AirCurve 10 ST-A - Medical device ResMed - Free user manual and instructions
Find the device manual for free AirCurve 10 ST-A ResMed in PDF.
Download the instructions for your Medical device in PDF format for free! Find your manual AirCurve 10 ST-A - ResMed and take your electronic device back in hand. On this page are published all the documents necessary for the use of your device. AirCurve 10 ST-A by ResMed.
USER MANUAL AirCurve 10 ST-A ResMed
RESMED ResMed Confidential Proprietary Information, not to be reproduced or made available to third parties without prior consent from ResMed and not to be used in any unauthorised way. DOC NO 288156 © ResMed Ltd 2018. PAGE 1 of 134
SPECIFICATION - PRINTED MATERIALS REV 1
Rev Change Date Document prepared by Document Checked by Note document (Name) (Name) drafted 1 K001608-00 24 July 2018 Melanie Theaker Lisa Oliver or delegate refer to change note. AirCurve 10 ST-A User Guide AMER Multi
Refer to the Change History on the English source listed for summary of changes in each version. English source This translation replaces… 1 288009r4 plus changes shown | 288131 below
Print according to master spec R370-7240
4. RESMED QC INSPECTION
For general sampling and inspection requirements, refer to AW1203-002 Appendix A, Printed or Unprinted Materials and Labels, Criteria 1, 2, 3, 4,5, 6, 8 and 9 Additional requirement a) None
| Sect n Update Tech Update of Wireless approval certificate id to add information for 4G Specs | Ecc ID: 2ACHL-AIR104G
IC ID: 9103A-AIR104G
Tech Update of Classification to be IEC 60601-1:2005+A1:2012 Specs Tech Update of Wireless information to match Machine data for Tech comms rev Specs 65 Cleaning | Add warning ‘Regularly clean the tubing assembly, water tub and mask for optimal therapy and to prevent the growth of germs that can adversely the patient's health.
ENGLISH Welcome The AirCurve"” 10 ST-A is a bilevel positive airway pressure device. À WARNING Read this entire guide before using the device. À CAUTION In the US, Federal law restricts this device to sale by or on the order of a physician. Indications for use AirCurve 10 ST-A The AirCurve 10 ST-A is indicated to provide non-invasive ventilation for patients weighing more than 30 Ib (13 kg) with respiratory insufficiency or obstructive sleep apnea (OSA) The iVAPS mode is indicated for patients weighing more than 66 Ib (30 kg). The AirCurve 10 ST-A is intended for home and hospital use The humidifier is intended for single patient use in the home environment and re-use in à hospital/institutional environment. Contraindications Positive airway pressure therapy may be contraindicated in some patients with the following pre- existing conditions: + severe bullous lung disease +" pneumothorax or pneumomediastinum + _pathologically low blood pressure, particularly if associated with intravascular volume depletion + dehydration + _cerebrospinal fluid leak, recent cranial surgery, or trauma. Adverse effects You should report unusual chest pain, severe headache, or increased breathlessness to your prescribing physician. An acute upper respiratory tract infection may require temporary discontinuation of treatment. The following side effects may arise during the course of therapy with the device + drying of the nose, mouth, or throat + nosebleed + bloating + _ear or sinus discomfort + eveirritation + skin rashes English
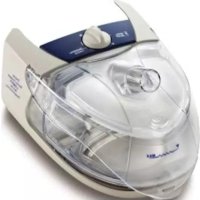
At a glance The AirCurve 10 includes the following: + Device with HumidAir” integrated humidifier + Water tub + Air tubing + Power supply unit + Travelbag + SD card (already inserted). Contact your care provider for a range of accessories available for use with the device including: + Air tubing (heated and non-heated): ClimateLineAir”, ClimateLineAir Oxy, SlimLine”", Standard +. Water tub: Standard water tub, cleanable water tub (can be disinfected) + Side cover for use without the humidifier + Filter: Hypoallergenic filter, standard filter + Air10" DC/DC converter (12V/24V) + SD card reader + Air10 oximeter adapter + Air10 USB adapter + Power Station Il + Air10 tubing elbow
About your device Aïr outlet Aïr filter cover Retention clip Power inlet Serial number and device number um 8 w N = About the control panel GSM Stop button O Dial CRD Home button HumidAir humidifier (water tub) Screen Adapter cover SD card cover
© LED alarm indicator Press to start/stop therapy. Press and hold for three seconds to enter power save mode. Turn to navigate the menu and press to select an option. Turn to adjust a selected option and press to save your change. Press to return to the Home screen. Different icons may be displayed on the screen at different times including A FenpTime @ Humidity 4$$ Humidifier warming 3e Humidifier cooling
Alarm muted ]| Wireless signal strength (green) All al Wireless transfer not enabled (gray)
à No wireless connection D Aïplane Mode English
À CAUTION Do not overfill the water tub as water may enter the device and air tubing.
