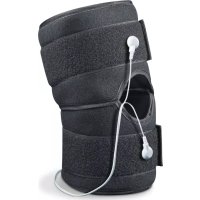
EM 34 - Pain therapy device BEURER - Free user manual and instructions
Find the device manual for free EM 34 BEURER in PDF.
Download the instructions for your Pain therapy device in PDF format for free! Find your manual EM 34 - BEURER and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. EM 34 by BEURER.
USER MANUAL EM 34 BEURER
Signs and symbols The following signs appear in the Safety Section (pages 2, 3, 5) and in this manual on pages 6, 10. READ THIS ENTIRE MANUAL, THE SAFETY SECTION
AND ALL INSTRUCTIONS AND WARNINGS COMPLETE-
LY AND CAREFULLY BEFORE USING THIS PRODUCT.
FOLLOW ALL SAFETY INSTRUCTIONS AND WARNINGS
TO AVOID HAZARDOUS SITUATIONS AND TO MAKE CORRECT USE OF THIS PRODUCT. ENGLISH Contents
9. Frequently Asked Questions ......................................12
This is the safety alert symbol. It alerts you to potential personal injury hazards. Obey all safety messages that follow this symbol to avoid pos
sible injury or death. WARNING WARNING indicates a hazardous situ- ation which, if not avoided, could re- sult in death or serious injury. CAUTION CAUTION indicates a hazardous situa- tion which, if not avoided, may result in minor or moderate injury. NOTICE NOTICE addresses practices not related to personal injury, such as product and/or property damage.3 WARNING To reduce the risk of fire, electric shock, or serious personal injury:
This device is for external use on human knees and elbows only. Do not use this device in the following situations:
If you have implanted electrical devices (e.g. a pace- maker).
If you have metal implants.
In combination with electronic life-sustaining devices, such as an artificial heart or lung.
If you have a fever above 102° F (39° C).
If you have cardiac arrhythmia or disorders of the heart’s impulse and conduction system.
After an operation, if muscle contractions could af- fect the healing process.
After consuming alcohol.
If you suffer from a seizure disorder (e.g. epilepsy).
If you are pregnant.
On acutely or chronically diseased (injured or irritat- ed) skin (e.g. inflamed skin - whether painful or not, reddened skin, rashes, e.g. allergies, burns, bruises, swellings, both open and healing wounds, and post- operative scars where the healing process could be affected).
On areas of skin that lack sensitivity.
In humid environments (e.g. in the bathroom) or when bathing or showering.
In the presence of electronic monitoring systems, such as heart monitors.
While asleep, driving a vehicle, or operating machin- ery.
While using other devices that transmit electrical im- pulses into your body.
Keep the device and accessories away from children. The device contains small parts that can be swallowed.
Store the device and accessories out of the reach of children. There is a risk of strangulation from the con- nection cable.
This unit is not intended for use by children or persons with physical, sensory (e.g. insensitivity to pain) or men- tal impairments or those who do not know how to use it due to a lack of experience or knowledge, unless they are supervised by a person responsible for their safe-4 ty or have received instructions from them on how to use the unit.
The device must not be used in combination with elec- tronic life-sustaining devices such as ventilators or other electronic medical products, such as electro
If your pain gets worse, becomes chronic, or persists for up to five days, contact your doctor.
Do not share this device with anyone else. This could lead to skin irritation or infection. The device is intended for private use only.
Do not move or use the cuff if the control unit is switched on.
Only connect the connection cable for the electrode plug to the intended control unit and the universal cuff that is included.
Do not pull on the connection cables during treatment.
Do not bend or pull on the ends of the connection cable.
Do not wear any electronic devices, such as watches, while using the device.
During treatment, do not touch the water contact elec- trodes on the universal cuff with your finger.
Do not use the device on a particular area more than three times a day with a 30-minute treatment period each time.
If the device does not work properly, or you feel unwell or experience pain, stop using it immediately.
