BPM Core - Blood pressure monitor WITHINGS - Free user manual and instructions
Find the device manual for free BPM Core WITHINGS in PDF.

User questions about BPM Core WITHINGS
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Blood pressure monitor in PDF format for free! Find your manual BPM Core - WITHINGS and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. BPM Core by WITHINGS.
USER MANUAL BPM Core WITHINGS
THANK YOU FOR CHOOSING WITHINGS BPM CORE
MERCI D'AVOIR CHOISI LE BPM CORE DE WITINGS
VIELEN DANK, DASS SIE SICH FÜR WITINGS BPM CORE ENTSCHEDEN HABEN
This guide is available in additional languages at: www.withings.com/guides
FR Ce guide est disponible dans plusieurs langues sur www.withtings.com/guides DE Diese Anleitung finden Sie in weiteren Sprachen unter: www.withtings.com/guides ESEsta guía está disponible enotiros idiomas en: www.withtings.com/guides IT Questa guida é disponible in autre lingue all'indirizzo: www.withtings.com/guides
Overview
Quick start from your smartphone
Visit go.withings.com from your smartphone or tablet, and follow the instructions to install the Health Mate app.
In the app, follow the instructions to install Withings BPM Core.
How to take a measurement
Before each measurement, follow these requirements:
- Use the product on the left upper arm
- Rest 5 minutes before the measurement
Sit down in a comfortable position, legs uncrossed, feet flat on the floor, arm and back supported. - Do not speak or move during the measurement
- You can wear one layer of clothes but it should not cover your left arm. The electrodes should be in contact with the skin.
Take the measurement in a calm and quiet area
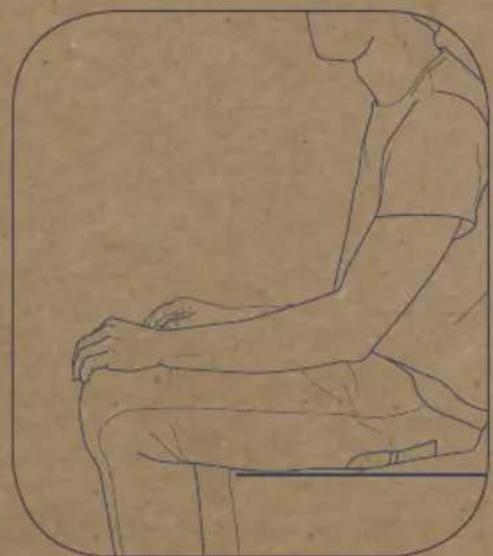
How to take a measurement
Wrap the cuff around your left arm. The tube should be positioned against your inner arm.
The stethoscope should be placed so that its flat surface is in contact with your chest. The electrodes should be in contact with the skin of your left arm.
How to take a measurement
The first part is the blood pressure measurement. Place your arm on a table and keep it level with your heart. Push the button to start BPM Core. The screen displays "START". Press the button again to start the measurement.
When the product displays the illustrated pictogram, you need to place your hand on the metal tube and the stethoscope against your chest. Adjust your arm position to ensure an accurate measurement.
How to take a measurement
At the end of the measurement, results are displayed on the screen and sent to the app via Wi-Fi or Bluetooth. If you didn't place your hand on the electrode, BPM Core did not record ECG and stethoscope signal and only displays blood pressure readings.
When you start the measurement, you can also select "Start x3" or "ECG" by swiping the right part of the screen. "Start x3" launches three blood pressure measurements in a row. "ECG" only records ECG and stethoscope signal.
In order to use your BPM Core, you need an iOS (10.0 or higher) or Android (6.0 and higher) device to install it. Thereafter, the product can be used in stand-alone, thanks to the Wi-Fi and Bluetooth® connections.
Important safety information
Consult your doctor during pregnancy, or if you suffer from arrhythmia and arteriosclerosis. Please read this section carefully before using the Smart blood pressure monitor, with ECG & digital stethoscope.
