KD798 - Blood pressure monitor TensioFlash - Free user manual and instructions
Find the device manual for free KD798 TensioFlash in PDF.

User questions about KD798 TensioFlash
0 question about this device. Answer the ones you know or ask your own.
Ask a new question about this device
Download the instructions for your Blood pressure monitor in PDF format for free! Find your manual KD798 - TensioFlash and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. KD798 by TensioFlash.
USER MANUAL KD798 TensioFlash

SISOMEDGROUPSA 112,AvenueKleber 75116Paris-France
DISTRIBUE PART DISTRIBUET BY
VISOEMEDGROUPSA
- Avenue Kleber 75116 PARIS-France contact@visioned-lab.fr Tel.: +33 [8] 92 350 334 [40/m) www.visionlab.com
www.visionmed-lab.com

KD-798


By Visiomed
LEXIQUE DES SYMBOLES / SYMBOL DEFINITIONS
Medical device in compliance with current European standards.
Please read before use.
TABLE OF CONTENTS
- WARNING 21
2.BLOOD PRESSURE 22 - PRECAUTIONS BEFORE USE 24
4 FEATURES OF THE TENSIOFLASH* KD-798 24 - MAIN DEVICE 24
6.CHANGING THE BATTERIES 25 - TIME AND DATE SETUP 25
- SOME SUGGESTIONS BEFORE MEASUREMENT 26
9.HOW TO USE THE WRIST CUFF 26 - HOW TO MEASURE BLOOD PRESSURE 27
- MEMORY STORAGE AND RECALL 27
- DEVICE MAINTENANCE 28
- PREVENT A MALFUNCTION 28
- TECHNICAL SPECIFICATIONS 29
15.TROUBLESHOOTINGGUIDE 30
The manufacturer reserves the right to modify the product technical specifications without prior notice.
Dear client,
Thank you for purchasing the VSIOMED upper arm automatic blood pressure monitor. We recommend that you read these instructions carefully, in order to measure your blood pressure effectively.
1. WARNING
- Follow the maintenance instructions in this manual.
- This device is suitable for home use.
- Use this device only for its intended use as described in this notice.
- Use this device in an ambient temperature range between 5 and 40^
- This device must always be stored in a clean, dry area.
- Do not expose this device to electric shock.
- Do not expose this device to extreme temperature conditions > 55^ C or < 20^ C .
- Do not expose this device to direct sunlight or water.
- Do not use this device outside.
- Never drop the device.
- Do not attempt to service this device yourself. - Do not attempt to open the device. In case of problems, contact your dealer.
- This device is not intended for use by persons (including children) with impaired physical, sensory or mental capabilities, or persons with no experience or knowledge of the device, unless they have undergone supervision or prior instructions concerning the use of this device by a person responsible for their safety.
- This electronic medical device requires specific precautions regarding electromagnetic compatibility. It must be installed and commissioned in accordance with the electromagnetic information provided in the Electromagnetic compatibility (EMC) Data table
- Measurements may be impaired if used near a TV, microwave oven, mobile phone or other devices with an electric field.
-
The measurement results are given for information only. If unsure about your results, please contact your doctor.
-
This device should not encourage self-medication or adjusting your treatment.
- Stop using the device in the event of an abnormality or malfunction.
2.BLOOD PRESSURE
Blood pressure varies considerably throughout the day and in hypertensive individuals these variations are even more pronounced. It is lower in the morning and increases in the afternoon and evening. It decreases during sleep. Blood pressure is lower in summer than in winter and increases substantially with age. Blood pressure also increases during exercise, intercourse, pregnancy, stress ...
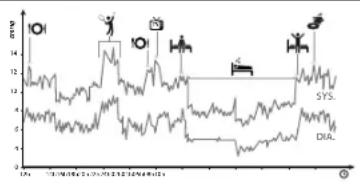
EXAMPLE OF BLOOD PRESSURE VARIATIONS IN A MAN OF 35 YEARS
Note: One or two readings will not provide a true indication of your normal blood pressure. It is important to take regular, daily measurements and to keep accurate records. Communicating your findings to your doctor can be a valuable aid in diagnosing and preventing potential health problem.