Do not modify the electrodes as this is potentially haz- ardous (max. recommended output value for the elec- trodes is 58.1 mA/sq in (9 mA/cm²), an effective current intensity 12.9 mA/sq in (2 mA/cm²) requires increased awareness).
Do not use while undertaking any activity where an un- expected reaction (e.g. strong muscle contractions even at low intensity) could be dangerous.
Do not use during strenuous activity (e.g. sports).
Pre-treating the skin with greasy creams or lotions is not recommended.
At first, use the device while sitting or lying down to minimize the risk of accidental injuries as a result of feeling faint. If you feel faint, switch off the device im- mediately, lie down and support the legs in an elevated position (approx. 5 - 10 min).
Hold the device away from sources of heat and do not use it within three feet (approx. 1 m) of shortwave or microwave devices (e.g. mobile phones).
The device is intended for self-treatment only.
If there are signs of wear and tear or damage or if the device is used improperly, return it to the manufactur- er or retailer.5
Do not connect the electrodes to other devices.
Do not use in near flammable substances, gases, or explosives.
Do not attempt to open or repair this device; contact customer service for all repairs.
The cuff and the electronic characteristics of this device could influence the safety and effectiveness of the elec
trical stimulation. Do not use any other TENS devices or cuffs with this product.
Before using the device, consult your doctor if any of the following apply:
Serious illness, high blood pressure, a blood coagu- lation disorder, propensity to thromboembolic condi- tions or recurrent malignant growths.
Skin diseases or open wounds.
Unexplained chronic pain in any part of the body.
Any sensory impairment that reduces the feeling of pain (e.g. metabolic disorders).
If you are receiving medical treatment.
If you are already having stimulation treatment for ex- isting conditions.
If you suffer from persistently irritated skin due to long-term stimulation at the same electrode site. NOTICE:
o not expose the device to direct sunlight or high tem- peratures.
rotect the device from dust, dirt and humidity. Never immerse the device in water or other liquids. Battery Handling Safety Precautions
- U se only the size and type of batteries specified.
- B e sure to follow the correct polarity when installing the batteries. Reversed batteries may cause damage to the device.
- D o not mix different types of batteries together (e.g. Al- kaline and Carbon-zinc or rechargeable batteries) or old batteries with fresh ones. Always replace batteries as a simultaneous set.
- I f the batteries in the device are depleted or the device will not be used for a long period of time, remove the batteries to prevent damage or injury from possible bat- tery leakage.
- D o not try to recharge batteries not intended to be re- charged; they can overheat and rupture (follow battery manufacturer’s directions).
o not dispose of batteries in fire, batteries may ex- plode or leak.6
- C lean the battery contacts and also those of the device prior to battery installation.
emove discharged batteries from the product and dis- pose/recycle in compliance with all applicable laws.
eep batteries away from children and pets. Batteries may be harmful if swallowed. Should a child or pet swal- low a battery, seek medical assistance immediately.
2. Important information
Signs and symbols The following symbols are used in these instructions for use, on the packaging and on the type plate for the device and accessories: Caution Note Note on important information Follow instructions for use Type BF applied part (cuff only) Direct current Keep dry Pacemaker
3. Parts and Controls
1. Hook and Loop Strap
6. ▲ Button (increases intensity)
9. ▼ Button (decreases intensity)
10. Intensity Level Indicators
11. Illuminated Logo
13. Battery Compartment Cover8
The Knee and elbow TENS is a single-channel TENS de- vice and falls into the category of electrostimulation units. Gentle electrical pulses are sent via water contact elec- trodes through the skin to the nerve fibers. The innovative Knee and elbow TENS provides temporary pain relief in the knee and elbow area. TENS, or transcutaneous electrical nerve stimulation, relates to the electrical stimulation of the nerves via the skin. TENS is an effective nonpharmacological method of treating dif- ferent types of pain that have a variety of causes. It has no side-effects if administered correctly. The method has been clinically tested and approved and can be used for simple self-treatment. The pain-relieving or pain-suppressing effect is achieved by inhibiting the transference of pain to nerve fibers (caused mainly by high-frequency impulses) and by increasing the secretion of endorphins in the body. Their ef- fect on the central nervous system reduces the sensation of pain. The method is scientifically substantiated and ap- proved as a form of medical treatment. Any symptoms that could be relieved using TENS must be checked by your doctor. Your doctor will also give you in
structions on how to carry out a TENS self-treatment re- gime. The device is designed to be used for temporary relief for sore and aching muscles in the upper extremities (arms) and lower extremities (legs) due to strain from exercise or normal household and working activities.