Intended use
The BPM Core is a digital monitor intended for use in measuring blood pressure, heart rate, to record a one derication ECG to detect atrial fibrillation, and uncover valvular heart diseases (aortic stenosis, aortic regurgitation, mitral stenosis). The device is intended to be used in a human adult population with an arm circumference between 9 inches to 17 inches (22 cm to 42 cm). The BPM Core is a medical device. Contact your physician if hypertensive values, AFib or VHD are indicated.
General safety and precautions
- Do not forcibly bend the arm cuff.
- Do not inflate the arm cuff when it is not wrapped around your arm.
- Do not apply strong shocks and vibrations to the blood pressure monitor or drop it.
- Do not take measurement after bathing, drinking alcohol, smoking, exercising or eating.
- Do not immerse the arm cuff in water.
- Do not use with a pacemaker, a defibrillator or other electric implant.
- Use on adults only.
- Do not use on children or pets.
Android is a trademark of Google Inc.
iOS is a trademark of Apple Inc.
Caution
General usage
Always consult your doctor. Self-diagnosis of measurement results and self treatment are dangerous. People with severe blood flow problems, or blood disorders, should consult a doctor before using the blood pressure monitor. Cuff inflation can cause internal bleeding. Operational factors such as common arrhythmias, ventricular premature beats, arterial sclerosis, poor perfusion, diabetes, age, pregnancy, pre-eclampsia or renal disease can affect the performance of the automated sphygmomanometer and/or its blood pressure reading. This device is a precision measuring equipment liable to be understood by lay user but it still should be handled with care. A long exposure of the device to lint, dust or sunlight might reduce its life time or damage it. Damaged cuff or sensor might lead to incorrect measurements. No usage for USB port. The user is an intended operator. Parts in contact with the skin: cuff and electrodes. Measurements can be affected by extreme temperatures, humidity & altitude.
- Do not leave the blood pressure monitor unattended with infants or persons who cannot express their consent.
- Do not use the blood pressure monitor for any purpose other than measuring blood pressure.
- Do not disassemble the blood pressure monitor.
- Do not operate the blood pressure monitor in a moving vehicle (car, airplane).
- Do not use a cellular phone near the device.
- Do not use the device with the USB cable plugged.
- Improper continuous pressure of cuff or too frequent measurements may interfere blood flow and result harmful injury to the users. Check if the use of the device does not result in prolonged impairment of your blood circulation.
- Do not apply the cuff over the users' arm bearing a wound or medical treatment and so on, as this can cause further injury.
- Cuff should be used on the arm on the side of a mastectomy.
- Use of the device can temporarily cause loss of function of simultaneously used equipment on the same limb.
After use
Cleaning
- Do not use an alcoholic-base or solvent agent to clean the device
- Clean the device with a soft and dry cloth
- The dirt on the cuff can be cleaned by a moisten cloth and soap
- Do not flush the device and cuff with much water
- Do not dismantle the device or disconnect the cuff or try to repair by yourself. If any problem happens, refer to the distributor
- Do not operate the device under severe environment of extreme temperature or humidity, or direct sunshine
- Do not shake the unit violently
- Do not submerge the device or any of the components in water
- Do not let the device under strong shocks, such as dropping the unit on the floor
Storage
- Store the device and the components in a clean and safe location
- If storage conditions are different from the usage conditions indicated in this document, please wait 30 minutes before taking a measurement.
Maintenance
If you can't fix the problems using the troubleshooting instructions, request service from your dealer. Manufacturer will make available on requested circuit diagrams, component part lists, descriptions, calibration instructions, or other information that will assist manufacturer's staff or authorized representative for repair. It is generally recommended to have the device inspected every 2 years, to ensure proper functioning and accuracy. Do not use the device while doing maintenance steps.
Error messages
Error message Causes Countermeasures
| Measurement could not be performed. Please try again. If the problem occurs again, please contact customer service. | Hardware auto- check error. Measurement could not be performed. Please try again. If the problem occurs again, please contact customer service. |
| Connection error. Quit the application and reconnect your blood pressure monitor. | Communication error between the Blood Pressure Monitor and the collateral device. |
| Please wait until the cuff is totally deflated before starting a new measurement. Stay still during measurement. | - Cuff is not deflated before measurement. - Noise interference. - Excessive user movements. |
Error messages
Error message Causes Countermeasures
Check that the Blood Pressure Monitor is correctly positioned on your arm and that measurement is performed in good conditions.