With the device on a stable level surface, grip the retention clip on the back of the device and pull up to open. Note: The retention clip is shown in the open position … (a) Plug the power connector into the device power inlet then {b} push down the rétention clip to secure in place. Connect one end of the power cord into the power supply unit and the other end into the power outlet. . Connect the air tubing firmly to the air outlet located on the rear of the device. Open the water tub and fill it with distilled water up to the maximum water level mark. Do not fill the water tub with hot water. . Close the water tub and insert it into the side of the device. . Connect the free end of the air tubing firmly onto the assembled mask. See the mask user guide for detailed information. Recommended masks are available on www.resmed.com Note: Ensure the device is placed so that the LED alarm indicator is clearly visible.
is enabled You will know that therapy is on when the Monitoring screen is displayed The pressure bar shows the inspiratory and expiratory pressures in green The green bar will expand and contract as you breathe in and out The screen will go black automatically after a short period of time. You can press Home or the dial to turn it back on. If power is interrupted during therapy, the device will automatically restart therapy when power is restored The AirCurve 10 device has a light sensor that adjusts the screen brightness based on the light in the room Stopping therapy
1. Remove your mask.
2. Press Start/Stop or if SmartStart is enabled, therapy will stop automatically after a few seconds.
Note: If Confirm Stop is enabled, a message is displayed asking if you want to stop therapy. Turn the dial to select Yes and then press the dial to stop therapy Once therapy has stopped, the Sleep Report gives you a summary of your therapy session Usage hours-Indicates the number of hours of therapy you received last session Mask Seal-Indicates how well your mask sealed: < Home
rar 7:15 © Good mask seal Mask al © Qi adjusting, see Mask Fit. Humidifier: & Humidifier-Indicates if your humidifier is working properly: © Humiciior Working @riciier might be faulty, contact your care provider. If set by your care provider, you will also see Events per hour-Indicates the number of apneas and hypopneas experienced per hour. More Info-Turn the dial to scroll down to view more detailed usage data English 5
Power save mode Your AirCurve 10 device records your therapy data. In order to allow it to transmit the data to your care provider, you should not unplug the device. However, you can put it into power save mode to save electricity. To enter power save mode: +_ Press and hold Start/Stop for three seconds. The screen goes black. To exit power save mode: +_ Press Start/Stop once. The Home screen is displayed. My Options Your AirCurve 10 device has been set up for your needs by your care provider, but you may find you want to make small adjustments to make your therapy more comfortable. Highlight My Options and press the dial to see your current settings. From here, you can personalize your options: Humidity Level 4 My Options. Mask Full Face ( Run Mask Fit > PNY, Run Warm Up > Sleep Report Airplane Mode Off Ramp Time Designed to make the beginning of therapy more comfortable, Ramp Time is the period during which the pressure increases from a low start pressure to the prescribed treatment pressure. You can set your Ramp Time to Off or between 5 to 45 minutes. My Options To adjust Ramp Time: < Hom 1. In My Options, turn the dial to highlight Ramp rem om Time and then press the dial. Humidiy Level 4 2. Turn the dial to adjust the ramp time to your preferred setting and press the dial to save the Mask Ful Fac change. Run Mask Fit > Run Warm Up > Airplane Mode Off
Ramp Down Ramp Down is intended to make stopping therapy more comfortable by reducing your pressure over a fixed 15 minute period. This option will only be available to you via your care provider To enable Ramp Down:
1. In My Options, turn the dial to highlight Ramp Down and then press the
RempTime 20min. dial Pr]. 2. Turn the dial to select On and then press the dial to save the change. Ramp Down CECI. To start Ramp Down: Mask MES) 1. Press the Start/Stop button Tube Standard Run Mask Fit > Note: If Confirm Stop is enabled, a message is displayed asking if you want to start Ramp Down: Turn the dial to select Yes and then press the dial to start Ramp Down. The Ramp Down icon D and time remaining wil be displayed at the bottom left of the screen Once Ramp Down is complete, the device will continue to run at low pressure. To stop therapy at any time, press Start/Stop Humidity Level The humidifier moistens the air and is designed to make therapy more comfortable. If vou are getting à dry nose or mouth, turn up the humidity. If you are getting any moisture in your mask, turn down the humidity. You can set the Humidity Level to Off or between 1 and 8, where 1 is the lowest humidity setting and 8 is the highest humidity setting. To adjust the Humidity Level: < Home 1. In My Options, turn the dial to highlight Humidity Expo An Level and then press the dial.
2. Turn the dial to adjust the humidity level and press
the dial to save the change. Humidity Level 4 Mask Full Face Run Mask Fit Run Warm Up Airplane Mode Off If you continue to get a dry nose or mouth, or moisture in your mask, consider using ClimateLineAir heated air tubing. ClimateLineAir together with Climate Control delivers more comfortable therapy, English 7
Mask Fit Mesk Fit is designed to help you assess and identify possible air leaks around your mask My Options To check Mask Fit:
1. Fitthe mask as described in the mask user guide.
2. In My Options, turn the dial to highlight Run Mask Fit and then press the
dial The device starts blowing air pa 3. Adjust the mask, mask cushion and headgear until you get a Good result. Stop Mask Fit À Mask Fit To stop Mask Fit, press the dial or Start/Stop. If vou are unable to get a good mask seal, talk to your care provider. More options There are some more options on your device which you can personalize Mask This option shows your mask type setting. If you use more than one type of mask, adjust this setting when switching between masks Run Warm Up This option allows you to pre-heat the water before starting therapy, so that the air is not cold or dry at the beginning of therapy. Ramp Down* This option is intended to make stopping therapy more comfortable by reducing your pressure over a fixed 15 minute period. SmartStart* When SmartStart is enabled, therapy starts automatically when you breathe into your mask. When you remove your mask, it stops automatically after few seconds. “When enabled by your are provider Working with alarms The device is fitted with an alarm feature that monitors your therapy and alerts you to changes that may affect your treatment. When power is connected to the device, the yellow LED alarm indicator will flash and the alarm will sound to confirm that the alarm is working. When an alarm is activated, the yellow LED alarm indicator will flash, the Monitoring ù alarm will sound and a message will appear on the screen Leak detecter, check your system setup and all ‘connections.
Muting activated alarms To mute the alarm:
1. Press the dial. A list of activated alarms will appear and the flashing alarm
mute icon ZX will be displayed in the top right corner of the screen The alarm will be muted for 2 minutes.
