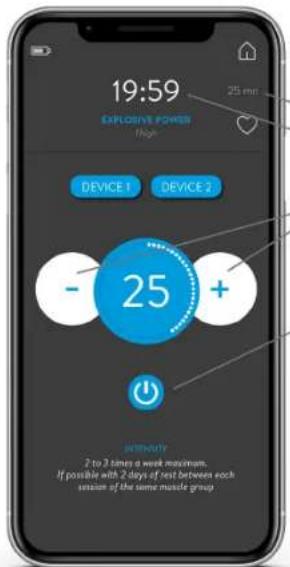
text_image
19:59 EXPLOSIVE POWER High DEVICE 1 DEVICE 2 - 25 + INTELLY 2 to 3 times a week maximum. If possible with 2 days of test between each session of the same muscle groupRUNNING PROGRAM
TOTAL PROGRAMME DURATION
REMAINING TIME COUNTDOWN
The default intensity is 1, press + or - to change it. The maximum intensity is 60. The intensity can also be set directly on the device.
PRESS THIS BUTTON TO STOP THE PROGRAM
Note:
Once your session is over, make sure you switch off the device before handling the electrodes. Replace them on the plastic (indicated side) and store them in a cool, dry place.
ENE
PREMIUM FUNCTION

PERSONALISED SUPPORT TO HELP YOU REACH YOUR SPORT OR HEALTH OBJECTIVES
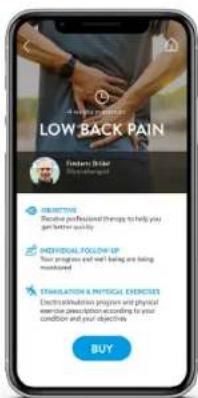
text_image
LOW BACK PAIN Frequent Binge Buy up an offer ESERTORT Receive professional things to help you get better quality NECESSICAL FOLLOW UP Your progress and wait being are being monitored EMILIZATION & PHYSICAL EXERCISES Electricity, administration program and physical exercise prescription according to your condition and your objectives BUY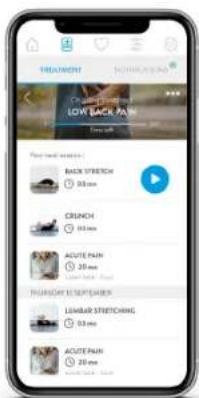
text_image
TREATMENT NICKLE BAN LOW BACK PAIN Back Stretch 0.0 mm CRENCH 0.0 mm ACUTE PAIN 20 mm LEOBAR STRETCHING 0.0 mm ACUTE PAIN 20 mmSelect or describe your problem, then fill in your profile and proceed to payment. Within 48 hours you will receive your personalised treatment to follow for a minimum of 4 weeks (electrostimulation and physical exercises) and follow the prescriptions to get better quickly. The healthcare professional will make sure that your exercises are carried out properly and that you feel good.
CARE INSTRUCTIONS

MAIN DEVICE
- Remove the cables from the device after use.
• Use a soft cloth to clean dirt on the device - Use a damp cloth with a neutral detergent or alcohol to clean if necessary.
- Do not use thinner or petrol to clean.
- Do not use alcohol on the back of the device
- Do not use any other detergents.
- Keep water or liquids away from the device
- Keep out of direct sunlight, heat, damp and dusty places.
ELECTRODES
- Do not use extra gel with the electrodes.
• Always replace and store the electrodes on the correct side of the plastic provided. - Use a little water to clean the electrode if necessary.
- Do not wash.
- Do not wipe the electrode with a cloth or paper.
• The lifespan of the electrodes varies between 30 and 50 uses depending on the quality of the skin, hairiness, perspiration and the care taken. - Electrodes are consumables; when they no longer adhere to the skin, it's a sign that they need changing.
- Do not stack or bend the electrodes.
- Do not touch the gel with your fingers.
- Apply to clean, dry skin, without cream or oil.
ENE
RECYCLINGIN