-User movement, or hot relaxed, or talking ...
Cuff is not correctly fastened.
Cuff broken
- Pump or valve failure
-Pressure overflow
- Noise interference
-User movement, or not relaxed, or talking
-User special characteristics
Battery level is too low
- Check that the Blood Pressure Monitor is correctly positioned on your arm and that measurement is performed in good conditions.
- Low battery level, Load your BPM Core with a micro USB cable.
- If user has special characteristics, please contact your physician.
- If the problem occurs again, please contact customer service.
Low battery level. Battery level is too low. Load the device with the provided charging
cable.
Troubleshooting
Problem Remedy
| No results for the ECG measurement | Ensure that the three electrodes are in direct contact with the skin. If yes, clean them with a tissue |
| A valvular heart disease analysis cannot be done | Check the stethoscope position, the flat surface must be in contact with the chest. Ensure that you did the required number of measurements in order to get a readings |
| The inflation action cannot be done or the air pressure cannot rise | 1. Check the cuff position and fasten the cuff correctly ad remeasure the pressure again2. Check the blood pressure monitor connection to the iOS or Android device |
| The low battery icon is displayed on the device | Reload the device with the cable provided |
| The blood pressure cannot be taken and the application shows an error message | 1. Re-fasten the cuff2. Relax yourself and sit down3. Keep the cuff and heart at the same level during the measurement period4. Keep silent and still during measurement5. If the patient has sever heart beat problem, then the blood pressure may not be read correctly |
| Under normal measuring circumstance, the reading at home is different from that of the clinics or each measurement has various reading | 1. The variation is due to the different environments2. The blood pressure is changing according to the physiological or psychological status of the human body3. Atrial fibrillation may not be permanent at early stage4. Show your recorded values to your physician |
Specifications Technical data
| Product description: | Digital automatic Blood Pressure Monitor with EKG and Heart sounds sensor |
| Model: WPM04 | |
| Blood pressure measurement method: Cuff oscillometric method | |
| Cuff inflation: Automatic inflation with air pump at 15 mmHg/s | |
| Pressure sensor: Gauge sensor | |
| Measurement range (pressure): | 0 to 285 mmHg, DIA 40 to 130 mmHg, SYS 60 to 230 mmHg |
| Measurement range (pulse): | 40 to 180 beats/min |
| Accuracy (pressure): Within +-3 mmHg or 2% of reading | |
| Accuracy (pulse): Within +-5% of reading | |
| Sensor: Semiconductor pressure sensor | |
| Operating conditions: | 5 to 40°C, 15 to 90% RH, atmospheric 86Kpa-106kpa, altitude: 2000m |
| Storage and transport conditions: | -25 to 70°C, 10 to 95% RH, atmospheric 86Kpa-106kpa, altitude: 2000m |
| Arm type: Use on left arm. | |
| Power source: 3,6 VDC Lithium Ion battery | |
| Weight: Approx. 430g | |
| Accessories: Charging cable, Instruction manual | |
| Product life: 3 years | |
| Wireless transmission: Wi-Fi and BLE |
Note: Specifications are subject to change without prior notice or any obligation on the parts of the manufacturer
Disposal
Actuation of European directives 2002/95/EC, 2002/96/EC and 2003/108/EC, for reduction in use of dangerous substances in the electric and electronic device and for garbage disposal.
The symbol applied on the device or its packaging means that at the end of its useful life the product must not be disposed of with domestic waste.

At the end of devices useful life, the user must deliver it to the able collecting centers for electric and electronic garbage, or give back to the retailer when purchasing a new device. Disposing of the product separately prevents possible negative consequences for the environment and for health, deriving from inadequate disposal. It also allows
the recovery of materials of which it's made up in order to obtain an important saving of energy and resources and to avoid negative effects to the environment and health. In case of abusive disposal of device by the user, will be applied administrative endorsements in compliance with current standard. The device and its parts is mared with regard to disposal, as appropriate, in accordance with national or regional regulations.