1 x knee and elbow universal cuff 1 x control unit 1 x connection cable 3 x 1.5 V AAA battery LR03
Battery Installation/Replacement
. Remove the Battery Compartment Cover from the bot- tom of the control unit.
. Insert three AAA size alkaline batteries (included), ob- serving the correct polarity as indicated inside the bat- tery compartment.
. Reattach the Battery Compartment Cover.9 Connecting Components
. Snap the ends of the power wire onto the power wire connectors on the cuff. The two ends are inter- changeable and do not have a specific left or right.
. Moisten the rubber electrode cov- ers with water, as well as the area on your body to be treated.
. Place the cuff around your knee or elbow, as desired. Ensure the rub- ber electrode covers are properly positioned over the desired treat- ment area and the opening in the cuff is over your knee/elbow.
. Tighten and secure the hook and loop straps.
. Insert the power wire plug into the power wire jack on the control unit.
. Press the On/Off Button to switch the device on. The device will beep three times and the logo will flash and then illuminate.
. Use the ▲/▼ buttons to select an intensity level from
1 - 25 that feels most comfortable. The “low” intensity
indicator will flash for levels 1-8, the “middle” intensity indicator will flash for levels 9-17, and the “high” inten- sity indicator will flash for levels 18-25. The higher the level within each range, the faster the indicator will flash. NOTE: When setting the level, the control unit will beep once. If it beeps twice, there may not be enough water on the rubber electrode covers to conduct enough cur- rent, or the power wire is not attached properly.
. This device will switch between two treatment modes by itself. If you prefer one treatment mode over the other, you can manually select it by pressing the On/Off Button during treatment; the Indicator above the “M” will illumin- ate briefly, indicating that the manual selection mode is active. To return to auto mode, press the On/Off Button again; the indicator above the “A” will illuminate briefly.
. To shut the device off at any time, press and hold the On/Off Button until the control unit emits one long beep and the logo shuts off. It will also shut off by itself after 30 minutes of use.
. Always remove the cuff directly after treatment.10
7. Care and Maintenance
Before cleaning, disconnect the power wire from the device and from the cuff. Cleaning the Cuff Clean the cuff in warm soapy water and then rinse well, making sure all soap is washed out. Pat cuff dry with a towel and then let air dry completely before use. Storage To store the device, first ensure it is switched off. Discon- nect the power wire from the control unit and from the cuff. Place all parts back into their original packaging and store in a cool, dry place out of the reach of children.