2. To retum to the previous screen, highlight OK and press the dial
Once the condition that activated the alarm is corrected, the alarm sound and flashing icon will stop. If the condition that activated the alarm remains after 2 minutes, the alarm will re-occur See the Alarms troubleshooting section for help with managing common alarm conditions Multiple alarms If multiple alarms are activated at the same time, the most recent alarm message will be displayed on the screen and any other activated alarms will be shown in the Alarms list. Monitoring AN Alarm To view the alarm list:
1. From the Monitoring screen, turn the dial clockwise until the last
Monitoring screen is displayed
2. To view the alarm details, highlight the alarm and press the dial.
Low MV Unmute Alarms Caring for your device Itis important that you regularly clean your AirCurve 10 device to make sure you receive optimal therapy. The following sections will help you with disassembling, cleaning, checking and reassembling your device: À WARNING Regularly clean the tubing assembly, water tub and mak for optimal therapy and to prevent the growth of germs that can adversely affect the patient's health. English 9
1. Hold the water tub at the top and bottom, press it gently and pull it away from the device.
2. Open the water tub and discard any remaining water.
3. Hold the cuff of the air tubing and gently pull it away from the device.
Grip the retention clip and pull up to release the power cord.
4. Hold both the cuff of the air tubing and the swivel of the mask, then gentiy pull apart.
Cleaning You should clean the device weekly as described. Refer to the mask user guide for detailed instructions on cleaning your mask
1. Wash the water tub and air tubing in warm water using mild detergent.
Do not wash in a dishwasher or washing machine.
2. Rinse the water tub and air tubing thoroughly and allow to dry out of direct sunlight and/or heat.
3. Wipe the exterior of the device with a dry cloth.
Checking You should regularly check the water tub, air tubing and the air filter for any damage.
1. Check the water tub:
+ Replace it if it is leaking or has become cracked, cloudy or pitted + Replace it if the seal is cracked or tom. + Remove any white powder deposits using a solution of one part household vinegar to 10 parts water.
2. Check the air tubing and replace it if there are any holes, tears or cracks
3. Check the air fier and replace it at least every six months. Replace it more often if there are any
holes or blockages by dirt or dust.
To replace the air filter:
1. Open the air filter cover and remove the old air filter.
The air filter is not washable or reusable.
2. Place a new air filter onto the air filter cover and then close it.
Make sure the air filter is fitted at all times to prevent water and dust from entering the device. Reassembling When the water tub and air tubing are dry, you can reassemble the parts.
1. Connect the air tubing firmly to the air outlet located on the rear of the device.
2. Open the water tub and fil it with distilled room temperature water up to the maximum water
3. Close the water tub and insert it into the side of the device.
4. Connect the free end of the air tubing firmly onto the assembled mask.
Therapy data Your AirCurve 10 device records your therapy data for vou and your care provider so they can view and make changes to your therapy if required. The data is recorded and then transferred to your care provider wirelessiy, if wireless network is available, or via an SD card. Data transmission Your AirCurve 10 device has the capability of cellular communication so that your therapy data can be transmitted to your care provider to improve the quality of your treatment. lt also allows your care provider to update your therapy settings in à more timely manner or upgrade your device software to ensure you receive the best therapy possible. The data is usually transmitted after therapy has stopped. In order to make sure that your data is transferred: + _ Leave your device connected to the power outlet at all times. + Meke sure that it is not in Airplane Mode. + Check the Wireless signal strength icon ulll for adequate wireless coverage. Notes: + Therapy data might not be transmitted if you use it outside of the country or region of purchase. + Devices with cellular communication might not be available in all regions. Please be aware that within the wireless network, the availability and quality of the network may be affected by terrain, buildings, and the weather. Wireless communication depends on network availability. Coverage is not available everywhere and varies by service. English 11
SD card An altemative way for your therapy data to be transferred to your care provider is via the SD card. Your care provider may ask you to send the SD card by mail or to bring it in. When instructed by vour care provider, remove the SD card. Do not remove the SD card from the device when the SD light is flashing because data is being written to the card. To remove the SD card:
1. Open the SD card cover.
2. Push in the SD card to release it. Remove the SD card from the device.
Place the SD card in the protective folder and send it back to your care provider. For more information on the SD card refer to the SD card protective folder provided with your device. Note: The SD card should not be used for any other purpose. Traveling You can take your AirCurve 10 device with you wherever you go. Just keep the following in mind: + Use the travel bag provided to prevent damage to the device. + Empty the water tub and pack it separately in the travel bag, + Make sure you have the appropriate power cord for the region you are traveling to. For information on purchasing, contact your care provider. + _lf you are using an external battery, you should turn off the humidifier in order to maximize the life of your battery. Do this by turning the Humidity Level to Off. Traveling by plane Your AirCurve 10 device may be taken on board as carry-on luggage. Medical devices do not count toward your carry-on luggage limit. You can use your AirCurve 10 device on a plane as it meets the Federal Aviation Administration {FAA) requirements. Air travel compliance letters can be downloaded and printed from Www.resmed.com. When using the device on a plane: + Make sure the water tub is completely empty and inserted into your device. The device will not work without the water tub inserted. + Turn on Airplane Mode.