Version 1.0 April 2019
FCC Statement
Federal Communications Commission (FCC) Statement 15.21
You are cautioned that changes or modifications not expressly approved by the part responsible for compliance could void the user's authority to operate the equipment. 15.105(b)
This equipment has been tested and found to comply with the limits for a Class B digital device, pursuant to part 15 of the FCC rules.
These limits are designed to provide reasonable protection against harmful interference in a residential installation. This equipment generates, uses and can radiate radio frequency energy and, if not installed and used in accordance with the instructions, may cause harmful interference to radio communications. However, there is no guarantee that interference will not occur in a particular installation.
If this equipment does cause harmful interference to radio or television reception, which can be determined by turning the equipment off and on, the user is encouraged to try to correct the interference by one or more of the following measures:
- Reorient or relocate the receiving antenna.
-
Increase the separation between the equipment and receiver.
-
Connect the equipment into an outlet on a circuit different from that to which the receiver is connected.
- Consult the dealer or an experienced radio/TV technician for help.
This device complies with Part 15 of the FCC Rules. Operation is subject to the following two conditions:
1) this device may not cause harmful interference and 2) this device must accept any interference received, including interference that may cause undesired operation of the device.
FCC RF Radiation Exposure Statement:
This equipment complies with FCC radiation exposure limits set forth for an uncontrolled environment. End users must follow the specific operating instructions for satisfying RF exposure compliance. This transmitter must not be co-located or operating in conjunction with any other antenna or transmitter. This device complies with Industry Canada license-exempt RSS standard(s). Operation is subject to the following two conditions: (1) this device may not cause interference, and (2) this device must accept any interference, including interference that may cause undesired operation of the device.
RF Statement
Medical Electrical Equipment needs special precautions regarding EMC and needs to be installed and put into service according to the EMC information provided in the following. | Interference may occur in the vicinity of equipment marked with | Portable and mobile RF communication equipment (e.g. cell phones) can affect Medical Electrical Equipment. | The use of accessories and cables other than those specified may result in increased emissions or decreased immunity | The device uses RF energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment | The device is suitable for use in all establishments, including domestic establishments and those directly connected to the public low voltage power supply network that supplies buildings used for domestic purposes | Portable and mobile RF communications equipment should be used no closer to any part of the device, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter | The device is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. Any other accessories, transducers and cables may result in increased emissions or decreased immunity and EMC performance. | The device should not be used adjacent to or stacked with other equipment and that if adjacent or stacked use is necessary, which should be observed to verify normal operation in the configuration in which it will be used. Medical Electrical Equipment needs special precautions regarding EMC and needs to be installed and put into service according to the EMC information provided in the following. Portable and mobile RF communication equipment (e.g. cell phones) can affect Medical Electrical Equipment. The use of accessories and cables other than those specified may result in increased emissions or decreased immunity of the unit.
Guidance and manufacturer's declaration-electromagnetic emissions
The Smart blood pressure monitor, with ECG & digital stethoscope is intended for use in the electromagnetic environment specified below. The customer or the user of the Smart blood pressure monitor, with ECG & digital stethoscope should assure that it is used in such an environment.
| Emissions test Compliance | Electromagnetic environment - guidance | |
| CE emissionsCISPR11 | Group 1 | The Smart blood pressure monitor, with ECG & digital stethoscope uses RF energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. |
| RE emissionsCISPR11 | Class B | |
| Harmonic emissionsIEC 61000-3-2 | Not applicable | The Smart blood pressure monitor, with ECG & digital stethoscope is suitable for use in all establishments, including domestic establishments and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes; |
| Voltage fluctuations/Flicker emissionsIEC 61000-3-3 | Not applicable | |
Declaration - electromagnetic emissions and immunity
for equipment and systems that are not life-supporting and are specified for use only in a shielded location
27
The Smart blood pressure monitor, with ECG & digital stethoscope declaration electromagnetic immunity
The Smart blood pressure monitor, with ECG & digital stethoscope system is intended for use in the electromagnetic environment specified below. The customer or the user of the Smart blood pressure monitor, with ECG & digital stethoscope system should assure that it is used in such an environment.