Essential informations for a correct use is indicated by the corresponding symbols. The following symbols can be seen on the device and on the labelling.
| SYMBOL TITLE | |
| IP22 | IP code of the device: the degree of protection of this device against the intrusion of solid foreign objects -- ≤ 12.5 mm in diameter (and against access to dangerous parts with a finger); the degree of impermeability is of the drip type (inclination of 150). |
| Disposal in compliance with directive 2002/96/EC (WEEE) | |
| This side up | |
| Date of manufacture | |
| Batch code | |
| CE0123 | CE mark, Certificate issued by TUV SUD. |
| Manufacturer | |
| Type BF applied part | |
| Warning: to avoid the risk of injury, read the manufacturer's instructions carefully. | |
| Symbol for «AUTHORISED REPRESENTATIVE IN THE EUROPEAN COMMUNITY» | |
| Fragile, handle with care | |
| Store the product in a dry place. Protect from water and rain. | |
| Product packaging is recyclable | |
| Importer | |
| Medical device | |
20 21
The product must not be disposed of with household waste.
Consumers are legally obliged to dispose of electrical equipment at a collection point in their town.
Contact your local council for more information or a retailer.
EN
INTRODUCTIONTR FR LE
DO YOU NEED HELP?
Our customer service team deals directly with all requests for advice or after-sales service. Our agents are fast and available by e-mail at cs@bluetens.com. We'll get back to you within a day (excluding weekends and public holidays).
Online troubleshooting:
https://www.bluetens.com/en/content/28-support-bluetens
The DS2 is guaranteed for two years. This guarantee does not cover damage caused by improper maintenance or use that does not comply with the relevant documentation, accidents (impact, fire, liquid), modifications, repairs, misuse or normal wear and tear. This warranty covers defects in materials or workmanship. This warranty only covers products purchased directly from Bluetens or an authorized Bluetens reseller.
In the event of a defect covered by the warranty, Bluetens will send you a return slip and will be responsible for the repair or exchange of your device as well as its return.
WARRANTY PERIOD
Your device is guaranteed for 2 years from the date of receipt of the product if purchased online, or from the date of purchase if purchased from a retailer.
NOT COVERED BY THE WARRANTY
Electrodes, cables and accessories included in the box are not covered by this warranty.
WARRANTY DISCLAIMER
Bluetens has not authorised any agency to repair its products. If you experience any problems with your device, please contact us. Bluetens will not be responsible for the results of any repair. The intervention of a third party on our products will void the 2-year warranty. The use of third-party electrodes or accessories could cause the device to malfunction and will void the warranty. The user should not attempt to repair the device or its accessories by himself. Each device has been inspected through systematic validation. The performance is stable and does not require calibration or validation. If your product does not achieve the expected performance and the basic function changes during normal use, please contact us. Please note that Bluetens will not be responsible for quality issues caused by improper use.

Bienvenue!
natural_image
Abstract black square with blue circular elements and radiating lines (no text or symbols)text_image
PLACE YOUR ITEMS ELECTROSES GOALLUMEZ LES DUO SPORT 2
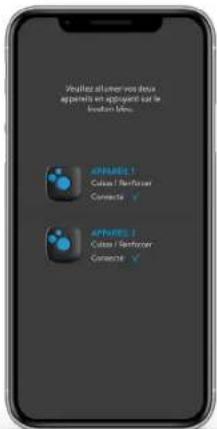
text_image
Analyze all other you do you apparents are applicable but be location time.
text_image
Diagram showing six labeled electronic devices and a battery pack with text labels like 'ELEKTRODEN' and 'ANLEITUNG'.natural_image
Abstract black square with blue circular elements and white lines radiating from center (no text or symbols)SCHALTEN SIE
IHR GERÄT EIN
text_image
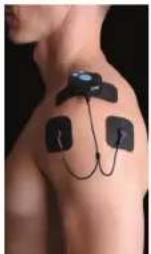
PODER EXPLOSIVO Engagement: 100% Cataleromotive 5 mm Sensitration 5 mm 2018SPOTCH This kind of fibers is requested when invested it powerful activities that demand remarkable effort during the many months of course. It is mainly on some, this kind of fibers works in the open and working environment. They learn to be ready for use. The progress is a very strong commitment to your daily professional team activity. STUALER 3 to 3 hours a week maximum. If possible with 2 days of next session of the same muscle group. GOHere is an overview of the possible electrode placements for the different cables and electrodes, on various parts of the body.
Épaule/Schulter/Hombro
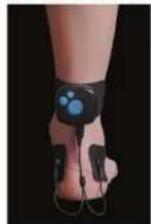
ANKLE
Cheville/Knöchel/Tobilio
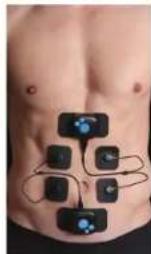
ABS
Abdos/Bauch/Abdominales
CALVES
Mollets/Waden/Pantorrillas