My Optio To turn on Airplane Mode:
1. In My Options, turn the dial to highlight Airplane
Mode and then press the dial
2. Turn the dial to select On and then press the dial to
save the change. The Airplane Mode icon} is displayed at the top right of the screen Airplane Mode RempTime 20min. Le Humidity Level 4 Mask Full Face Run Mask Fit > Run Warm Up > Airplane Mode Of À CAUTION Do not use the device with water in the water tub on a plane due to the risk of inhalation of water during turbulence. Troubleshooting If you have any problems, have à look at the following troubleshooting topics. If you are not able to fix the problem, contact your care provider or ResMed. Do not try to open the device General troubleshooting Problem/possible cause Solution Airis leaking from around my mask Mask may be fited incorrect. Make sure your mask is fitted correctly. See your mask user guide for fitting instructions or use the Mask Fit function to check your mask fit and seal 1 am getting a dry or blocked nose Humidity level may be set 100 low Adjust the Humidity Level If you have ClimateLineAir heated air tubing, see the ClimateLineAir user guide 1 am getting droplets of water on my nose, in the mask and air tubing Humidity level may be set too high Adjust the Humidity Level If you have ClimateLineAir heated air tubing, see the ClimateLineAir user guide My mouth is very dry and uncomfortable Air may be escaping through your mouth Increase the Humidity Level You may need à chin strap to keep your mouth closed or a full face mask Air pressure in my mask seems too high (it feels like 1 am getting too much air) Ramp may be turned off Use the Ramp Time option. English 13
Problem/possible cause Solution Aïr pressure in my mask seems too low (it feels like 1 am not getting enough air) Ramp may be in progress A Ramp Down may be in progress: ne Non-vented mask is used. Mask vents might be blocked. Expiratory pressure (EPAP] may be set 100 low My screen is black Backlight on the screen may have turned off. Itturns off automatically after à short period of time. Power may not be connected I have stopped therapy, but the device is still blowing air Device is cooling down. My water tub is leaking Water tub may not be assembled correct. Water tub may be damaged or cracked My therapy data has not been sent to my care provider Power may not be connected Wireless coverage may be poor. The No wireless connection icon f] is displayed on the top right of the screen. no wireless network available: Wait for air pressure to build up or turn Ramp Time off Press Start/Stop to stop therapy then press Start/Stop to restart and continue therapy, Only use a vented mask Check if you have sufficient venting. Unblock mask vents if necessary. Talk to your care provider about your settings Press Home or the dial to turn it back on. Connect the power supply and make sure the plug is fully inserted. Note: The retention clip should be in the open position when inserting the plug. For instructions refer to the Setup section. Device blows a small amount of air in order to avoid condensation in the air tubing, lt will stop automatically after 30 minutes Check for damage and reassemble the water tub correct. Contact your care provider for a replacement. Connect the power supply and make sure the plug is fully inserted. Note: The retention clip should be in the open position when inserting the plug. For instructions refer to the Setup section. Make sure that the device is placed where there is coverage lie, on your bedside table, not in a drawer or on the floor) and away from other communication devices. The Wireless signal strength icon til indicates good coverage when al bars are displayed, and poor coverage when fewer bars are displayed. Make sure that the device is placed where there is coverage lie, on your bedside table, not in a drawer or on the floor) and away from other communication devices. If instructed to do so, send the SD card to your care provider. The SD card also contains your therapy data.
Problem/possible cause Solution Device may be in Airplane Mode. Data transfer is not enabled for your device. Tum off Airplane Mode, see Traveling by plane. Talk to your care provider about our settings. My screen and buttons are flashing but there is no alarm sound or message Software upgrade is in progress Software upgrade takes approximately 10 minutes to complete. Displays message: Read only card, please remove, unlock and re-insert SD card SD card switch may be in the lock {read-only) position Alarms troubleshooting Move the switch on the SD Card from the lock position À to the unlock position mf” and then re-insert it Problem/possible cause Solution Display disappears and an alarm is activated Power failure Power cord is disconnected or mains power has been turned off during therapy. Remove your mask until power is restored Ensure the power cord is connected and the mains power switch if available) is on. Displays message: High leak detected, check your water tub, tub seal or side cover Water tub may not be inserted propery. Water tub seal may not be inserted properiy. Make sure the water tub is correctly inserted. Open the water tub and make sure that the seal is correctiy inserted. Displays message: High leak detected, connect your tubing Air tubing may not be connected properly. Mask may be fited incorrect. Displays message: Tubing blocked, check your tubing Air tubing may be blocked Make sure the air tubing is firmiy connected at both ends Make sure your mask is fitted correctly. See your mask user guide for fitting instructions or use the Mask Fit function to check your mask fit and seal Check the air tubing and remove any blockages. Press the dial to clear the message and then press Start/Stop to restart the device. Displays message: Leak detected, check your system setup and all connections Mask may be fited incorrect. Displays message: Low MV detected Minute ventilation level has dropped below the pre-set alarm level. Make sure your mask is fitted correctly. See your mask user guide for fitting instructions or use the Mask Fit function to check your mask fit and seal Contact your care provider. English 15
Problem/possible cause Solution Displays message: Apnea detected The device detects an apnea that has exceeded the pre-set Breath normally to disable the alarm alarm Ifthe problem persists contact your care provider. Displays message: Low Sp0: detected SpOz has dropped below the pre-set alarm level Check the attachment of the sensor Ifthe problem persists contact your care provider. Displays message: No SpO data, check your oxi sensor attachment to module/finger Oximeter sensor is not attached properly. Ensure that the oximeter sensor is attached properly to the module and to your finger. Oximeter sensor may be faulty. Ifthe message appears repeatediy, the oximeter sensor might be faulty. Replace the oximeter. Displays message: Non-vented mask detected, use vented mask or unblock your mask vents Non-vented mask is used. Only use a vented mask Mask vents might be blocked. Check if you have sufficient venting. Unblock mask vents if necessary. Expiratory pressure (EPAP] may be set 100 low Talk to your care provider about your settings Displays message: System fault, refer to user guide, Error 004 Device may have been left in à hot environment Allow to col before re-use. Disconnect the power supply and then reconnect it to restart the device. Air filter may be blocked Check the air filter and replace it if there are any blockages. Disconnect the power supply and then recannect it to restart the device. Air tubing may be blocked Check the air tubing and remove any blockages. Press the dial to clear the message and then press Start/Stop to restart the device. There may be water in the air tubing Empty the water from the air tubing. Disconnect the power supply and then reconnect it ta restart the device. Displays message: System fault, refer to user guide, Error 022 Power cord may not be correctiy inserted into the device. Remove the power cord from the device and then re-insert it. Ensure that the power cord is fuly inserted into the device Note: the retention clip should be in the open position when inserting the plug. For instructions refer to the Setup section. Ifthe problem continues, contact your local ResMed dealer or ResMed office. Do not open the device. Al other error messages, for example, System fault, refer to user guide, Error XX An unrecoverable error has occurred on the device. Contact your care provider. Do not open the device
Reassembling parts Some parts of your device are designed to easily come off in order to avoid damage to the parts or the device. You can easily reassemble them as described below. To insert the water tub seal:
1. Place the seal into the lid
2. Press down along all edges of the seal until
To reassemble the water tub lid: itis firmiy in place
1. Insert one side of the lid into the pivot hole of the base.
2. Slide the other side down the ridge until it clicks into place.
General warnings and cautions À WARNING Make sure that you arrange the air tubing so that it will not twist around the head or neck. Make sure the power cord and plug are in good condition and the equipment is not damaged. Keep the power cord away from hot surfaces. If you notice any unexplained changes in the performance of the device, if it is making unusual sounds, if the device or the power supply are dropped or mishandled, or if the enclosure is broken, discontinue use and contact your care provider or your ResMed Service Center. Do not open or modify the device. There are no user serviceable parts inside. Repairs and servicing should only be performed by an authorized ResMed service agent. Beware of electrocution. Do not immerse the device, power supply or power cord in water. liquids are spilled into or onto the device, unplug the device and let the parts dry. Always unplug the device before cleaning and make sure that all parts are dry before plugging it back in. Supplemental oxygen must not be used while smoking or in the presence of an open flame. Always make sure that the device is turned on and airflow generated before the oxygen supply is turned on. Always turn the oxygen supply off before the device is turned off, so that unused oxygen does not accumulate within the device enclosure and create a risk of fire. Do not perform any maintenance tasks while the device is in operation. The device should not be used adjacent to or stacked with other equipment. If adjacent or stacked use is necessary, the device should be observed to verify normal operation in the configuration in which it will be used. The use of accessories other than those specified for the device is not recommended. They may result in increased emissions or decreased immunity of the device. Regularly check the antibacterial filter for signs of moisture or other contaminants, particularly during nebulization or humidification. Failure to do so could result in increased breathing system resistance. The device has not been tested or certified for use in the vicinity of X-ray, CT or MRI equipment. Do not bring the device within 13 ft (4 m) of X-ray or CT equipment. Never bring the device into an MR environment. Therapy settings should not be changed remotely for patients in a hospital setting. Remote changes in a hospital setting may not be appropriate for certain patients, as these setting changes may not be communicated to all hospital personnel treating the patient. Hospital staff should liaise with the patient’ regular care provider such that the desired therapy outcome is achieved. À CAUTION Use only ResMed parts and accessories with the device. Non-ResMed parts may reduce the effectiveness of the treatment and/or damage the device. Use only vented masks recommended by ResMed or by the prescribing doctor with this device. Fitting the mask without the device blowing air can result in rebreathing of exhaled air. Make sure that the mask vent holes are kept clear and unblocked to maintain the flow of the fresh air into the mask. Be careful not to place the device where it can be bumped or where someone is likely to trip over the power cord.
+ Blocking the air tubing and/or air inlet of the device while in operation could lead to overheating of the device. + Keep the area around the device dry, clean and clear of anything (eg, clothes or bedding) that could block the air inlet or cover the power supply unit. + Do not place the device on its side as water might get into the device. + _ incorrect system setup may result in incorrect mask pressure reading. Ensure the system is correctly set up. +_ Do not use bleach, chlorine, alcohol, or aromatic-based solutions, moisturizing or antibacterial soaps or scented oils to clean the device, the humidifier or air tubing. These solutions may cause damage or affect the humidifier performance and reduce the life of the products. + _ If you use the humidifier, always place the device on a level surface lower than your head to prevent the mask and air tubing from filling with water. + _ Leave the water tub to cool for ten minutes before handling to allow the water to cool and to make sure that the water tub is not too hot to touch. + _ Make sure that the water tub is empty before transporting the device. Note: The device is not intended to be operated by persons (including children) with reduced physical, sensory or mental capabilities without adequate supervision by a person responsible for the patient's safety. Technical specifications 90W power supply unit AC input range: 100-240V, 50-60Hz 1.0-1.5A, Class I 115, 400H2 1.5A, Class (nominal for aircrait use) DC output PA Er TEA Typical power consumption 53W (57VA) Peak power consumption: 104W (108VA) Environmental conditions Operating temperature HAE to 495°F (25°C to +35°C) Note: The air flow for breathing produced by this therapy device can be higher than the temperature of the room Under extreme ambient temperature conditions {104°F/40°C) the device remains safe: Operating humidity: 10 to 85% relative humidity, non-condensing Operating altitude: Sea level to 8,500 (2,591 mi air pressure range 1013 hPa to 738 hPa Storage and transport temperature: 49 to +140°F (-20°C to +60°C) Storage and transport humnidity. 5 to 95% relative humidity, non-condensing Electromagnetic compatibility The AirCurve 10 complies with all applicable electromagnetic compatibility requirements (EMC) according to IEC 60601-1- 2:2014, for residential, commercial and light industry environment. It is recommended that mobile communication devices are kept at least 1 m away from the device. For further details see the Guidance and manufacturer's declaration — electromagnetic emissions and immunity section: Classification: IEC 60601-1:2005+A1:2012 Class I (double insulation), Type BF, Ingres protection IP22 English 19