Immunity test IEC 60601 test level Compliance level Electromagnetic environment - guidance
| Conducted RF IEC 61000-4-6 | 3 Vrms 150 kHz to 80 MHz | N/A N/A | N/A |
| Radiated RF IEC 61000-4-3 3 V/m 80 MHz to 2.5 GHz | N/A Portable and mobile RF communications equipment should be used no closer to any part of the EQUIPMENT or SYSTEM including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter. Interference may occur in the vicinity of equipment marked with the following symbol. | ||
Declaration - electromagnetic emissions and immunity
for equipment and systems that are not life-supporting and are specified for use only in a shielded location
Declaration - electromagnetic immunity
The Smart blood pressure monitor, with ECG & digital stethoscope system is intended for use in the electromagnetic environment specified below. The customer or the user of the Smart blood pressure monitor, with ECG & digital stethoscope system should assure that it is used in such an environment.
Immunity test IEC 60601 test level Compliance level Electromagnetic environment - guidance
| Electrostatic discharge (ESD)IEC 61000-4-2 | Contact: ±8 kV Air: ±2 kV, ±4 kV, ±8 kV, ±15 kV | Contact: ±8 kV Air: ±2 kV, ±4 kV, ±8 kV, covered with synthetic material, the relative humidity should be at least 30 %. | Floors should be wood, concrete or ceramic tile. If floors are covered with synthetic material, the relative humidity should be at least 30 %. |
| Electrical fast transient/ burst IEC 61000-4-1 | 2 kV for power supply lines 1 kV for input/output lines | N/A N/A | Mains power quality should be that of a typical commercial or hospital environment. |
| Surge IEC 61000-4-5 1 kV differential mode | N/A | A Mains power quality should be that of a typical commercial or hospital environment. | |
| N/A | |||
| Voltage dips, short interruptions and voltage variations on power supply input lines IEC 61000-4-11 | -5 % UT(95% dip in UT) for 0.5 cycle, -40 % UT (60 % dip in UT) for 5 cycles, -70 % UT(30 % dip in UT) for 25 cycles, -5 % UT (95 % dip in UT) for 5 sec | N/A Mains power quality should be that of a typical commercial or hospital environment. If the user of the EQUIPMENT or SYSTEM requires continued operation during power mains interruptions, it is recommended that the EQUIPMENT or SYSTEM be powered from an uninterruptible power supply or a battery. | |
| Power frequency (50/60 Hz) magnetic field IEC 61000-4-8 | 30 A/m 50 Hz or 60 Hz | 30 A/m 50 Hz and 60 Hz | Power frequency magnetic fields should be at levels characteristic of a typical location in a typical commercial or hospital environment. |
Informations importantes
Configuration require
Version 1.0 April 2019
Withings Two (2) Years commercial guarantee Withings BPM Core | Smart blood pressure monitor, with ECG & digital stethoscope
Withings SA, 2 rue Maurice Hartmann, 92130 Issy-les-Moulineaux, France ("Withings") warrants the Withings-branded hardware product ("Withings Product") against defects in materials and workmanship when used normally in accordance with Withings' published guidelines for a period of TWO (2) YEARS from the date of original retail purchase by the end-user purchaser ("Warranty Period"). Withings' published guidelines include but are not limited to information contained in technical specifications, safety instructions or quick start guide. Withings does not warrant that the operation of the Withings Product will be uninterrupted or error-free. Withings is not responsible for damage arising from failure to follow instructions relating to the Withings Product's use.
Information to users in Canada
This device complies with Part 15 of the FCC Rules and with Industry Canada licence-exempt RSS standard(s).
Operation is subject to the following two conditions:
(1) This device may not cause harmful interference, and
(2) This device must accept any interference received, including interference that may cause undesired operation.
Changes or modifications made to this equipment not expressly approved by Withings may void the FCC authorization to operate this equipment.
Radiofrequency radiation exposure information: The radiated output power of the device is far below the FCC radio frequency exposure limits. Nevertheless, the device shall be used in such a manner that the potential for human contact during normal operation is minimized.