8. Troubleshooting Guide
Problem Possible cause Solution The device does not switch on Batteries are depleted Replace the bat
teries Batteries are not inserted correctly Reinsert the bat
teries according to the polarity indica- tors inside the bat- tery compartment The elec
trical pulses are weak Cuff is not con- tacting skin prop- erly Reposition the cuff to ensure it con
tacts the skin suf- ficiently Electrodes are not wet enough Shut off the de
vice and remoisten electrodes11 Treatment is uncomfort
tioning Replace the cuff Intensity level set too high Lower intensity level Electrodes are not wet enough Shut off the de
functioning Lower intensity level and rotate the power wire plug 90°. If problem persists, replace the power wire Stimulation is ineffect
ive Electrodes pos- itioned incorrectly Remove elec- trodes and repos- ition, ensuring cuff has sufficient skin contact The skin turns red and/or a stabbing pain occurs The universal cuff is not positioned correctly on the skin Ensure that the universal cuff sits comfortably on the skin and cannot move The universal cuff is dirty Clean the universal cuff as described in these ins- truc
trodes has a scratch Replace the uni- versal cuff Water contact electrodes are not sufficiently moist Switch off the de
maturely Cuff has become too loose Stop treatment and retighten/re- position cuff Power wire is loose Stop treatment and reinsert power wire Batteries are de
pleted Replace batteries
9. Frequently Asked Questions
When should I use this device? Apply the cuff as soon as you feel pain in your knees/ elbows. If you begin using this device immediately, you can alleviate or even avoid chronic pain. The earlier you begin using the device, the higher the probability that pain can be alleviated. How often should I use the device? Do not use the device on a particular area more than three sessions per day, 30 minutes per session. Start the first treatment at low intensity until you are familiar with the de- vice. Stop treatment as soon as the pain has receded or disappeared. If, after using the device, you experience pain due to overexertion, stop use for two days. If you experience further pain due to overexertion, lower the intensity level during subsequent uses and reduce the treatment time. When should I stop using the device? Stop using the device if you feel a stabbing pain during stimulation. If you notice side-effects following use (skin irritation, reddening and burning of the skin, headache, feel- ing unwell), stop using the device. Stop using the device if pain does not improve after several treatments, becomes chronic or lasts for more than five days. What type of pain should I use the device for? The device works best to treat acute pain, meaning you can easily determine precisely where the pain is located. Acute pains are generally located in a specific area of the body and occur for less than three months. Chronic pain can occur in different parts of the body and over a period of six months or more. The device is unable to assist with chronic pain and you should consult a doctor instead. This device cannot treat injuries or the source of pain. It is intended only to provide short-term pain relief with the aim of improving your day-to-day life.13
Output waveform Biphasic rectangular pulse Pulse length 300 μs Pulse frequency 2-60Hz or 2/100Hz Output voltage max. 90 Vpp (500 ohms) Output current max. 180 mApp (500 ohms) Power supply 3 x 1.5 V AAA batteries LR03 Intensity Levels 0 to 25 Operating conditions 41 ° – 104 °F (5 °– 40 °C) at a relative humidity of 30– 75% Storage conditions 14 °– 131 °F (-10 °– 55 °C) at a relative humidity of 10– 90% Dimensions 3.3 x 2.8 x 1.2 in (85 x 72 x 31 mm) Weight Control unit: 2.96 oz (84 g) including batteries Cuff: 4 oz (113 g) The serial number can be found on the unit itself or in the bat- tery compartment.
11. FCC Compliance Statement
This device complies with Part 15 of the FCC Rules. Operation is subject to the following two conditions: (1) this device may not cause harmful interference; and (2) this device must ac- cept any interference received, including interference that may cause undesirable operation. NOTE: This equipment has been tested and found to comply with the limits for a Class B digital device, pursuant to Part 15 of the FCC Rules. These limits are designed to provide reason
able protection against harmful interference in a residential in- stallation. This equipment generates, uses, and can radiate radio frequency energy, and if not installed and used in accordance with the instructions, may cause harmful interference to radio communications. However, there is no guarantee that interfer
ence will not occur in a particular installation. If this equipment does cause harmful interference to radio or television reception, which can be determined by turning the equipment off and on, the user is encouraged to try to correct the interference by one or more of the following measures:
- R eorient or relocate the receiving antenna.
ncrease the separation between the equipment and receiver.