Sensors Pressure sensors: Internally located at device outlet, analog gauge pressure type, -5 to +45 cm HaO (-5 to +45 hPa) Flow sensor: Internally located at device inlet, digital mass flow type, -70 10 +180 L/min Maximum single fault steady pressure Device will shut down in the presence of a single fault if the steady state pressure exceeds: 30 cm HO (30 hPa) for more than 6 sec or 40 cm HO (40 hPa) for more than 1 sec. Sound Pressure level measured according to ISO 80601-2-70:2015 (CPAP mode) SlimLine: 25 dBA with uncertainty of 2 dBA Standard 25 dBA with uncertainty of 2 dBA SlimLine or Standard and humidification: 27 dBA with uncertainty of 2 dBA Power level measured according to 1S0 80601-2-70:2015 (CPAP mode): SlimLine: 33 dBA with uncertainty of 2 dBA Standard 33 dBA with uncertainty of 2 dBA SlimLine or Standard and humidification: 35 dBA with uncertainty of 2 dBA Declared dual-number noise emission values in accordance with ISO 4871:1996. Alarm volume settings Low (nominal 54 dBAÏ, Medium (nominal 60 dBA), High (nominal 73 dBA) Physical - device and water tub Dimensions {W x D x H} 10.04 x 5.91" x 4.57" 255 mm x 150 mm x 116 mm) Air outlet (complies with ISO 5356-1:2004): 22mm Weight (device and standard water tubl: 47.1 0211336 gl Weight (device and cleanable water tub): 47.1 02 (1336 gl Housing construction: Flame retardant engineering thermoplastic Water capacity To maximum fil line 380 mL. Standard water tub - material Injection molded plastic, stainless steel and silicone seal Cleanable water tub - material Injection molded plastic, stainless steel and silicone seal Temperature Maximum heater plate 1549F (68°C) Cutout 1659F (74%C) Maximum gas temperature: < 108% (< 41°C) Aïr filter Standard Material: Polyester non woven fiber Average arrestance: >75% for -7 micron dust Hypoallergenic: Material: Acrylic and polypropylene fibers in a polypropylene carrier Efficiency: >98% for -7-8 micron dust; >80% for -0.5 micron dust Aircraft use ResMed confirms that device meets the Federal Aviation Administration (FAA) requirements (RTCA/DO-160, section 21, category M for all phases of air travel.
Wireless module Technology used Frequencies Mex RF power output CDMA (USA only), 4G (USA and Canada) 36,
The AirCurve 10 device complies with FCC Rules and Industry Canada Rules. The AirCurve 10 device should be used at a minimum distance of 0.8" (2 cm) from the body during operation. Additional information regarding the FCC Rules and IC compliance for this device can be found on www.resmed.com/downloads/devices Operating pressure range
S, ST. T, PAC, IVAPS
CPAP Supplemental oxygen Maximum flow Pneumatic flow path Design life Device, power supply unit Cleanable water tub: Standard water tub, air tubing General The patient is an intended operator. Operator position 3 to 30 cm H:0 4 to 20 cm H:0 15 L/min {S, ST, T, PAC, CPAP), 4 L/min (iVAPS)
6 months The device is designed to be operated within arm's length. An operator should position their line of sight within an angle of 30 degrees from a plane perpendicular to the screen. English
2 85 go 25 85 go 30 85 go AH - Absolute Humidity in mg/L 2 BTPS - Body Temperature Pressure Saturated Air tubing Air tubing Material ClimateLineAir Flexible plastic and electrical components ClimateLineAir Oxy Flexible plastic and electrical components SlimLine Flexible plastic Standard Flexible plastic 3m Flexible plastic Heated air tubing temperature cut-out: < 106°F (< 41°C) Notes: Nominal system output AH', BTPS2 Setting 4 Setting 8 6 >10 6 >10 6 >10 6 >10 6 >10 6 >10 Length Inner diameter 66° (2m) 0° (15 mm) 64 (19m) 0.75" (19 mm) 6' (1.8 m) 0.6 (15 mm) 66° (2m) 0.75" (19 mm) 910" (3 m) 0.75" (19 mm) + The manufacturer reserves the right to change these specifications without notice + The electrical connector end of the heated air tubing is only compatible with the air outlet at the device end and should not be fitted to the mask + Do not use electrically conductive or antistatic air tubing + The temperature and relative humidity settings displayed are not measured values. Displayed values Value Range Pressure sensor at air outlet: Mask pressure 3-30 cm H20 Flow derived values: Leak 0-120 L/min Tidal volume 0-4000 mL Respiratory rate 0-50 BPM Minute ventilation 0-30 L/min Ti 0.1-4.0 sec LE ratio 1:100-2:1 Display resolution
Value Accuracy" Pressure measurement! Mask pressure? #[0.5 cm H:0 (0.5 hPa) + 4% of measured value] Flow and flow derived values" Flow +6 L/min or 10% of reading, whichever is greater, at O to 150 L/min positive flow Leak? +12 L/min or 20% of reading, whichever is greater, O to 60 L/min Tidal volume? 420% Respiratory rate2 41.0 BPM Minute ventilation’? 420% l'Results are expressed as STPD [Standard Temperature and Pressure, Dry. 101.kPa at an aperating temperature of 68°F (20°C), dry 2 Accuracy may be reduced by the presence of leaks, supplemental oxygen, tidal volumes <100 mL ar minute ventilation <3 L/min Measurement accuracy verfied as per ISO 10651-6:2004 for Home Care Ventiatary Support Devices {Figure 101 and Table 101) using naminal ResMed mask vent flous. Measurement system uncertainties In accordance with 1S0 80601-2-70:2015 the measurement uncertainty of the manufactures test equipment is: For measures of flow #15 L/min or + 2.7% of reading [whichever is greater) For measures of volume (< 100 mL} +5 mLor 6% of reading [whichever is greater) For measures of volume (> 100 mL} +20 mL or 3% of reading (whichever is greater} For measures of pressure + 0.15 cm H20 (0.15 hPa} For measures of time +10ms Pressure accuracy - CPAP Maximum static pressure variation at 10 cm H20 (10 hPa) according to ISO 80601-2-70:2015 Standard air tubing SlimLine air tubing Without humidification + 0.5 cm HO [+ 0.5 hPa) 205 cm HO (4 0.5 hPa) With humidification +05 cm HO [+ 0.5 hPa) 205 cm HO (4 0.5 hPa) Maximum dynamic pressure variation according to ISO 80601-2-70:2015 Device without humidification and Standard air tubing / Device with humidification and Standard air tubing Pressure [cm H20 (hPa)] 10 BPM 15 BPM 20 BPM 4 05/05 05/05 08/08 8 05/05 05/05 08/08 12 05/05 05/05 08/08 16 05/05 05/05 08/08 20 05/05 05/05 08/08 Device without humidification and SlimLine air tubing / Device with humidification and SlimLine air tubing Pressure [cm H20 (hPa)] 10 BPM 15 BPM 20 BPM 4 05/05 05/05 08/08 8 05/05 05/05 08/08 12 05/05 05/05 08/08 16 05/05 05/05 08/08 20 05/05 05/05 08/08 English