The World Health Organisation (WHO) developed the Blood Pressure Classification indicated in the following chart (1999 - revised in 2003). This classification, however, is only a general guideline, because blood pressure varies from person to person according to age, weight, and health status.
It is advisable to consult your doctor to determine your normal blood pressure.
| BLOOD PRESSURE CLASSIFICATION CHART (IN MM HG) | ||||
| Category Systolic (upper) Diastolic (lower) | WHO indication in colour | |||
| Optimal less than 120 and less than 80 green | ||||
| Normal between 120 and 129 and/or between 80 and 84 green | ||||
| Normal high between 130 and 139 and/or between 85 and 89 green | ||||
| Stage 1 - Mild Hypertension | between 140 and 159 and/or between 160 and 179 and/or between 180 | and/or between 100 and 109 | orange | |
| Stage 2 - Moderate Hypertension | ||||
| Stage 3 - Severe Hypertension | ||||
| Isolated systolic hypertension | greater than or equal to 140 | and less than 90 red | ||
| Note: There is no universally accepted definition of hypotension (low blood pressure), but systolic pressure below 99 mm Hg is usually regarded as hypotension. | ||||
- Do not confuse self-monitoring with self-diagnosis. Blood pressure readings should be interpreted by a physician or trained healthcare professional, who is familiar with your medical history. Measurements do not replace your doctor's treatment or regular check-ups.
- If you are taking medication, consult with your physician to determine the most appropriate time to measure your blood pressure.
- Do not use this device on children and the elderly without medical advice.
- This device should not encourage self-medication or adjusting your treatment.
- Do not use the device in a moving vehicle. This can result in erroneous measurements.
- Blood pressure measurements determined with this device are equivalent to those obtained by a trained observer using the cuff/stethoscope auscultation method, within the limits prescribed by the American National Standard for Electronic or automated sphygmomanometers.
- Do not use any other cuff than the one provided.
4. FEATURES OF THE TENSIOFLASH ^® KD-798
-
Indication of SYS, DIA, Pulse/Min. measurements and WHO international blood pressure classification level
2.Cardiac arrhythmia detection -
Automatically stores the 60 last readings.
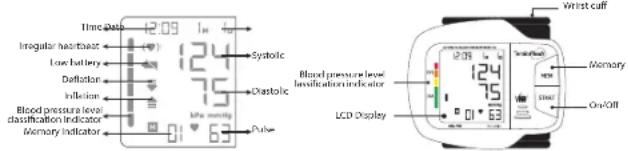
5. MAIN DEVICE
6. CHANGING THE BATTERIES
When the LCD screen displays "Battery", the batteries are low or depleted. 1. Open the battery cover underneath the device.
-
Replace the used battery, taking care to observe the indicated polarity. A polarity error may cause damage and void the guarantee. Never insert rechargeable batteries. Use disposable batteries. Do not mix new batteries with used batteries.
-
Replace the cover.
Do not dispose of old batteries with household waste. Take your used batteries to a collection point.
If not used for a long period of time, be sure to remove the battery from the device to prevent leakage of liquid that may damage the device.
Do not use batteries other than the batteries mentioned, do not recharge non-rechargeable batteries, or throw them into a fire.
7. TIME AND DATE SETUP
- Press the START and MEM button simultaneously. The month flashes.
- Use the MEM button to set the month.
- Press the START button to save your settings and display the day.
4.Use the MEM button to set the day. - Follow the steps to set the hour and then the minutes.
- Press START to confirm your selection.
- The date and time must be adjusted at each battery change.

8. SOME SUGGESTIONS BEFORE MEASUREMENT
- Avoid eating, smoking, and exercising for 30 minutes before taking a measurement. Rest for at least 15 minutes before taking a reading.
- Stress raises blood pressure. Avoid taking measurements when stressed.
- It is best to take your blood pressure on the left arm.
- The measurement should be taken in a quiet place, you should be relaxed, in a seated position, your left arm on a table so that the cuff is level with your heart.