LOWER ABDOMEN
Bas du ventre/Unterbauch/Parte inferior del abdomen

FIND ALL THE ELECTRODE PLACEMENTS IN THE APPLICATION
RETROUVEZ L'INTÉGRALITÉ DES PLACEMENTS DES ÉLECTRODES DANS L'APPLICATION
ALLE ELEKTRODENPOSITIONEN FINDEN SIE IN DER APP
Instant electric voltage/current when using Electrodes:
Load 500Ω: MAX 120V / MAX 240mA
Load 1000Ω: MAX 150V / MAX 150mA
TENS
| N° PROGRAM OUTPUT PARAMETERS | ||
| 1 Acute Pain Frequency: 80Hz - Impulse: 150μs - Treatment time: 20 minutes | ||
| 2 | Very acute pain | Frequency: 100Hz - Impulse: 250μs - Treatment time: 20 minutes |
| 3 Cervical Pain Frequency: 2Hz - Impulse: 200μs - Treatment time: 20 minutes | ||
| 4 | Intense recovery 1 | Frequency: 2-4-6-8-10-9-7-5-3-1 Hz - Each step 3 minutesImpulse: 150μs - Treatment time: 30 minutes |
| 5 Endorphin 1 Frequency: 5Hz - Impulse: 250μs - Treatment time: 30 minutes | ||
| 6 Chronic pain 1 Frequency: 80Hz - Impulse: 200μs - Treatment time: 60~120 minutes | ||
| 7 | Decontracting massage | Frequency: 1Hz - Impulse: 150μs - Treatment time: 30minutes |
| 8 | Active recovery 1 | Frequency: 10-9-8-7-6-5-4-3-2-1 Hz - Each step 3 minutesImpulse: 150μs - Treatment time: 30 minutes |
EMS
N° PROGRAM OUTPUT PARAMETERS
| 1 | Strengthening n°1 | Warm-up: Frequency: 8Hz - Impulse: 150μs 5 minutesTraining: Frequency variable - Impulse: 150μs8Hz~50Hz Inc 1.5 seconds - 60Hz Hold 6 seconds60Hz~8Hz Dec 1.5 second - 8Hz Hold 6 SecondsPhase time est: 15 second - Loop for: 25 minutesCool-down: Frequency: 3Hz Impulse: 300μs 5 minutesTreatment time: 35 minutes |
| 2 Strength 2s | Sequence: Frequency: 50 Hz - Impulse variableImpulse: 50~250μs inc 1.5 secondsImpulse: 250μs hold 2 secondsImpulse: 200~65μs dec 1.5 secondsPause: 4 seconds - Loop for: 20 minutes | |
| 3 | Muscle sharpening 1 | Warm-up: Frequency: 6Hz - Impulse: 150μs 5 minutesTraining: Frequency variable - Impulse: 150μs6Hz~30Hz Inc 4 seconds - 30Hz Hold 10 seconds30Hz~4Hz Dec 1 second - 4Hz Hold 6 SecondsPhase time est: 21 second - Loop for: 25 minutesCool-down: Frequency: 3Hz - Impulse: 300μs 5 minutesTreatment time: 40 minutes |
| 4 Muscle tone 1 | Warm-up: Frequency: 100Hz - Impulse: 150μs 5 minutesTraining: Frequency: 4Hz~20Hz~4Hz Inc/Dec step by 1Hz Impuse 150Phase time est: 22.5 second - Loop for: 20 minutesCool-down: Frequency: 3Hz - Impulse: 150μs 5 minutesTreatment time: 30 minutes | |
| 5 | Prevention muscle loss 1 | Frequency variable - Impulse: 200μs15Hz~80Hz Inc 2.2 seconds - 80Hz Hold 10 seconds80Hz~15Hz Dec 1.3 second - 15Hz Hold 8 SecondsPhase time est: 21.5 second - Loop for: 30 minutes |
ELECTROMAGNETIC COMPATIBILITY
The device has been thoroughly tested and inspected to assure proper performance and operation.
The EMC Test Report Number of TUV: 68.730.20.0074.01
| Configuration Description | |
| AC Powered 230VAC | 50Hz |
| AC Powered | 100VAC/50Hz & 240VAC/50Hz(Only for Voltage dips and Voltage interruptions) |
OUTPUT PARAMETERS
| Output Frequency | 1Hz | 10Hz | 50Hz | 100Hz | 200Hz | 500Hz | 1KHz | 1,2KHz |
| Output Pulse 400uS | 300uS 100uS | 100uS 500uS | 50uS 50uS | 34uS 24uS | ||||
| Output Voltage Vpp | 0-120V 0-120V 0-10V | 0-85V | 0-85V 0-57V | 0-57V 0-57V | ||||
MODES OF OPERATION:
| Mode Description | |
| Charging | The EUT's USB-C port is connected to a single-port USB-C cable (length: 80 cm) and is charged by an AC / DC adapter. |