- C onnect the equipment into an outlet on a circuit different from that to which the receiver is connected.
onsult the dealer or an experienced radio/TV technician for help. Changes or modifications not expressly approved by the party responsible for compliance could void the user’s authority to operate the equipment.14
Limited Lifetime Warranty For Original Purchaser Your Beurer Knee and Elbow TENS Unit, Model EM34, is warranted to be free from defects in materials and work- manship for the life of the product under normal condi- tions of intended use and service. This warranty extends only to the original retail purchaser and does not extend to retailers or subsequent owners. We will, at our option, repair or replace the Beurer Knee and Elbow TENS Unit, Model EM34, without additional charge, for any part or parts covered by these written war
ranties. No refunds will be given. Repair or replacement is our only responsibility and your only remedy under this written warranty. If replacement parts for defective ma
terials are not available, Beurer reserves the right to make product substitutions in lieu of repair or replacement. For warranty service contact our customer service depart- ment at 1-800-536-0366 or at info@beurer.com to provide a description of the problem. If the problem is deemed to be within the scope of the limited lifetime warranty, you will be asked to mail the product at your costs in its ori- ginal package with proof of purchase, your name, address and phone number. If the problem is not deemed to be within the scope of the limited lifetime warranty, we will provide a quotation for repair respectively replacement and return shipping fee. This warranty does not cover damage caused by misuse or abuse; accident; the attachment of unauthorized ac
cessory; alteration to the product; improper installation; misapplication; lack of reasonable care with respect to the product; unauthorized repairs or modifications; im
proper use of electrical/power supply; old worn batteries; normal wear; loss of power; dropped product; malfunc- tion or damage of an operating part as a result of failure to comply with instructions for use or to provide manufac
turer’s recommended maintenance; transit damage; theft; neglect; vandalism; or environmental conditions; loss of use during the period the product is at a repair facility or otherwise awaiting parts or repair; or any other condi
tions whatsoever that are beyond the control of Beurer. This warranty is void if the product is ever used in a com- mercial or business environment. The maximum liability of Beurer under this warranty is limited to the purchase price actually paid by the customer for the product covered by the warranty, as confirmed by proof of purchase, regard
less of the amount of any other direct or indirect damage suffered by the customer. This warranty is effective only if the product is purchased and operated in the country in which the product is pur
Subject to errors and changes.15 chased. A product that requires modifications or adapta- tion to enable it to operate in any other country than the country for which it was designed, manufactured, ap- proved and/or authorized, or repair of products damaged by these modifications is not covered under this warranty. THE WARRANTY PROVIDED HEREIN SHALL BE THE SOLE AND EXCLUSIVE WARRANTY. ANY IMPLIED WAR
RANTIES, OBLIGATIONS, OR LIABILITES, INCLUDING
ATION OF THIS APPLICABLE WRITTEN WARRANTY. Some states do not allow limitations on how long an im- plied warranty lasts, so the above limitations may not apply to you. IN NO EVENT SHALL BEURER BE LIABLE FOR ANY
SPECIAL, INCIDENTAL, INDIRECT OR CONSEQUENTIAL
DAMAGES FOR BREACH OF THIS OR ANY OTHER WAR
RANTY, EXPRESS, IMPLIED OR ANY OTHER THEORY OF LIABILITY, WHATSOEVER. Some states do not al- low the exclusion or limitation of special, incidental, or consequential damages, so the above limitation may not apply to you. Distributed by: Beurer North America, LP 900 N Federal Hwy, Ste 300 Hallandale Beach, FL 33009 USA Made in China.16
- Theuseofaccessoriesorreplacementparts,otherthanthosesold/specifiedbythemanufacturermayresultinincreasedemissionsor decreased immunity of the device.