- Sit with your feet flat on the floor (legs uncrossed), place your left arm on the table with your palm facing upward.
- Remain still and do not talk during the measurement.
- Record your blood pressure and pulse results in order to show them to your doctor. You should take your blood pressure measurements regularly at the same time of day.
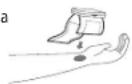
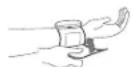

9. HOW TO USE THE WRIST CUFF
- Remove all jewellery (watch, bracelet ...) from your left hand.
- Palm up, slip on the cuff so that the monitor display faces you in the reading direction.
- The cuff should be one or two centimetres from your palm.
- When the cuff is properly positioned, hold it in place by pressing the tab firmly against the Velcro.
Notes:
- Always measure the same arm (normally left) for a better interpretation of the results. If you cannot put the cuff on your left arm, you can use your right arm.
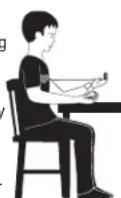
10. HOW TO MEASURE BLOOD PRESSURE
- Press the START button. All the display characters are shown on the screen.
- The device displays "zero" on the screen when it is ready to take the measurement. The cuff inflates automatically. When the screen displays the maximum value the pressure is released, the heart beats are detected. The air is automatically evacuated from the cuff.
- After the measurement is complete, the systolic and diastolic pressure readings and pulse rate are displayed.
- The result will be automatically stored.
- Press START again to turn the device off. If you forget, the device will stop automatically after about 1 minute.
-
- If there is too much pressure from the cuff, or you feel uncomfortable, please press the START button immediately to quickly deflate the cuff.
Notes:
- The value measured will be too low if the cuff is held higher than the heart, or will be too high if the cuff is held lower than the heart.
- The device automatically stores the last 2 sets of 30 readings. If more than 2x30 readings, the earliest data will be deleted.
- During measurement do not speak or move as it may affect the accuracy of the result.
- If you want to stop during the measurement process, just press the START button to stop immediately.
- Consult your doctor if in doubt about using the device.
11. MEMORY STORAGE AND RECALL
- Press the MEM button to display the stored results. The device displays the number of recorded readings. If no result stored, will be displayed.
- The screen will display the last measurement. Press the MEM button to review data in ascending order. The time and date are stored for all results.
-
To delete measurements from the memory, press the MEM button until 0 is displayed on the screen.
-
Press the START button to turn off the monitor.
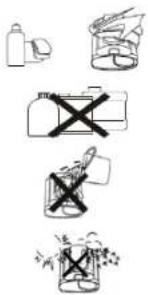
12. DEVICE MAINTENANCE
- Use a soft dry cloth to clean the device. If necessary, use a cloth lightly dampened with tap water.
- Do not use alcohol, benzene, thinner or other harsh chemicals to clean the device or cuff.
- Before using please wash your hands. Do not wash or wet the cuff.
- Avoid placing the device in contact with direct sunlight, high humidity or dust.
- Fold the pressure cuff and store the device in its box. Note: If the blood pressure monitor is left unused for long periods, remove the batteries from the device (otherwise, the batteries may leak and damage the device)
13. PREVENT A MALFUNCTION
- Do not drop the device, avoid jolts and shocks.
- Never insert anything into the device.
- Do not disassemble the device.
- Do not put the device in water.
- The device may not meet its performance specifications if stored or used outside the temperature and humidity ranges specified in the specifications.
- The device retains its safety and performance characteristics for a minimum of 10,000 measurements or three years of use.