| EMS + Lien BT | The EUT is powered by a rechargeable 3.7VDC lithium battery, Its USB-C port is connected to a double electrode cable (4 ends, length: 110 cm), and the electrode cable terminals are connected to electrode pads (Size: 50 x 100 mm). The part of the electrode cable 40 cm away from the EUT is immersed in 0.9% saline solution. The EUT is connected to the APP via Bluetooth, and its treatment functions are controlled by the APP or by the EUT itself. Next, the EUT is set to Nev.1 Acute Pain mode (Pain) and set to maximum power for the operation. |
| TENS + Lien BT | The EUT is powered by a rechargeable 3.7VDC lithium battery, Its USB-C port is connected to a dual-electrode cable (4 ends, length: 110 cm), and the electrode cable terminals are connected to electrode pads (Size: 50 x 100 mm). The part of the electrode cable that is 40 cm away from the EUT is placed in 0.9% saline solution. The EUT is connected to the APP via Bluetooth, and its processing functions are controlled by the APP or by itself. Next, the EUT is set to TENS mode and adjusted to maximum power for operation. |
EMISSION TESTS EN 60601-1-2:2015, IEC 60601-1-2:2014
| Test Condition: Group1, Class B | Test Result |
| Radiated Emission 30MHz to 1000MHz | Pass |
| Conducted Emission on AC 150kHz to 30MHz | Pass |
| Harmonic Class A | Pass |
| Flicker | Pass |
IMMUNITY TESTS EN 60601-1-2:2015, IEC 60601-1-2:2014
| Test Condition: Group1, Class B Test Result | |
| Electrostatic Discharge (IEC 61000-4-2) ± 2kV ± 4kV ± 6kV ± 8kV ± 15kV | Contact: ± 8kV Air: ± 2kV ± 4kV ± 8kV ± 15kV |
| Radiated Immunity (IEC 61000-4-3) 80MHz to 2700MHz 10V/m385MHz 27V/m450MHz 28V/m710MHz, 745MHz, 780MHz 9V/m810MHz, 870MHz, 930MHz 28V/m1720MHz, 1845MHz, 1970MHz 28V/m2450MHz 28V/m5240MHz, 5500MHz, 5785MHz 9V/m | 10V/m, 80% Am at 1kHz27V/m PM at 18Hz28V/m FM ± 5 kHz deviation at 1kHz sine9V/m PM at 217 Hz28V/m PM at 18Hz28V/m PM at 217 Hz28V/m PM at 217 Hz9V/m PM at 217 Hz |
| Electrical Fast Transient (IEC 61000-4-4)For AC Power port: ± 2kV , 100 kHz repetition frequencyFor signal input/output port: ± 1kV , 100 kHz repetition frequency | For AC port:Power supply lines: ± 2kV |
| Surge (IEC 61000-4-5)For AC Power port: ± 0.5kV , ± 1kV (Line to line) ± 0.5kV , ± 1kV , ± 2kV (Line to Ground)For signal input/output port: ± 2kV (Line to Ground) | For AC port:(L-N): ± 0.5kV ± 1kV |
| Conducted Immunity (IEC 61000-4-6)For AC Power port, patient coupling port and signal input/output port:150KHz to 80MHz 3V (80% Am at 1kHz)ISM and amateur radio bands between 150KHz to 80MHz 6V (80% Am at 1kHz) | For AC port and patient coupling port: 150KHz to 80MHz 3V (80% Am at 1kHz) ISM and amateur radio bands between 150KHz to 80MHz 6V (80% Am at 1kHz) |
| Voltage Dips and Interruption (IEC 61000-4-11)0%, 70%, 0% of UT | For AC power port:0% for 0.5 cycle (at 0°, 45°, 90°, 135°,180°, 225°, 270° and 315°), 0% for 1 cycle (at 0°), 70% for 25 cycles (at 0°), 0% for 250 cycles |
| Power Frequency Magnetic Field (IEC 61000-4-8) 50Hz, 60Hz 30A/m | 50Hz: 30A/m60Hz: 30A/m |
OTHER LANGUAGES

THIS USER MANUAL IS AVAILABLE IN OTHER LANGUAGES AT THE FOLLOWING LINK:
https://www.bluetens.com/en/content/48-user-manuals

Take care of yourself
Prenez soin de vous
Achten Sie auf sich
Cuídese
www.bluetens.com