Reference to standards
This device complies with the following normative documents: COUNCIL DIRECTIVE 93/42/EEC of 14 June 1993 concerning medical devices as amended by Directive 2007/47/EC
EN ISO 13485: Medical devices - Quality management systems - Requirements for regulatory purposes. Reference to standards contd. EN ISO 14971: Medical devices - Application of risk management to medical devices. IEC/EN 60601-1: Medical electrical equipment - Part 1: General requirements for basic safety and essential performance. EN 1060-3: Non-invasive sphygmomanometers.
Part 3: Supplementary requirements for electromechanical blood pressure measuring systems. | EN 1060-4: Non-invasive sphygmomanometers. Test procedures to determine the overall system accuracy of automated non-invasive sphygmomanometers. | IEC/EN 60601-1-1: General requirements for basic safety and essential performance - Collateral Standard: Requirements for medical electrical equipment and medical electrical systems used in the home healthcare environment. | IEC 80601-2-30: Medical electrical equipment - Part 2-30: Particular requirements for the basic safety and essential performance of automated non-invasive sphygmomanometers. | EN 300 328: Electromagnetic compatibility and Radio spectrum Matters (ERM); Wideband transmission systems; Data transmission
equipment operating in the 2,4 GHz ISM band and using wide band modulation techniques; Harmonized EN covering the essential requirements of article 3.2 of the Directive (2014/53/EU). | EN 301 489-1: Electromagnetic compatibility and Radio spectrum Matters (ERM); ElectroMagnetic Compatibility (EMC) standard for radio equipment and services; Part 1: Common technical requirements. | EN 301 489-17: Electromagnetic compatibility and Radio spectrum Matters (ERM); ElectroMagnetic Compatibility (EMC) standard for radio equipment and services; Part 1: Common technical requirements | IEC/EN60601-1-2: Medical electrical equipment: Part 1-2: General requirements for basic safety and essential performance-collateral standard electromagnetic compatibility | EN 55011: Industrial, scientific and medical equipment - Radio-frequency disturbance characteristics - Limits and methods of measurement. | FCC part B 15B: Electromagnetic Compatibility. | FCC Rule Part 15.247 Cat: DSS (Bluetooth) FCC Rule Part 15.247 Cat: DTS (BT4.0). | EN ISO 10993-1: Biological evaluation of medical devices -- Part 1: Evaluation and testing within a risk management process. | EN ISO 10993-5: Biological evaluation of medical devices -- Part 5: Tests for in vitro cytotoxicity | EN ISO 10993-10: Biological evaluation of medical devices. Tests for irritation and skin sensitization | IEC/EN 60601-1-6: Medical electrical equipment - Part 1-6: General requirements for basic safety and essential performance - Collateral standard: Usability | ANSI/AAMI/ISO 81060-2 Non-invasive sphygmomanometers Part 2: Clinical validation of automated measurement type | EN ISO 15223-1: Medical devices - Symbols to be used with medical device labels, labelling and information to be supplied - Part 1: General requirements | IEC 60601-2-47:2012: Medical electrical equipment - Basic safety and essential performance of ambulatory electrocardiograph systems | IEC 57:2012: Testing and reporting performance results of cardia rhythm and ST-segment measurement algorithms

Manufacturer/Fabricant/Hersteller:
WITHINGS SA,
Ingress of water or particulate matter
Dry storage environment
Complies with waste electrical and
electronic equipment directive
Type BF Applied Part (cuff)
FR PIECE appliquee de type BF (brassard)
Read this manual before use
EN Withings SA hereby declares that the device Withings BPM Core is in conformity with the essential requirements and other relevant requirements of Directive 2014/53/EU.
The declaration of conformity can be found at: withings.com/compliance.
Our goods come with guarantees that cannot be excluded under the Australian Consumer Law. You are entitled to a replacement or refund for a major failure and compensation for any other reasonably foreseeable loss or damage. You are also entitled to have the goods repaired or replaced if the goods fail to be of acceptable quality and the failure does not amount to a major failure.


Need help?
withings.com/support
IM_WPM04_ENFRDE_003
withings
Withings BPM Core | Smart blood pressure monitor, with ECG & digital stethoscope | V1.0 - Apr.2019 © 2019 Withings SA. All rights reserved.