- Thedeviceshouldnotbeusedadjacenttoorstackedwithotherequipmentandthatifadjacentorstackeduseisnecessary,thedevice should be observed to verify normal operation in the configuration in which it will be used. Guidance and manufacture’s declaration – electromagnetic emissions EM34 is intended for use in the electromagnetic environment specified below. The customer or the user of EM34 should assure that it is used in such an environment. Emissions test Compliance Electromagnetic environment – guidance RF emissions CISPR 11 Group 1 EM34 uses RF energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. RF emissions CISPR 11 Class B EM34 is suitable for use in all establishments, including domestic establishments and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes. Harmonic emissions IEC 61000-3-2 Not applicable Voltage fluctuations/flicker emissions
Not applicable49 Table 2 For all me equipment and me systems Guidance and manufacturer’s declaration – electromagnetic im- munity Guidance and manufacturer’s declaration – electromagnetic immunity EM34 is intended for use in the electromagnetic environment specified below. The customer or the user of EM34 should assure that it is used in such an environment. Immunity test IEC 60601 test level Compliance level Electromagnetic environment – guidance Electrostatic discharge (ESD)
± 6 kV contact ± 8 kV air ± 6 kV contact ± 8 kV air Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic materi- al, the relative humidity should be at least 30 %. Electrical fast transient/burst
± 2 kV for power supply lines ± 1 kV input/output lines Not applicable Mains power quality should be that of a typical commercial or hospital environment. Surge IEC 61000-4-5 ± 1 kV differential mode ± 2 kV common mode Not applicable Mains power quality should be that of a typical commercial or hospital environment. Voltage dips, short interrup- tions and voltage variations on power supply input lines IEC 61000-4-11 < 5 % U
for 5 sec Not applicable Mains power quality should be that of a typical commercial or hospital environment. If the user
EM34 requires continued operation during power mains interruptions, it is recommended that EM34 is be powered from an uninterrupti- ble power supply or a battery. Power frequency (50/ 60 Hz) magnetic field IEC 61000-4-8 3 A/m 3 A/m Power frequency magnetic fields should be at levels characteristic of a typical location in a typical commercial or hospital environment. NOTICE: U
is the a.c. mains voltage prior to application of the test level.50 Table 3 For all me equipment and me systems that are not life-supporting Guidance and manufacturer’s declaration – electromagnetic immunity Guidance and manufacturer’s declaration – electromagnetic immunity EM34 is intended for use in the electromagnetic environment specified below. The customer or the user of EM34 should assure that it is used in such an environment. Immunity test IEC 60601 test level Compliance level Electromagnetic environment – guidance Conducted RF
Not applicable 3 V/m 80 MHz to 2,5 GHz 3 V/m Portable and mobile RF communications equipment should be used no closer to any part
EM34 , including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter. Recommended separation distance: d = 1,2 d = 1,2 80 MHz to 800 MHz d = 2,3 800 MHz to 2,5 GHz Where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and d is the recommended separation distance in metres (m). Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey,
should be less than the compliance level in each frequency range.
Interference may occur in the vicinity of equipment marked with the following symbol: NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies. NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people.51
Field strength from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and land mobile radios, amateur radio, AM and FM radios broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which EM34
used exceeds the applicable RF compliance level above, EM34 should be observed to verify normal operation. If abnormal performance is ob- served, additional measures may be necessary, such as reorienting or relocating EM34
Over the frequency range 150 kHz to 80 MHz, field strengths should be less than 3 V/m. Table 4 For all me equipment and me systems that are not life-supporting Recommended separation distances between portable and mobile RF communications equipment and EM34 Recommended separation distances between portable and mobile RF communications equipment and EM34 EM34 is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of EM34 can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and EM34 as recommended below, according to the maximum output power of the communications equipment. Rated maximum output power of transmitter (W) Separation distance according to frequency of transmitter (m) 150 kHz to 80 MHz d = 1,2 80 MHz to 800 MHz d = 1,2 800 MHz to 2,5 GHz d = 2,3 0,01 0,12 0,12 0,23 0,1 0,38 0,38 0,73 1 1,2 1,2 2,3 10 3,8 3,8 7,3
For transmitters rated at a maximum output power not listed above, the recommended separation distance d in meters (m) can be estimated using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer. NOTE 1 At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies. NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people.52 758.881-0517
 ManualGo.com
ManualGo.com