Flow (maximum) at set pressures The following are measured accordingly to ISO 80601-2-70:2015 at the end of the specified air tubing Pressure AirCurve 10 and AirCurve 10, AirCurve 10 and AirCurve 10, cm H20 (hPa) Standard humidification and SlimLine humidification and Lmin Standard L/min ClimateLineAir Lmin LUmin
Guidance and manufacturer’s declaration electromagnetic emissions and immunity Medical electrical equipment needs special precautions regarding EMC and needs to be installed and put into service according to EMC information provided in this document. The AirCurve 10 device has been designed to meet EMC standards. However, should you suspect that the device performance (eg, pressure or flow) is affected by other equipment, move the device away from the possible cause of interference: Guidance and manufacturer's declaration —electromagnetic emissions The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that the device is used in such an environment. Emissions test Compliance Electromagnetic environment — guidance RF emissions CISPR 11 Group 1 The device uses RF energy only for its internal function. Therefore, its RF emissions are very low and are not likely to cause any interference in nearby electronic equipment. RF emissions CISPR 11 Class B The device is suitable for use in all establishments, including domestic establishment and those directly connected to the public low-voltage network that supplies buildings used for domestic purposes Harmonic Emissions Class A
Voltage Fluctuations/Flicker Emissions Complies
Guidance and manufacturer’s declaration - electromagnetic immunity The device is intended for use in the electromagnetic environment specified below. The customer or the user of the device should assure that the device is used in such an environment. Immunity test IEC6O60-1-2test Compliancelevel Electromagnetic environment — guidance level Electrostatic #6 KVcontact 28 KV contact Floors should be wood, concrete or ceramic tie. I discharge (ESD) +8 KV air 215 KV air floors are covered with synthetic material, the relative IEC 61000-4-2 humidity should be at least 30% Electrical fast 42 KV for power 22W Mains power quality should be that of a typical transient/burst supply lines commercial or hospital environment. IEC6100044 +1 KV forinput/output +1 KV for input/output lines lines English 25
Immunity test 1EC60601-1-2test Compliance level Electromagnetic environment — guidance level Surge AT KV Genet 21 KVdierentia Mains power quality shouldbe that or a tpical IC6100025 mode mode commercial or hospital environment. 42V common mode #2 KV common mode Voltage dips,… <5% Ut(>95% dipin 100V Mains power quality should be that of a typical short Ut for 0.5 cycle commercial or hospital environment interruptions 40% Ut(60% dipin 240V I the user of the device requires continued operation and voltage Ut)for5 cycles during power mains interruptions, its recommended variations on 7096 Ut (30% dipin that the device be powered from an uninteruptible power supply Ut for 25 cycles power source input lines , IEC 61000-4-11 Dose” dipin Power 3A/m 30 Am Power frequency magnetic fields should be at levels frequency characteristi of à typical location in a typical (50/60 Hz) commercial or hospital environment magnetic field
Conducted RE 3 Vrms 3Vims Portable and mobile RF communications equipment IC6100026 150kHzto80 MH? 10 kHz to 80 MHz should be used no closer to any part of the device, including cables, than the recommended separation distance calculated from the equation applicable to the frequency of the transmitter RadiatedRF 3 V/m 10m Recommended separation distance IC6100043 8OMHio25GH2 BOMHo25GH2 d-0.35ÿP d=0.35 ÿP 80 MHz to 800 MHz d=0.70 /P 800 MHz to 2.5 GHz Where (P) is the maximum output power rating of the transmitter in watts (W) according to the transmitter manufacturer and d is the recommended separation distance in meters (m). Field strengths from fixed RF transmitters, as determined by an electromagnetic site survey, # should be less than the compliance level in each frequency range. ? Interference may occur in the vicinity of equipment marked with the following symbol: (4? 2Field strengths from fixed transmitters, such as base stations for radio [cellular/cordless) telephones and land mobile radios, amateur radio, AM and FM radio broadcast and TV broadcast cannot be predicted theoretically with accuracy. To assess the electromagnetic environment due to fixed RF transmitters, an electromagnetic site survey should be considered. If the measured field strength in the location in which the device is used exceeds the applicable RF compliance level above, the device should be observed to verify normal operation. If abnormal performance is observed, additional measures may be necessary, such as reorienting or relocating the device. “Over the frequency range 150 kHz to 80 MHz, field strengths should be less than 3 V/m. Notes: + Utis the AC mains voltage prior to application of the test level. + At 80 MHz and 800 MHz, the higher frequency range applies. + These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people:
Recommended separation distances between portable and mobile RF communications equipment and the device The device is intended for use in an environment in which radiated RF disturbances are controlled. The customer or the user of the device can help prevent electromagnetic interference by maintaining à minimum distance between portable and mobile RF communications equipment {transmitters) and the device as recommended below, according to the maximum output power of the communications equipment. Rated maximum output Separation distance according to frequency of transmitter (m} power oftransmitter 150 kHz to 80 MHz 80 MHz to 800 MHz 800 MHz to 2.5 GHz (wi d=0.35 ÿP d= 0.35 P d=07ÿP
For transmitters rated at a maximum output power not listed above, the recommended separation distance din metres (m) can be determined using the equation applicable to the frequency of the transmitter, where P is the maximum output power rating of the transmitter in watts (W] according to the transmitter manufacturer. Notes: + At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies. + These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people Symbols The following symbols may appear on the product or packaging, @ Read instructions before use. À Indicates a warning or caution. [I Follow instructions before use. sl Manufacturer. [ECIREP] European Authorized Representative. Batch code. [REF] catalog number. [SN ] Serial number. [DN ] Device number. () On / off. À Device weight 1P22 Protected against finger sized objects and against dripping water when tilted up to 15 degrees from specified orientation. 2 Direct current. Type BF applied part [LI] class 11 equipment. &° Humidity limitation. sl” Temperature limitation. (£) Non-ionising radiation: & China pollution contra! logo 1. ® @ China pollution control logo 2. RX Only Prescription only {in the US, Federal law restricts these devices to sale by or on the order of a physician) AAMAX maximum water level. (1 use distilled water only. © Operating altitude 26% Atmospheric pressure limitation. @ Complies with RTCA DO-160 section 21, category M. € MR unsafe {do not use in the vicinity of an MRI device} English 27
== Environmental information This device should be disposed of separately, not as unsorted municipal waste. To dispose of your device, you should use appropriate collection, reuse and recycling systems available in your region. The use of these collection, reuse and recycling systems is designed to reduce pressure on natural resources and prevent hazardous substances from damaging the environment If you need information on these disposal systems, please contact your local waste administration: The crossed-bin symbol invites you to use these disposal systems. If you require information on collection and disposal of your ResMed device please contact your ResMed office, local distributor or go to www.resmed.com/environment. Servicing The AirCurve 10 device is intended to provide safe and reliable operation when operated in accordance with the instructions provided by ResMed. ResMed recommends that the AirCurve 10 device be inspected and serviced by an authorized ResMed Service Centre if there is any sign of wear or concern with device function. Otherwise, service and inspection of the products generally should not be required during their design life Limited warranty ResMed Ltd (hereafter 'ResMed') warrants that your ResMed product shall be free from defects in material and workmanship from the date of purchase for the period specified below. Product Warranty period + Mask systems [including mask frame, cushion, headgear and tubing}—excluding 90 days Single-use devices + Accessories —excluding single-use devices + Flex:type finger pulse sensors + Humidifier water tubs + Batteries for use in ResMed internal and external battery systems 6 months + Clip-type finger pulse sensors 1 year + CPAP and bilevel device data modules + Oximeters and CPAP and bilevel device oximeter adapters + Humidifier cleanable water tubs + Titration control devices + CPAP, bilevel and ventilation devices [including external power supply units) 2 years + Humidifiers + Battery accessories + Portable diagnostic/screening devices This warranty is only available to the initial consumer. It is not transferable. If the product fails under conditions of normal use, ResMed will repair or replace, at its option, the defective product or any of its components This Limited Warranty does not cover: a) any damage caused as a result of improper use, abuse, modification or alteration of the product; bl repairs carried out by any service organization that has not been expressly authorized by ResMed to perform such repairs; and c) any damage or contamination due to cigarette, pipe, cigar or other smoke. Warranty is void on product sold, or resold, outside the region of original purchase.
Warranty claims on defective product must be made by the initial consumer at the point of purchase This warranty replaces all other expressed or implied warranties, including any implied warranty of merchantability or fitness for a particular purpose. Some regions or states do not allow limitations on how long an implied warranty lasts, so the above limitation may not apply to you: ResMed shall not be responsible for any incidental or consequential damages claimed to have resulted from the sale, installation or use of any ResMed product. Some regions or states do not allow the exclusion or limitation of incidental or consequential damages, so the above limitation may. not apply to you: This warranty gives you specific legal rights, and you may also have other rights which vary from region to region. For further information on your warranty rights, contact your local ResMed dealer or ResMed office Further information If you have any questions or require additional information on how to use the device, contact your care provider. English 29
 ManualGo.com
ManualGo.com