- TECHNICAL SPECIFICATIONS
| Product name | TensioFlash - Automatic wrist blood pressure monitor |
| Model KD-798 | |
| Measurement method | Oscillometric system |
| Classification Class IIa medical device | |
| Indication Digital LCD display | |
| Measuring range | Cuff pressure: 0~300 mm Hg. Pulse: 40~180 Beats/min |
| Accuracy | Pressure +/- 3 mm Hg Pulse +/- 5% |
| Inflation Inflation by electric pump | |
| Deflation Pressure released by valve | |
| Memory Stores | 60 last readings. |
| Power source 2 x 1.5V "AAA" alkaline batteries | |
| Battery life | >150 readings (once a day, at room temperature: 22°C) |
| Working Temp. & humidity | 5~40°C (40~104°F) <85% RH |
| Storage Temp. & humidity | -20~55°C (-4~131°F) ≤95% RH |
| External dimensions | 84mm (L)/ 65mm (W)/ 30mm (H) |
| Weight Approx. | 125g (Device and Batteries) |
| Accessories | Cuff (wrist circumference 14~19.5 cm). Instruction manual, Case, 2 batteries -1.5V AAA |
| Voltage 3 Volts | DC |
The manufacturer reserves the right to modify the product technical specifications without prior notice.
15. TROUBLESHOOTING GUIDE
If any of the following problems occur when using your TensioFlash®, please refer to this troubleshooting guide. If the problem persists, please contact our customer service at 0892350334 (0,40€/mn).
| PROBLEM POSSIBLE CAUSE SOLUTION | ||
| Nothing is displayed when the START/STOP button is pressed | No battery inserted Insert batteries | |
| The batteries are used Insert new batteries | ||
| The batteries are not placed in the correct polarity direction | Observe battery polarity | |
| Screen displays abnormal results | The cuff position was not correct or it was not properly tightened | Apply the cuff correctly and try again |
| The body posture was not correct during the measurement | Review the suggestions before measurement | |
| You moved during the measurement or you were not sufficiently relaxed. | Do not move during the measurement. Relax for 10 minutes before taking your blood pressure. | |
| Arrhythmia | The device is not suitable for people with severe cardiac arrhythmia | |
| Screen displays "Er 5" | Cuff pressure is above 300mmHg | Measure again after 5 minutes. If the error persists contact customer service |
| Screen displays "Er 6" | More than 3 minutes with cuff pressure over 15mmHg | |
| Screen displays "Er 7" | EEPROM access error | |
| Screen displays "Er 8" | Device parameter checking error | |
| Screen displays "Er A" | Pressure sensor parameter error |
∀⊥∀D W
ELECTROMAGNETIC COMPATIBILITY INFORMATION
Table 1 For all ME EQUIPMENT and ME SYSTEMS
| Guidance and manufacture's declaration - electromagnetic emissions | ||
| The KD-738 is intended for use in the electromagnetic environment specified below.The customer or the user of the KD-798 should assure that it is used in such an environment. | ||
| Emissions test Compliance | Electromagnetic environment | guidance |
| RF emissionsCISPR 11 | Group 1 | The KD-798 uses RF energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. |
| RF emissionsCISPR 11 | Class B | The KD-798 is suitable for use in all establishments other than domestic and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes. |
| Harmonic emissionsIEC 61000-3-2 | Not applicable | |
| Voltage fluctuations/flicker emissionsIEC 61000-3-3 | Not applicable | |
Table 2 For all ME EQUIPMENT and ME SYSTEMS
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The KD 798 is intended for use in the electromagnetic environment specified below. The customer or the user of the KD-798 should assure that it is used in such an environment. | |||
| IMMUNITY test | IEC 60601test level | Compliance level | Electromagnetic environment - guidance |
| Electrostatic discharge (ESD)IEC 61000-4-2 | ±6 kV contact±8 kV air | ±6 kV contact±8 kV air | Floors should be wood, concrete or ceramic tile.If floors are covered with synthetic material, the relative humidity should be at least 30 %. |
| Power frequency(50/60 Hz)magnetic fieldIEC 61000-4-8 | 3 A/m 3 A/m | Power frequency magnetic fields should be at levels characteristic of a typical location in a typical commercial or hospital environment. | |
Table 3 For all ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
| Guidance and manufacturer's declaration - electromagnetic immunity | |||
| The KD-798 is intended for use in the electromagnetic environment specified below. The customer or the user of the KD-798 should assure that it is used in such an environment. | |||
| IMMUNITY test | IEC 60601test level | Compliance level | Electromagnetic environment - guidance |
| RADIATED RF IEC 61000-4-3 3V/m | 80 MHz to 2.5 GHz | 3 V/m Portable and | mobile RF communications equipment should be used no closer to any part of the KD-798, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter. |
| Recommended separation distance: d = 1.2 √P 80 MHz to 800 MHz d = 2.3 √P 800 MHz to 2,5 GHz Where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and d is the recommended separation distance in meters (m). Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey.a should be less than the compliance level in each frequency range.b Interference may occur in the vicinity of equipment marked with the following symbol: | |||
| NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies. NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. a Field strengths from fixed transmitters, such as base stations for radio (cellular/cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the KD-798 is used exceeds the applicable RF compliance level above, the KD-798 should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as re-orienting or relocating the KD-798. b Over the frequency range 150 kHz to 80 MHz, field strengths should be less than 3V/m. | |||
Table 4 For ME EQUIPMENT and ME SYSTEMS that are not LIFE-SUPPORTING
| Recommended separation distances between portable and mobile RF communications equipment and the KD-798 | |||
| The KD-798 is intended for use in an electromagnetic environment in which radiated RF disturbances are controlled. The customer or the user of the KD-798 can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and the KD-798 as recommended below, according to the maximum output power of the communications equipment. | |||
| Rated maximum output of transmitW | Separation distance according to frequency of transmitter m | ||
| 150 kHz to 80 MHz d = 1.2 √p | 80 MHz to 800 MHz d = 1.2 √p | 800 MHz to 2,5 GHz d = 2.3 √p | |
| 0.01 0.12 0.20.23 | |||
| 0.1 0.38 0.38 0.73 | |||
| 1 1.2 1.2 2.3 | |||
| 10 3.8 3.8 7.3 | |||
| 100 12 12 23 | |||
| For transmitters rated at a maximum output power not listed above, the recommended separation distance d in meters (m) can be determined using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer. NOTE 1 At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies. NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people. | |||
A⊥N∀R∀M
CARTE DE GARANTIE - GUARANTEE CARD
Date d'achat / Purchase date
Date :/ /
EN:Visioned will repair or replace this product free of charge in the case of defective parts or manufacturing defects, in accordance with the conditions mentioned below as follows: DURATION:24 MONTHS RETURN TO WORKSHOP
LIMITS AND EXCLUSIONS: This guarantee concerns only the original final purchaser. A purchase invoice, or another proof of purchase, with this guarantee card will be required to obtain an after-sales service, in accordance with this guarantee. This guarantee card will not be extended to another person only the original final purchaser. This guarantee becomes void if the serial numbers on the product are modified, replaced, illegible, absent, or if repair has been carried out by a service not approved, including the user.
This guarantee covers only the defects of the material or parts, occurring during normal use of the product. It does not cover the damage caused during the transport of the apparatus, causes due to repairs being carried out by the distributor, by any modifications undertaken, any connection of equipment not approved by Visiomed, or causes contrary to those written in the user manual or notice. Moreover, the present guarantee does not cover damage due to falls, bad handling, bad installations, damage by fire, floods, lightning, or any other natural disaster. This guarantee does not cover the packing of the material, the accessories, the defects caused by commercial exposure of the product, show room, sale space, demonstration etc... Normal maintenance, cleaning and the replacement of parts where wear is normal, are not covered by the terms of this guarantee. Visiomed and its representatives and agents will not in any case be held responsible for any damage and consecutive damages due to the mishandling of this product. This guarantee is the only valid one at Visiomed*, any other guarantee (commercial guarantee) except this one will not be taken into account.
IMPORTANT: During the guarantee period if you are dissatisfied with the repairs of this product, please contact the Visiomed customer service.

EN: Disposal of Waste Equipment by Users in Private Households in the European Union.
For more information about where you can drop off your waste equipment for recycling, please contact your local city office or your household waste disposal service.
07/2017 KD-798
EN: TensioFlash is a brand registered by Visiomed Group SA. The constructor keeps the right to modify the photographs and characteristics without previous notice. Registered design. The logos and trademarks are the property of their respective owners.
